Advanced Manufacturing of 3,4-Dimethoxyphenyl Acetonitrile for Cardiovascular API Production
The pharmaceutical industry's relentless pursuit of safer and more efficient synthetic routes for cardiovascular medications has brought significant attention to the manufacturing of key intermediates like 3,4-dimethoxyphenyl acetonitrile. As disclosed in patent CN101475511B, a novel methodology has been established that fundamentally alters the production landscape for this critical building block used in the synthesis of Verapamil Hydrochloride and Bevantolol. This patent introduces a robust three-step sequence involving decarboxylation, aldoxime formation, and dehydration, which collectively offer a superior alternative to legacy processes plagued by toxicity and inefficiency. For R&D directors and procurement strategists, understanding the nuances of this technology is essential for securing a reliable supply chain for high-purity pharmaceutical intermediates. The method not only addresses the urgent need for greener chemistry by eliminating hazardous cyanide salts but also demonstrates exceptional economic viability through simplified operational procedures and enhanced yield profiles.
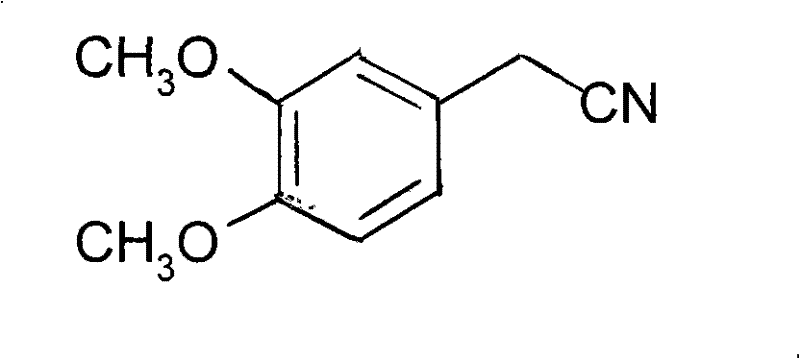
Traditionally, the synthesis of benzyl cyanides, including 3,4-dimethoxyphenyl acetonitrile, relied heavily on the nucleophilic substitution of haloalkanes with cyanide ions. This conventional approach, while chemically straightforward, presents formidable challenges in a modern regulatory environment due to the extreme toxicity of prussiate (cyanide) reagents. The handling of sodium cyanide requires specialized infrastructure, rigorous safety protocols, and costly waste treatment systems to neutralize lethal byproducts, all of which inflate the operational expenditure and extend lead times for production batches. Furthermore, side reactions such as hydrolysis or incomplete substitution often compromise the purity of the crude product, necessitating energy-intensive purification steps like rectification. In contrast, the novel approach detailed in the patent circumvents these pitfalls by utilizing a precursor derived from epoxy-potassium propionate. This strategic shift allows manufacturers to bypass the direct use of cyanide entirely, replacing it with a sequence that leverages abundant and benign reagents like hydroxylamine hydrochloride and potassium dihydrogen phosphate.
Mechanistic Insights into Decarboxylation and Phase-Transfer Catalyzed Dehydration
The core innovation of this synthesis lies in its elegant manipulation of functional group transformations to achieve the nitrile moiety without direct cyanation. The process initiates with the decarboxylation of 3-(3,4-dimethoxyphenyl)-2',3'-epoxy-potassium propionate in an aqueous medium buffered by KH2PO4. This step is critical as it generates the reactive 3,4-dimethoxy phenylacetaldehyde intermediate under mild thermal conditions, typically between 0°C and 40°C. The subsequent conversion to the aldoxime involves the reaction of this aldehyde with hydroxylamine hydrochloride in the presence of sodium bicarbonate, which acts as an acid scavenger to drive the equilibrium toward oxime formation. The final and perhaps most technically sophisticated step is the dehydration of the aldoxime to the nitrile. This is achieved using a strong base in conjunction with a phase-transfer catalyst (PTC) such as Tetrabutyl ammonium bromide (TBAB). The PTC facilitates the transport of hydroxide ions into the organic phase, promoting the elimination of water from the oxime group to form the carbon-nitrogen triple bond with high efficiency.
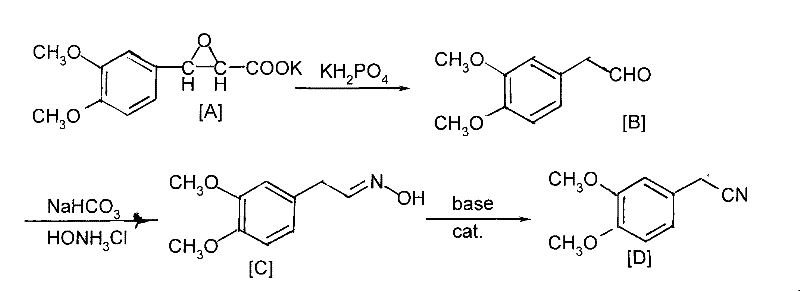
From an impurity control perspective, this mechanistic pathway offers distinct advantages over acid-catalyzed dehydration methods which often suffer from Beckman rearrangement side reactions leading to amide contaminants. By employing a basic dehydration system mediated by phase-transfer catalysis, the process selectively targets the removal of the hydroxyl group from the oxime without affecting the electron-rich dimethoxy aromatic ring. The patent data indicates that this specificity results in crude products with HPLC purity exceeding 99%, significantly reducing the burden on downstream purification units. Moreover, the use of water as a solvent in the initial decarboxylation step simplifies the workup procedure, as the organic product can be easily extracted into solvents like toluene or ethyl acetate. This biphasic system not only enhances reaction kinetics but also allows for the continuous removal of water during the final reflux stage, driving the dehydration equilibrium to completion and minimizing the formation of unreacted oxime residues.
