Advanced Enantioselective Synthesis of Hemicaronic Aldehyde Derivatives for High-Performance Pyrethroids
Advanced Enantioselective Synthesis of Hemicaronic Aldehyde Derivatives for High-Performance Pyrethroids
The global demand for high-efficacy, low-toxicity agrochemicals has placed immense pressure on the supply chain for chiral intermediates, particularly those used in the synthesis of pyrethroids. Patent CN1020451C presents a groundbreaking enantioselective process for preparing derivatives of hemicaronic aldehyde with precise trans or cis structures. This technology addresses a critical bottleneck in the manufacturing of chrysanthemic acid analogues by providing a direct route to optically active compounds, bypassing the inefficiencies of traditional racemic synthesis. By leveraging chiral pool starting materials, this method ensures that the resulting intermediates possess the exact stereochemistry required for biological activity, thereby offering a reliable agrochemical intermediate supplier solution for manufacturers seeking to optimize their production lines.
The significance of this patent lies in its ability to control the stereochemistry at multiple centers simultaneously. Unlike older methods that often result in complex mixtures of diastereomers requiring extensive chromatographic separation, this process utilizes a defined sequence of reduction, olefination, and cyclopropanation to lock in the desired configuration early in the synthesis. This approach not only enhances the purity of the final product but also streamlines the overall workflow, making it an attractive option for cost reduction in agrochemical intermediate manufacturing. The following analysis delves into the mechanistic nuances and commercial implications of this sophisticated synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of hemicaronic aldehyde and related pyrethroid intermediates has historically relied on the preparation of racemic mixtures followed by resolution. This classical approach is inherently inefficient, as it theoretically caps the maximum yield at 50% for the desired enantiomer, with the remaining material often discarded or requiring energy-intensive recycling processes. Furthermore, the separation of diastereomers with similar physical properties often necessitates the use of preparative HPLC or multiple recrystallization steps, which drastically increases solvent consumption and waste generation. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, creating vulnerabilities in the supply chain for high-purity agrochemical intermediates.
Additionally, conventional routes often struggle with regioselectivity during the formation of the cyclopropane ring. Non-stereoselective cyclopropanation can lead to a mixture of cis and trans isomers, complicating the downstream synthesis of the final active ingredient. The presence of unwanted isomers can act as impurities that affect the stability and efficacy of the final pesticide formulation. Consequently, manufacturers face significant challenges in meeting stringent regulatory standards for impurity profiles, often requiring additional purification stages that further erode profit margins and delay time-to-market for new products.
The Novel Approach
The methodology described in CN1020451C revolutionizes this landscape by introducing an enantioselective pathway that begins with optically pure diesters derived from tartaric acid. By starting with a chiral scaffold, the synthesis inherently biases the formation of subsequent stereocenters, allowing for the targeted production of either the 1R-trans or 1R-cis configuration. This strategic use of chiral auxiliaries eliminates the need for late-stage resolution, effectively doubling the theoretical yield compared to racemic processes. For supply chain heads, this means a more predictable and robust production schedule with significantly reduced raw material overheads.
Moreover, the novel approach employs a versatile sequence of reactions that can be tuned to produce specific geometric isomers on demand. By selecting appropriate cyclopropanation agents—such as isopropylidene triphenylphosphorane for trans-structures or sulfurane reagents for cis-structures—the process offers unparalleled flexibility. This adaptability allows manufacturers to pivot quickly between different product grades based on market demand without retooling entire production lines. The result is a highly efficient, scalable process that aligns perfectly with the goals of commercial scale-up of complex agrochemical intermediates, ensuring a steady flow of high-quality materials to the formulation stage.
