Industrial Scale Synthesis of Vinpocetine: A Technical Breakdown for Global Procurement
The global demand for cerebrovascular therapeutics has driven intense scrutiny on the manufacturing protocols of key active pharmaceutical ingredients, specifically Vinpocetine. As detailed in patent CN102040606A, a robust semi-synthetic methodology has been established that transitions production away from unreliable botanical extraction towards a controlled, four-step chemical synthesis. This patent outlines a comprehensive pathway starting from Tabersonine, an indole alkaloid extracted from African Voacanga seeds, effectively bypassing the bottlenecks of traditional Vincamine sourcing. The technical significance of this route lies in its ability to guarantee product purity exceeding 99% while maintaining a yield profile suitable for multi-tonnage commercial output. For procurement strategists and R&D leaders, understanding this specific workflow is critical, as it represents a shift from agrarian dependency to industrial chemical precision. The structural integrity of the final molecule, characterized by its unique eburnane skeleton and ethyl ester functionality, is preserved through mild reaction conditions that prevent racemization or degradation.
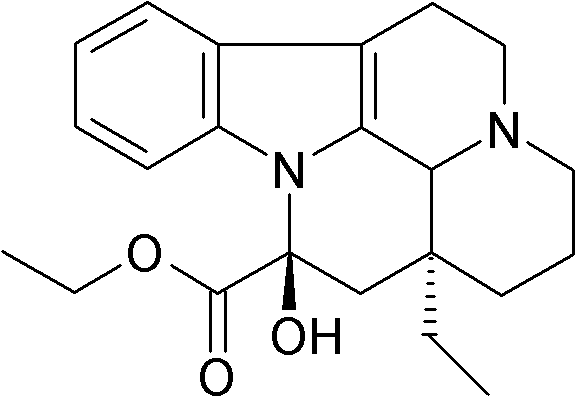
Furthermore, the integration of this synthetic route addresses the chronic supply instability inherent in the pharmaceutical intermediate sector. By decoupling the production of Vinpocetine from the seasonal harvest cycles of Vinca minor, manufacturers can secure a continuous pipeline of high-purity material. The patent explicitly details the optimization of each unit operation, from the initial hydrogenation to the final crystallization, ensuring that impurities are systematically removed at every stage. This level of process control is essential for meeting the stringent regulatory requirements of major markets such as the FDA and EMA. Consequently, adopting this technology allows supply chain directors to mitigate risks associated with raw material scarcity and price volatility, establishing a more resilient foundation for long-term drug formulation projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Vinpocetine has been plagued by two distinct methodologies, both of which suffer from severe commercial and technical drawbacks that hinder scalable manufacturing. The first approach, total chemical synthesis, involves constructing the complex pentacyclic ring system from scratch. While theoretically sound, this method is practically inefficient due to the excessive number of reaction steps required, leading to cumulative yield losses that render the process economically unviable for industrial application. The second conventional method relies on the direct extraction of Vincamine from the leaves of Vinca minor plants. This biological route is inherently fragile; it is subject to geographical constraints, climatic variations, and agricultural pests, resulting in fluctuating raw material quality and availability. Moreover, the extraction process itself is labor-intensive and often yields crude products laden with structurally similar alkaloid impurities that are difficult to separate, compromising the final API purity.
The Novel Approach
In stark contrast, the novel semi-synthetic approach described in the patent data leverages Tabersonine as a superior starting material, offering a streamlined pathway that resolves the inefficiencies of previous methods. By utilizing Tabersonine, which is more abundantly available from Voacanga species, the process eliminates the geographical bottlenecks associated with Vinca minor. The four-step sequence—hydrogenation, oxidation-reduction, hydrolysis, and esterification—is designed for operational simplicity and high throughput. Crucially, this method introduces specific purification protocols, such as recrystallization from petroleum ether and methanol washes, which effectively strip away side products and unreacted intermediates. The result is a manufacturing protocol that not only achieves a content purity of 99% but also demonstrates a robustness that allows for seamless scaling from pilot batches to full commercial production without the need for complex chromatographic separations.
