Advanced Aqueous Phase Synthesis of Probenecid for Commercial Scale-Up and Purity Optimization
Advanced Aqueous Phase Synthesis of Probenecid for Commercial Scale-Up and Purity Optimization
The pharmaceutical industry is constantly seeking robust manufacturing pathways that balance high purity with environmental sustainability, and the technology disclosed in patent CN103613521A represents a significant leap forward in the production of Probenecid. This specific intellectual property outlines a novel aqueous phase synthesis method that fundamentally restructures the traditional chemical approach by utilizing water as the primary reaction medium instead of volatile organic solvents. By shifting the paradigm from oxidation-based routes to a diazotization and sulfonyl chlorination sequence, this method addresses critical pain points regarding waste management and operator safety that have long plagued the manufacturing of this anti-gout agent. For R&D directors and procurement specialists evaluating long-term supply contracts, understanding the mechanistic advantages of this water-phase technology is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting modern regulatory standards. The following analysis dissects the technical merits of this patent to demonstrate how it enables cost reduction in API manufacturing while ensuring consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Probenecid has relied heavily on the oxidation of electron-poor aromatic rings, specifically utilizing p-toluenesulfonic acid or p-toluenesulfonamide as starting materials which require aggressive oxidizing agents. As illustrated in the traditional reaction pathway below, these legacy methods necessitate the use of potassium dichromate or potassium permanganate, introducing severe environmental liabilities due to the generation of heavy metal-containing wastewater that is difficult and expensive to treat.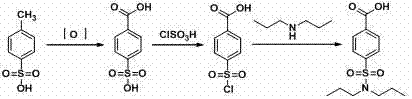 Furthermore, the reliance on organic solvents throughout the amidation and purification stages increases the risk of fire hazards and complicates the solvent recovery processes, leading to inflated operational expenditures. The presence of residual heavy metals such as chromium or manganese in the final product stream also poses a significant regulatory hurdle, requiring additional purification steps that inevitably lower the overall atom economy and extend the production cycle time. Consequently, many facilities utilizing these conventional oxidative routes face increasing pressure to suspend operations due to tightening environmental regulations and the prohibitive costs associated with hazardous waste disposal.
Furthermore, the reliance on organic solvents throughout the amidation and purification stages increases the risk of fire hazards and complicates the solvent recovery processes, leading to inflated operational expenditures. The presence of residual heavy metals such as chromium or manganese in the final product stream also poses a significant regulatory hurdle, requiring additional purification steps that inevitably lower the overall atom economy and extend the production cycle time. Consequently, many facilities utilizing these conventional oxidative routes face increasing pressure to suspend operations due to tightening environmental regulations and the prohibitive costs associated with hazardous waste disposal.
The Novel Approach
In stark contrast to the oxidative degradation of methyl groups, the innovative aqueous phase synthesis described in the patent data constructs the sulfonamide functionality through a constructive diazotization strategy starting from p-aminobenzoic acid. This approach completely bypasses the need for toxic heavy metal oxidants and minimizes the usage of organic solvents, thereby drastically simplifying the downstream purification workflow and reducing the environmental footprint of the manufacturing facility. By employing liquid sulfur dioxide and hydrochloric acid to generate the sulfonyl chloride reagent in situ, the process avoids the handling risks associated with highly corrosive reagents like chlorosulfonic acid or thionyl chloride while maintaining high reaction efficiency. This strategic shift not only enhances the safety profile of the plant but also improves the economic viability by reducing the consumption of expensive raw materials and lowering the energy requirements for solvent removal. For supply chain heads, this translates to a more resilient production capability that is less susceptible to disruptions caused by environmental compliance audits or raw material scarcity.
Mechanistic Insights into Diazotization and Sulfonyl Chlorination
The core of this technological breakthrough lies in the precise control of the diazotization reaction, where p-aminobenzoic acid is converted into a reactive diazonium salt under strictly regulated acidic conditions. The patent specifies maintaining the reaction temperature between -20°C and 20°C during the addition of sodium nitrite, a critical parameter that ensures the stability of the diazonium intermediate and prevents premature decomposition which could lead to phenolic by-products. This careful thermal management is essential for maximizing the conversion rate and ensuring that the subsequent sulfonyl chlorination step proceeds with high selectivity towards the desired para-substituted product. The use of water as the solvent in this stage also facilitates better heat dissipation compared to organic media, allowing for safer scaling of the exothermic diazotization process without the risk of thermal runaway. Understanding these kinetic constraints is vital for process chemists aiming to replicate this high-yield pathway in a commercial reactor setting.
Following the formation of the diazonium species, the introduction of liquid sulfur dioxide into the hydrochloric acid medium generates the active sulfonyl chlorinating agent which reacts efficiently to form p-carboxybenzenesulfonyl chloride. 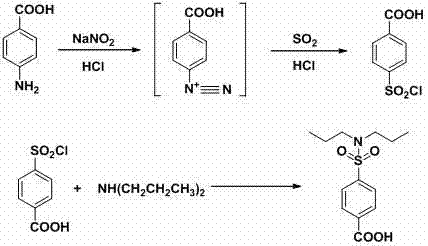 The subsequent reaction with dipropylamine is conducted in an aqueous environment at temperatures ranging from 15°C to 100°C, where the controlled batch addition of the sulfonyl chloride prevents localized exotherms and ensures uniform particle formation. This mechanistic pathway inherently suppresses the formation of polysubstituted impurities often seen in organic solvent systems, resulting in a crude product with purity levels exceeding 98% before final recrystallization. The elimination of transition metal catalysts means there is no need for expensive metal scavenging resins or complex chelation steps, further streamlining the isolation of the high-purity pharmaceutical intermediate. Such a clean reaction profile is indicative of a mature process ready for commercial scale-up of complex pharmaceutical intermediates.