Mechanistic Insights into Enantioselective Cyclopropanation and Oxidative Cleavage
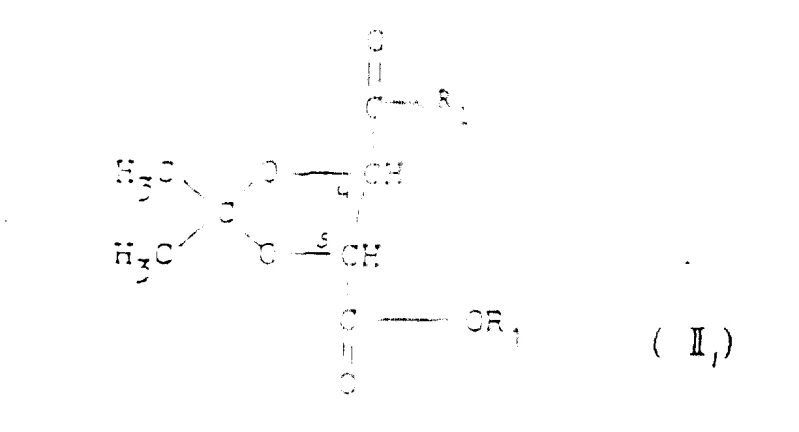
The core of this synthetic strategy involves a meticulously orchestrated series of transformations that preserve chirality while constructing the complex molecular architecture. The process initiates with the controlled reduction of a chiral diester (Formula II) using diisobutylaluminium hydride (DIBAL-H) at cryogenic temperatures (-78°C). This step is critical; the low temperature prevents over-reduction or epimerization, ensuring the quantitative formation of the corresponding dialdehyde (Formula III) with retained optical purity. The dialdehyde then serves as a pivotal intermediate, undergoing a Wittig-Emmons-Horner reaction with specific phosphonate esters to extend the carbon chain and introduce the necessary unsaturation for ring closure.
Following the olefination, the construction of the gem-dimethyl cyclopropane ring is achieved through reaction with specialized carbenoid equivalents. For the trans-configuration, the patent specifies the use of isopropylidene triphenylphosphorane, which reacts with the unsaturated diester to form the cyclopropane ring with high stereoselectivity. Conversely, for the cis-configuration, a sulfurane reagent is employed. This divergence in reagent choice highlights the precision of the method, allowing chemists to dictate the spatial arrangement of the methyl groups on the cyclopropane ring. The resulting intermediates (Formula V) contain a dioxolane protecting group which stabilizes the molecule during subsequent handling and purification steps.
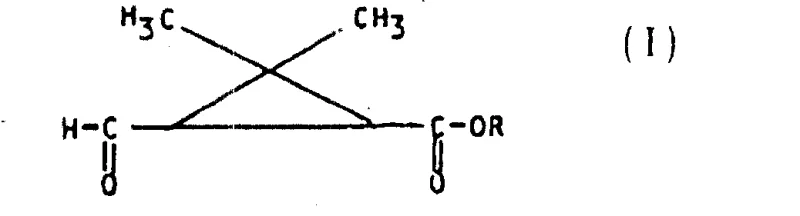
The final transformation involves the removal of the protecting group and the oxidative cleavage of the carbon-carbon double bond to reveal the aldehyde functionality. This is accomplished using strong oxidants such as sodium periodate or potassium permanganate, often in the presence of acid. This step effectively snaps the extended chain back to the aldehyde, yielding the target hemicaronic aldehyde derivative (Formula I). The mechanism ensures that the stereochemistry established in the earlier steps is preserved throughout the harsh oxidative conditions, delivering a product with exceptional enantiomeric excess. This rigorous control over the reaction pathway is what makes this technology a cornerstone for producing high-purity OLED material precursors or advanced pharmaceutical intermediates where chirality is paramount.
How to Synthesize Hemicaronic Aldehyde Efficiently
The synthesis of these valuable chiral intermediates requires strict adherence to the reaction conditions outlined in the patent to ensure optimal stereochemical outcomes. The process is designed to be robust yet sensitive to temperature and stoichiometry, particularly during the reduction and cyclopropanation phases. Operators must maintain an inert atmosphere and utilize anhydrous solvents to prevent side reactions that could compromise the yield or purity of the intermediates. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive roadmap for replicating this high-value chemistry in a pilot or production setting.
- Perform a controlled reduction of the chiral diester precursor (Formula II) using diisobutylaluminium hydride (DIBAL-H) at low temperatures (-78°C) to generate the corresponding dialdehyde (Formula III).
- React the dialdehyde with a Wittig-Emmons-Horner reagent (phosphonate ester) to extend the carbon chain and form the unsaturated diester intermediate (Formula IV) with specific geometric configuration.
