Advanced D-IP3 and L-IP3 Synthesis: Scalable Routes for High-Purity Signaling Molecules
The landscape of biochemical research and pharmaceutical development relies heavily on the availability of high-purity signaling molecules, particularly inositol phosphates like D-inositol 1,4,5-trisphosphate (D-IP3). Patent CN1122030C introduces a transformative synthetic methodology that addresses the longstanding bottlenecks in producing these critical intermediates. Historically, the synthesis of enantiomerically pure inositol derivatives has been plagued by inefficient purification steps, but this invention offers a robust alternative by prioritizing crystallization over chromatography. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process efficiency. The core innovation lies in the strategic use of menthyl chloroformate for chiral resolution, allowing for the isolation of key intermediates like D4P through simple recrystallization rather than labor-intensive column separation. This shift not only enhances the purity profile of the final API intermediate but also drastically simplifies the manufacturing workflow, making it highly attractive for commercial scale-up of complex signaling molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as the widely cited Ozaki synthesis, have long presented substantial economic and logistical challenges for industrial manufacturers. The traditional approach necessitates multiple column chromatography steps to separate various inositol diastereomers, a process that is inherently costly and difficult to scale. The patent explicitly notes that conventional silica gel usage can cost approximately $150/kg, creating a heavy financial burden when processing large batches. Furthermore, chromatographic separations often result in lower overall yields due to product loss on the column and require vast quantities of organic solvents, complicating waste management and environmental compliance. For supply chain heads, these inefficiencies translate into longer lead times and higher volatility in raw material costs. The reliance on non-commercially available reagents in some older methods further exacerbates supply risks, making consistent production of high-purity OLED material or pharmaceutical grades nearly impossible without significant process redesign.
The Novel Approach
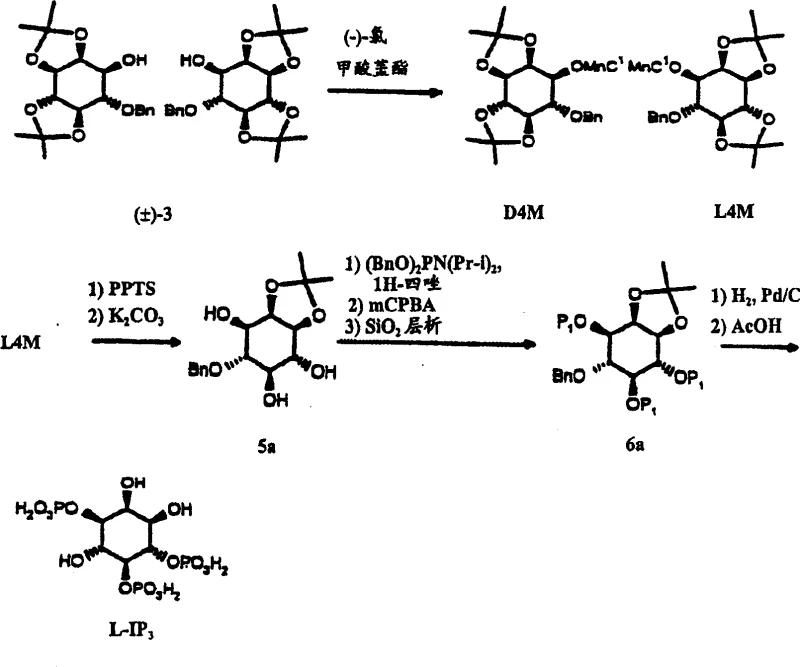
The methodology described in CN1122030C circumvents these issues by introducing a crystallization-driven purification strategy. By converting the racemic intermediate (±)-3 into a menthyl ester derivative (4P), the process creates diastereomers with distinct physical properties that allow for separation via methanol crystallization. This eliminates the need for repeated silica gel columns for the initial chiral resolution. As illustrated in the reaction scheme below, the pathway from D-inositol to D4P and subsequently to D-IP3 is streamlined to minimize chromatographic interventions. Only one specific intermediate, the tribenzyl phosphate, requires SiO2 chromatography, representing a massive reduction in operational complexity compared to prior art. This approach ensures that cost reduction in API manufacturing is achieved not just through cheaper reagents, but through fundamental process intensification that favors scalable unit operations like filtration and crystallization over batch chromatography.