How to Synthesize 3,4-Dimethoxyphenyl Acetonitrile Efficiently
The implementation of this synthetic route requires precise control over stoichiometry and reaction parameters to maximize the benefits outlined in the patent literature. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates and reducing solvent consumption. Operators must pay close attention to the molar ratios of the phase-transfer catalyst and the base in the final step, as these dictate the rate of dehydration and the overall yield. The following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility and safety at scale.
- Perform decarboxylation on 3-(3,4-dimethoxyphenyl)-2',3'-epoxy-potassium propionate in water using KH2PO4 to prepare 3,4-dimethoxy phenylacetaldehyde.
- Conduct an aldoxime reaction by adding sodium bicarbonate and hydroxylamine hydrochloride directly to the extracted organic solution.
- Execute dehydration using a phase-transfer catalyst and alkali under reflux with water division, followed by recrystallization in ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cyanide-free synthesis route translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of highly toxic cyanide salts removes a significant bottleneck in raw material sourcing and logistics, as the transportation and storage of prussiates are subject to stringent international regulations and security checks. By substituting these hazardous materials with stable solids like hydroxylamine hydrochloride and potassium dihydrogen phosphate, manufacturers can streamline their inventory management and reduce the risk of supply disruptions caused by regulatory compliance issues. Furthermore, the simplified waste profile of this process means that effluent treatment costs are substantially lowered, contributing to a more sustainable and economically resilient production model.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of expensive safety infrastructure required for cyanide handling. Without the need for specialized detoxification units for cyanide waste, capital expenditure for new facilities is reduced, and operating expenses for existing plants are minimized. Additionally, the patent claims that the synthetic cost is significantly lower than prior art methods, attributed to the high atom economy of the decarboxylation step and the use of inexpensive, commodity-grade reagents. The high purity of the final product also reduces the loss of material during recrystallization, ensuring that a greater proportion of the input mass is converted into saleable high-value intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available chemicals such as toluene, ethyl acetate, and common inorganic salts ensures that the supply chain remains robust against market volatility. Unlike specialized catalysts or restricted precursors, the reagents used in this process are produced globally in massive quantities, guaranteeing consistent availability and competitive pricing. This stability is crucial for long-term contract manufacturing organizations (CMOs) that need to guarantee delivery schedules to pharmaceutical clients. The ability to source materials locally in multiple regions further mitigates the risk of geopolitical disruptions affecting the continuity of API intermediate production.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated for industrial mass production with reaction conditions that are easily managed in standard stainless steel reactors. The use of water in the initial step and the ability to recycle organic solvents like toluene align perfectly with green chemistry principles, facilitating easier permitting and environmental compliance. The absence of heavy metals or persistent organic pollutants in the waste stream simplifies the discharge process, allowing facilities to operate with a smaller environmental footprint. This alignment with sustainability goals is increasingly becoming a prerequisite for partnerships with top-tier multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: How does this synthesis method improve safety compared to traditional cyanide substitution?
A: Traditional methods rely on toxic sodium cyanide for nucleophilic substitution, posing severe safety and environmental risks. This patent utilizes a cyanide-free pathway involving decarboxylation and aldoxime dehydration, significantly reducing hazardous waste handling requirements and improving operator safety profiles.
Q: What represents the key cost-saving advantage in this manufacturing process?
A: The process eliminates the need for expensive heavy metal catalysts and complex waste treatment associated with cyanide. Furthermore, the use of inexpensive reagents like potassium dihydrogen phosphate and common phase-transfer catalysts, combined with high yields exceeding 80% in the final step, drastically lowers the overall cost of goods sold.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial mass production. The reaction conditions are mild (0°C to 40°C for early steps), utilize water as a primary solvent for the initial step, and employ robust phase-transfer catalysis, ensuring scalability and consistent quality with HPLC purity reaching over 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dimethoxyphenyl Acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering 3,4-dimethoxyphenyl acetonitrile with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for GMP API synthesis. Our facility is equipped to handle the specific solvent systems and phase-transfer catalysis conditions described in advanced patents, guaranteeing a product that is free from toxic cyanide residues and ready for immediate downstream processing.
We invite you to collaborate with us to optimize your supply chain for Verapamil and related therapeutics. By leveraging our expertise in this specific synthetic route, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance the reliability and profitability of your pharmaceutical projects.