The subsequent reaction with dipropylamine is conducted in an aqueous environment at temperatures ranging from 15°C to 100°C, where the controlled batch addition of the sulfonyl chloride prevents localized exotherms and ensures uniform particle formation. This mechanistic pathway inherently suppresses the formation of polysubstituted impurities often seen in organic solvent systems, resulting in a crude product with purity levels exceeding 98% before final recrystallization. The elimination of transition metal catalysts means there is no need for expensive metal scavenging resins or complex chelation steps, further streamlining the isolation of the high-purity pharmaceutical intermediate. Such a clean reaction profile is indicative of a mature process ready for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Probenecid Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and addition sequences outlined in the patent to achieve the reported yields of 85-91%. The process begins with the preparation of the diazotization liquid, followed by the generation of the sulfonyl chloride reagent, and concludes with the amidation step where the final molecular architecture is assembled. Operators must pay close attention to the dropping times, which are controlled between 3 to 5 hours for key reagent additions, to maintain the reaction equilibrium and prevent the accumulation of unstable intermediates. While the general framework is established, the specific operational parameters such as stirring rates and precise pH adjustments during acidification are critical for optimizing crystal morphology and filtration characteristics.
- Perform diazotization of p-aminobenzoic acid using hydrochloric acid and sodium nitrite at controlled low temperatures between -20°C and 20°C.
- Generate sulfonyl chloride reagent by passing liquid sulfur dioxide into dilute hydrochloric acid, then react with the diazotization product to form p-carboxybenzenesulfonyl chloride.
- React p-carboxybenzenesulfonyl chloride with dipropylamine in water at 15-100°C, followed by acidification and filtration to isolate pure Probenecid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this aqueous phase technology offers substantial benefits that directly impact the bottom line and supply chain reliability for global buyers. By eliminating the dependency on heavy metal oxidants and volatile organic solvents, manufacturers can significantly reduce the costs associated with hazardous waste treatment and environmental compliance reporting. This reduction in regulatory burden allows for a more streamlined operation where resources can be allocated towards capacity expansion rather than pollution control infrastructure. Furthermore, the simplified workup procedure reduces the overall cycle time per batch, enhancing the throughput capability of existing manufacturing assets without requiring major capital investment in new equipment. These factors collectively contribute to a more competitive pricing structure and a more stable supply of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of expensive heavy metal oxidants like potassium dichromate and the avoidance of organic solvents leads to a direct decrease in raw material procurement costs and waste disposal fees. Since the process utilizes water as the main medium, the energy consumption required for solvent recovery and distillation is drastically minimized, resulting in lower utility bills per kilogram of product. Additionally, the high atom utilization rate means less raw material is wasted as by-products, further optimizing the cost of goods sold and improving margin potential for long-term contracts. These efficiencies make the process economically superior to traditional oxidative routes that suffer from low yields and high remediation costs.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as p-aminobenzoic acid and liquid sulfur dioxide reduces the risk of supply disruptions caused by the scarcity of specialized oxidizing agents. The robustness of the water-based system also means that production is less sensitive to variations in ambient conditions, ensuring consistent output quality throughout the year. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing buyers to plan their inventory with greater confidence and safety stock levels. A reliable supplier utilizing this technology can guarantee continuous availability even during periods of strict environmental enforcement that might shut down conventional plants.
- Scalability and Environmental Compliance: The absence of toxic heavy metals in the wastewater stream simplifies the effluent treatment process, making it easier to scale production from pilot batches to multi-ton annual capacities without violating discharge limits. This environmental compatibility future-proofs the manufacturing site against increasingly stringent global regulations on chemical emissions and heavy metal residues. The simplified operational steps also reduce the training burden for plant personnel, minimizing the risk of human error during scale-up and ensuring that safety protocols are easily maintained. Such a sustainable approach aligns with the corporate social responsibility goals of major pharmaceutical companies seeking green chemistry partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this aqueous synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing supply chains.
Q: How does the aqueous phase method improve environmental compliance compared to traditional routes?
A: The aqueous phase method eliminates the use of heavy metal oxidants like potassium dichromate or potassium permanganate, thereby removing the burden of toxic heavy metal wastewater treatment and significantly reducing environmental pollution risks associated with chromium or manganese disposal.
Q: What are the purity and yield specifications achieved with this new synthetic route?
A: This optimized process achieves a product yield of 85-91% calculated based on p-aminobenzoic acid, with a final purity exceeding 98%, which meets the stringent standards required for pharmaceutical applications and reduces the need for extensive recrystallization.
Q: Does this process avoid the use of hazardous organic solvents and reagents?
A: Yes, the protocol utilizes water as the primary reaction medium and avoids toxic reagents such as thionyl chloride or chlorosulfonic acid in large quantities, replacing them with safer liquid sulfur dioxide and hydrochloric acid systems that simplify operational safety and storage requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Probenecid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the aqueous phase synthesis of Probenecid can be seamlessly transferred from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the highest international standards for identity, assay, and impurity profiles. Our commitment to technical excellence ensures that clients receive a high-purity Probenecid product that is fully compliant with pharmacopoeial requirements and ready for downstream formulation.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener manufacturing process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to build a sustainable and efficient supply chain for your anti-gout medication portfolio.