Mechanistic Insights into Phosphoramidite Phosphorylation and Oxidation
The chemical elegance of this synthesis is perhaps best observed in the phosphorylation and oxidation steps that construct the triphosphate core. The process utilizes N,N-diisopropyl-dibenzylphosphoramidite, often referred to as the Fraser-Reid reagent, to introduce the phosphorus groups onto the inositol ring. This reagent is highly reactive and allows for the efficient formation of phosphite triesters under mild conditions. Following phosphorylation, the protocol employs an in situ oxidation step using meta-chloroperbenzoic acid (mCPBA). This immediate oxidation converts the unstable phosphite triesters into the more robust phosphate triesters, preventing degradation and ensuring high fidelity in the final structure. For technical teams, understanding this mechanism is crucial for impurity control, as the in situ nature of the oxidation minimizes the exposure of reactive intermediates to moisture or hydrolytic conditions. The patent reports yields as high as 92% for this specific transformation, highlighting the robustness of the phosphoramidite coupling strategy in this specific steric environment.
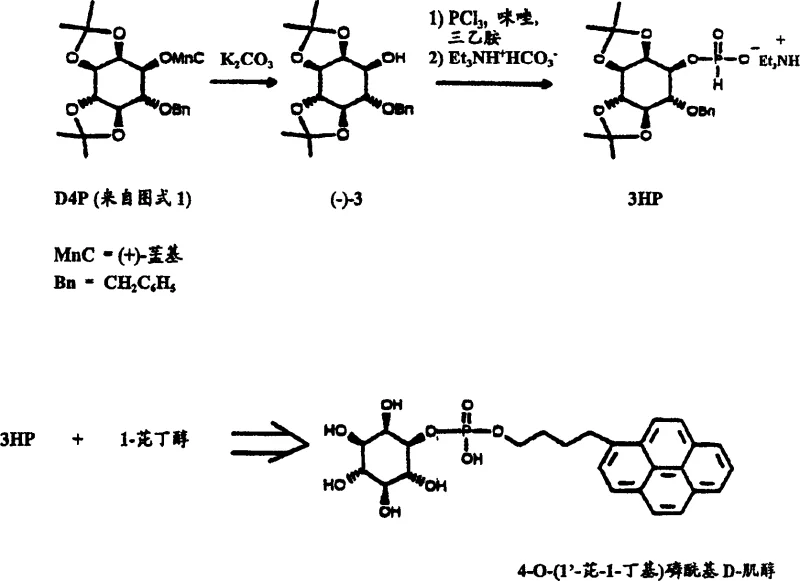
Impurity control is further enhanced by the stability of the protecting groups chosen. The use of isopropylidene acetals provides orthogonal protection that can be selectively removed using pyridinium p-toluenesulfonate (PPTS) without affecting the benzyl ether or the newly formed phosphate esters. This selectivity is vital for maintaining the integrity of the 1,4,5-substitution pattern, which is essential for the biological activity of D-IP3. Additionally, the final hydrogenolysis step using palladium on activated carbon cleanly removes the benzyl protecting groups. The patent specifies that acidification with acetic acid post-hydrogenolysis yields the final free acid form of D-IP3 with high optical purity. This rigorous control over stereochemistry and functional group manipulation ensures that the resulting high-purity inositol phosphates meet the stringent specifications required for cell signaling research and potential therapeutic applications.
How to Synthesize D-IP3 Efficiently
The synthesis of D-IP3 outlined in this patent offers a clear roadmap for laboratories aiming to produce this valuable signaling molecule with minimal resource expenditure. The process begins with the protection of myo-inositol, followed by a regioselective benzylation that sets the stage for chiral resolution. The critical breakthrough is the use of menthyl chloroformate to differentiate enantiomers, allowing the desired D-series intermediate to be harvested via crystallization. Subsequent deprotection, phosphorylation, and global deprotection yield the final trisphosphate. For detailed operational parameters, reagent ratios, and specific workup procedures, please refer to the standardized guide below which encapsulates the critical process controls identified in the patent documentation.