- Execute a gem-dimethyl cyclopropanation reaction using a sulfurane or phosphorane reagent to construct the cyclopropane ring, followed by hydrolysis and oxidative cleavage (using periodate or permanganate) to yield the final hemicaronic aldehyde (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this enantioselective technology offers profound advantages that extend beyond simple chemical yield. By shifting from a resolution-based model to a direct asymmetric synthesis, companies can drastically simplify their manufacturing workflows. This simplification reduces the number of unit operations required, which in turn lowers capital expenditure on equipment and decreases the operational footprint of the production facility. The elimination of resolution steps also means a significant reduction in solvent usage and waste disposal costs, aligning with modern sustainability goals and environmental compliance standards.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the doubling of theoretical yield by avoiding the discard of unwanted enantiomers. In a racemic process, 50% of the material is essentially waste unless a complex recycling loop is established. This enantioselective route converts nearly all starting material into the desired isomer, leading to substantial cost savings on raw materials. Furthermore, the use of commodity chemicals like tartaric acid derivatives as chiral sources is far more economical than purchasing expensive chiral catalysts or resolving agents, driving down the overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Dependence on complex resolution processes often introduces bottlenecks, as the separation efficiency can fluctuate with batch quality. This new method relies on deterministic chemical transformations where the outcome is dictated by the stoichiometry and structure of the reagents rather than subtle physical differences. This predictability enhances supply chain reliability, ensuring consistent delivery schedules and reducing the risk of stockouts. Additionally, the starting materials are derived from the abundant chiral pool, mitigating the risk of supply disruptions associated with niche synthetic reagents.
- Scalability and Environmental Compliance: The reactions described, such as DIBAL-H reduction and Wittig olefination, are well-understood in industrial chemistry and can be scaled from kilogram to multi-ton quantities with established engineering controls. The process avoids the use of heavy metal catalysts often found in asymmetric hydrogenation, simplifying the purification of the final product and reducing the burden of heavy metal waste treatment. This cleaner profile facilitates easier regulatory approval and supports the production of green, eco-friendly agrochemicals that meet increasingly strict global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enantioselective synthesis. These answers are derived directly from the experimental data and claims within patent CN1020451C, providing clarity on the feasibility and scope of the technology. Understanding these details is crucial for R&D teams evaluating the integration of this pathway into their existing portfolios.
Q: What is the primary advantage of this enantioselective process over conventional racemic synthesis?
A: The primary advantage is the ability to directly synthesize specific enantiomers (such as 1R-trans or 1R-cis configurations) from chiral pool starting materials like tartrate derivatives. This eliminates the need for costly and inefficient resolution of racemic mixtures, significantly improving overall atom economy and reducing downstream purification costs.
Q: Which key reagents are critical for maintaining stereochemical integrity in this pathway?
A: The process relies heavily on the stereochemical fidelity of the starting diester (derived from tartaric acid) and the controlled conditions of the reduction step using DIBAL-H. Additionally, the choice of cyclopropanation agent (e.g., isopropylidene triphenylphosphorane for trans or sulfurane for cis) dictates the final geometric configuration of the cyclopropane ring.
Q: Is this synthetic route suitable for large-scale commercial production of pyrethroid intermediates?
A: Yes, the route utilizes standard organic transformations such as reduction, olefination, and oxidation which are well-established in industrial settings. The use of commercially available chiral precursors and robust reagents like DIBAL-H and periodates suggests high scalability potential for manufacturing high-purity agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hemicaronic Aldehyde Supplier
The technological breakthroughs detailed in CN1020451C represent a significant leap forward in the synthesis of chiral cyclopropane derivatives, yet translating laboratory success to commercial reality requires expert execution. NINGBO INNO PHARMCHEM stands at the forefront of this translation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive reagents and cryogenic conditions required for this process, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and dedicated process development team, we guarantee the consistent supply of high-enantiomeric excess intermediates essential for next-generation pyrethroids.
We invite global partners to collaborate with us to unlock the full potential of this enantioselective technology. Whether you require custom synthesis of specific cis or trans isomers or optimization of the oxidative cleavage step for your specific application, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how we can drive efficiency and quality in your supply chain together.
