Scalable Synthesis of Diphenyl Ether Insecticide Intermediates via Optimized Wittig Chemistry
Scalable Synthesis of Diphenyl Ether Insecticide Intermediates via Optimized Wittig Chemistry
Introduction to Advanced Agrochemical Intermediate Synthesis
The development of potent insecticidal agents relies heavily on the precise construction of complex molecular architectures, particularly those featuring diphenyl ether scaffolds which are renowned for their biological efficacy. Patent CN88100991A discloses a sophisticated methodology for preparing compounds of formula (I) that exhibit significant pesticidal activity, addressing the critical need for robust synthetic routes in the agrochemical sector. This technology leverages a strategic combination of Wittig olefination and subsequent catalytic reduction to establish the carbon backbone with high fidelity. For global R&D teams, understanding the nuances of this pathway is essential for evaluating the feasibility of integrating these intermediates into broader crop protection portfolios. The ability to access these structures efficiently directly impacts the speed at which new formulations can reach field testing phases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing substituted diphenyl ether derivatives often suffer from significant inefficiencies that hinder commercial viability. Conventional methods frequently rely on multi-step sequences involving harsh reaction conditions that can compromise the integrity of sensitive functional groups, such as halogens or fluorinated moieties, which are crucial for pesticidal potency. Furthermore, older methodologies may struggle with regioselectivity, leading to complex mixtures of isomers that require extensive and costly purification processes to separate the biologically active stereoisomer from inactive byproducts. These challenges not only inflate the cost of goods sold but also introduce variability in supply quality, creating bottlenecks for procurement managers who require consistent batch-to-batch performance. The accumulation of phosphorus waste from stoichiometric reagents in classical approaches also poses environmental compliance hurdles that modern facilities strive to avoid.
The Novel Approach
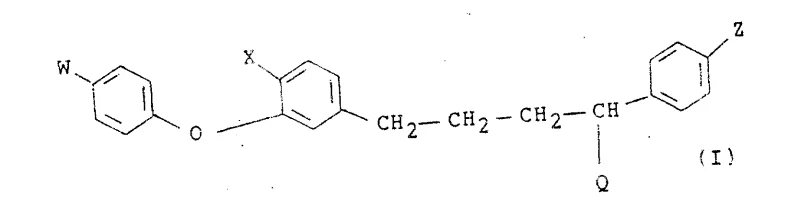
The innovative strategy outlined in the patent data presents a transformative solution by utilizing a Wittig reaction between specific phosphorus ylides and fluorinated ketones to construct the central diene system with remarkable precision. This approach allows for the modular assembly of the molecule, enabling chemists to easily vary substituents on the aromatic rings to tune biological properties without redesigning the entire synthetic pathway. By employing stabilized or semi-stabilized ylides derived from allyl halides, the reaction favors the formation of the desired E-configuration, significantly reducing the burden on downstream purification units. Following the olefination, a catalytic hydrogenation step smoothly reduces the double bonds to yield the saturated alkyl chain found in the final active ingredient precursors. This streamlined two-stage sequence (olefination followed by reduction) minimizes unit operations and enhances overall throughput.
Mechanistic Insights into Wittig Olefination and Reduction
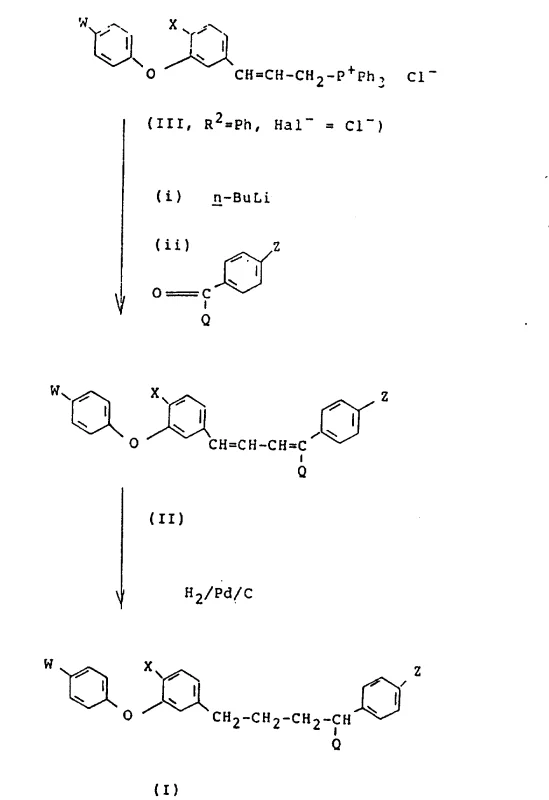
A deep mechanistic understanding of the Wittig reaction employed here reveals why this pathway offers superior control over impurity profiles compared to alternative coupling methods. The reaction proceeds through the formation of a betaine intermediate which collapses into an oxaphosphetane ring before eliminating triphenylphosphine oxide to form the carbon-carbon double bond. The choice of base, typically n-Butyl Lithium, is critical for generating the ylide species quantitatively without inducing side reactions on the sensitive ether linkage or the halogenated aromatic rings. The electronic nature of the substituents on the ketone partner, particularly the electron-withdrawing trifluoromethyl groups, influences the reactivity and stereoselectivity of the olefination, ensuring that the thermodynamic E-isomer is predominantly formed. This level of mechanistic predictability is vital for R&D directors who need to guarantee that the impurity spectrum remains within strict regulatory limits for agrochemical registration.