- Convert myo-inositol to (±)-2,3: 4,5-di-O-isopropylidene inositol via double acetonation, followed by regioselective benzylation using cesium fluoride and benzyl bromide.
- Resolve the racemic mixture by acylation with (+)-menthyl chloroformate to form D4P, isolating the desired enantiomer through methanol crystallization.
- Deprotect the 4,5-isopropylidene group, phosphorylate using dibenzyl phosphoramidite, oxidize with mCPBA, and finalize via catalytic hydrogenolysis and acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the purification train. By replacing multiple chromatography steps with crystallization, the process significantly reduces the consumption of silica gel and organic solvents. This reduction directly correlates to lower raw material costs and decreased waste disposal fees, contributing to substantial cost savings in fine chemical manufacturing. Furthermore, crystallization is a unit operation that is far easier to validate and scale in a GMP environment compared to flash chromatography, thereby reducing the risk of batch failure and ensuring consistent supply continuity. The reliance on commercially available reagents like benzyl bromide, cesium fluoride, and mCPBA ensures that the supply chain is not vulnerable to the shortages often associated with exotic or custom-synthesized catalysts.
- Cost Reduction in Manufacturing: The elimination of extensive column chromatography removes a major cost center from the production budget. Silica gel is an expensive consumable, and its removal from the early stages of synthesis lowers the variable cost per kilogram of the intermediate. Additionally, the ability to recover and recycle solvents from crystallization mother liquors is generally higher than from chromatography fractions, further enhancing the economic viability of the process. This efficiency allows for competitive pricing structures when sourcing these complex intermediates, making it an ideal solution for cost-sensitive research programs.
- Enhanced Supply Chain Reliability: The synthetic route utilizes robust, commodity-grade chemicals that are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency. Moreover, the stability of key intermediates, such as the H-phosphonate 3-HP which remains stable for months at low temperatures, allows manufacturers to build strategic inventory buffers. This capability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream customers receive their materials without delay even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in silica gel waste and solvent volume aligns with modern green chemistry principles. Scaling a chromatography-heavy process often requires disproportionately large equipment footprints and waste treatment capacities. In contrast, a crystallization-based process scales linearly and generates less hazardous waste. This makes the technology not only economically superior but also environmentally sustainable, facilitating easier regulatory approval and compliance with increasingly strict environmental regulations in chemical manufacturing zones.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and claims within CN1122030C, providing a factual basis for evaluating the technology's suitability for your specific application. Whether you are concerned about the optical purity of the final product or the stability of the intermediates during storage, the patent provides clear guidance on managing these variables effectively.
Q: How does this new synthesis method reduce production costs compared to traditional Ozaki methods?
A: The patented method significantly reduces reliance on column chromatography, which traditionally requires expensive silica gel (approx. 0/kg). By utilizing crystallization for intermediate purification, specifically using menthyl esters, the process minimizes solvent waste and operational time.
Q: What represents the key chiral resolution strategy in this patent?
A: The process employs (+)-menthyl chloroformate to derivatize the racemic inositol intermediate. This creates diastereomers that can be separated via simple crystallization from methanol, avoiding the need for costly chiral HPLC separation.
Q: Is the intermediate 3-HP stable for long-term storage?
A: Yes, the patent data indicates that the intermediate 6-O-benzyl-2,3:4,5-di-O-isopropylidene inositol H-phosphonate (3-HP) is a colorless liquid that remains stable for several months when stored at -20°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-IP3 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality signaling molecules play in advancing pharmaceutical and biochemical research. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1122030C can be translated into reliable industrial output. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of D-IP3 or L-IP3 meets the exacting standards required for cell biology assays and drug discovery programs. Our commitment to technical excellence means we can navigate the complexities of phosphorylation and chiral resolution with precision.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can lower your overall project costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-purity intermediates into your supply chain.