Following the construction of the diene framework, the catalytic hydrogenation step serves as a powerful tool for impurity control and final structure refinement. The use of palladium on carbon catalysts under controlled hydrogen pressure allows for the selective reduction of the olefinic bonds while leaving the aryl halides and ether linkages intact, demonstrating excellent chemoselectivity. This step effectively removes any residual Z-isomers that might have formed during the Wittig step, as the steric differences often lead to differential reduction rates or allow for crystallization-driven purification of the saturated product. Moreover, the hydrogenation process converts the reactive diene into a stable alkyl chain, improving the shelf-life and handling safety of the intermediate for downstream formulation. The robustness of this reduction ensures that the final product meets the stringent purity specifications required for high-performance insecticidal applications.
How to Synthesize Diphenyl Ether Intermediates Efficiently
The practical execution of this synthesis requires careful attention to reagent quality and reaction parameters to maximize yield and minimize waste generation. The process begins with the preparation of the phosphorus-containing reagents from readily available allyl halides, followed by the condensation with the fluorinated ketone building blocks under inert atmosphere conditions. Detailed standardized operating procedures are essential to maintain the low-temperature requirements during ylide generation and to manage the exothermic nature of the subsequent quenching steps. For process chemists looking to implement this route, adherence to the specific molar ratios and solvent choices described in the technical literature is paramount to achieving the reported efficiency. The following guide outlines the critical operational phases necessary to translate this laboratory-scale innovation into a reliable manufacturing process.
- Preparation of allyl halide intermediates via vinylation of substituted benzaldehydes followed by hydrohalogenation.
- Conversion of allyl halides into phosphonium salts or phosphate esters to generate active Wittig reagents.
- Execution of Wittig condensation with fluorinated ketones followed by catalytic hydrogenation to saturate the diene system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, adopting this optimized synthetic route offers substantial strategic benefits for procurement managers and supply chain leaders focused on cost efficiency and reliability. The elimination of complex transition metal cross-coupling catalysts in favor of phosphorus-based chemistry significantly reduces the raw material costs associated with expensive palladium or nickel ligands, leading to direct savings in the bill of materials. Additionally, the high stereoselectivity of the Wittig step reduces the need for resource-intensive chromatographic separations, allowing for simpler crystallization or distillation workflows that lower utility consumption and waste disposal fees. This streamlined process architecture enhances the overall economic viability of producing these high-value agrochemical intermediates, making it a compelling option for long-term sourcing contracts. Companies can leverage these efficiencies to negotiate more competitive pricing structures while maintaining healthy margins in a volatile market.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for precious metal catalysts often required in alternative cross-coupling strategies, thereby removing the cost volatility associated with commodities like palladium. By utilizing stoichiometric phosphorus reagents that are commercially abundant, the process ensures a stable and predictable cost base for large-scale production campaigns. Furthermore, the high conversion rates observed in the hydrogenation step minimize the loss of valuable fluorinated starting materials, ensuring that every kilogram of input contributes maximally to the final output. These factors combine to create a manufacturing profile that is resilient against raw material price fluctuations and conducive to significant cost reduction in agrochemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as triphenylphosphine and common solvents like tetrahydrofuran ensures that the supply chain is not vulnerable to single-source bottlenecks or geopolitical disruptions affecting specialized reagents. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling suppliers to respond rapidly to fluctuations in demand from downstream formulators. This flexibility is crucial for maintaining continuous production lines and avoiding stockouts that could delay the launch of seasonal crop protection products. Consequently, partners can rely on a steady flow of high-quality intermediates that support just-in-time inventory strategies and reduce the need for excessive safety stock holdings.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as filtration and distillation that are easily transferred from pilot plants to multi-ton reactors without significant re-engineering. The primary byproduct, triphenylphosphine oxide, can be potentially recycled or disposed of using established waste management protocols, simplifying environmental compliance compared to processes generating heavy metal waste. This alignment with green chemistry principles reduces the regulatory burden on manufacturing sites and supports corporate sustainability goals. The ability to scale from 100 kgs to 100 MT/annual commercial production ensures that the technology can grow alongside market demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology in an industrial setting. These insights are derived from a thorough analysis of the patent specifications and are intended to clarify the operational advantages and chemical constraints of the process. Understanding these details helps stakeholders make informed decisions about integrating these intermediates into their supply chains. The answers reflect the consensus on best practices for handling fluorinated agrochemical building blocks and managing the associated reaction workflows.
Q: How is stereochemical purity controlled during the Wittig olefination step?
A: The process utilizes specific phosphonium salts under strongly basic conditions to favor the E-isomer configuration, which is critical for biological activity.
Q: What are the advantages of using phosphate esters over phosphonium salts?
A: Phosphate ester variants (Horner-Wadsworth-Emmons modification) often offer easier workup procedures and reduced phosphine oxide waste generation compared to traditional triphenylphosphine routes.
Q: Is the catalytic hydrogenation step scalable for industrial production?
A: Yes, the reduction of the diene system uses standard palladium-on-carbon catalysts under moderate pressure, making it highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyl Ether Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of global agrochemical innovation pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of diphenyl ether derivatives meets the exacting standards required for pesticide registration. Our commitment to technical excellence means that we do not just supply chemicals; we provide solutions that enhance the reliability and performance of your final formulations.
We invite you to collaborate with us to explore how this advanced synthetic route can optimize your procurement strategy and reduce overall project costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner. Let us help you secure a competitive advantage in the agrochemical market through superior supply chain execution and technical expertise.