Mechanistic Insights into Tabersonine-to-Vinpocetine Conversion
The core of this synthetic strategy relies on a precise manipulation of the indole alkaloid framework, beginning with the catalytic hydrogenation of Tabersonine. In this initial step, the double bond within the D-ring is selectively reduced using a palladium on carbon (Pd/C) catalyst under moderate hydrogen pressure (3-5 MPa) and temperatures ranging from 50 to 60°C. This transformation yields Vincadifformine, setting the stereochemical stage for subsequent modifications. The choice of Pd/C over platinum oxide is a deliberate economic decision, as it offers comparable catalytic activity at a fraction of the cost, while the controlled pressure conditions ensure safety and reproducibility in large-scale reactors. Following hydrogenation, the process moves to a critical oxidation-reduction sequence where Vincadifformine is treated with m-chloroperbenzoic acid (m-CPBA) to form an intermediate N-oxide, which is subsequently reduced using Triphenylphosphine. This step is mechanistically vital as it rearranges the molecular skeleton to form the essential Vincamine structure, preserving the chiral centers necessary for biological activity.
Impurity control is rigorously managed throughout the latter half of the synthesis, particularly during the hydrolysis and esterification phases. The conversion of Vincamine to Vincamine Acid via alkaline hydrolysis using potassium hydroxide in anhydrous methanol is monitored closely via thin-layer chromatography to prevent over-hydrolysis or degradation of the sensitive indole nucleus. The final esterification step employs a dual-catalyst system involving sulfuric acid and a cation exchange resin. This combination accelerates the formation of the ethyl ester while the resin acts as a scavenger for basic impurities, thereby simplifying the workup procedure. The final product is isolated through a series of pH adjustments and solvent extractions using dichloromethane, followed by crystallization from methanol. This multi-stage purification ensures that trace metals from the catalyst and organic byproducts are reduced to negligible levels, satisfying the rigorous impurity profiles demanded by modern pharmacopeias for cerebral vasodilators.
How to Synthesize Vinpocetine Efficiently
The execution of this synthesis requires strict adherence to the specified reaction parameters to maximize yield and minimize waste generation. The process begins with the dissolution of high-purity Tabersonine in anhydrous methanol, followed by the addition of the palladium catalyst and pressurization with hydrogen gas. Once the hydrogenation is complete, the catalyst is filtered off, and the solvent is concentrated to induce crystallization of the intermediate Vincadifformine. The subsequent oxidation step demands careful temperature control between 25 and 28°C to manage the exothermic nature of the peracid reaction, followed by a prolonged stirring period with Triphenylphosphine to ensure complete reduction. Detailed standardized operating procedures for each unit operation, including specific solvent volumes, reflux times, and drying protocols, are essential for replicating the patent's success in a GMP environment.
- Hydrogenation of Tabersonine using Palladium on Carbon catalyst under 3-5 MPa pressure to form Vincadifformine.
- Oxidation with m-chloroperbenzoic acid followed by reduction with Triphenylphosphine to yield Vincamine, including solvent recovery.
- Alkaline hydrolysis of Vincamine using Potassium Hydroxide in methanol to produce Vincamine Acid.
- Acid-catalyzed esterification using ethanol and cation exchange resin to finalize Vinpocetine with 99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this semi-synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The most significant advantage lies in the decoupling of production from volatile agricultural supply chains. By shifting the raw material base to Tabersonine, which can be sourced from more stable supply regions compared to Vinca minor, companies can secure long-term contracts with predictable pricing structures. Furthermore, the process design inherently supports cost reduction through the implementation of closed-loop recycling systems. Specifically, the recovery of Triphenylphosphine oxide from the filtrate allows for its regeneration and reuse in subsequent batches. This circular economy approach drastically lowers the consumption of high-value reagents, directly impacting the cost of goods sold (COGS) without compromising product quality.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the strategic selection of reagents and the implementation of recovery loops. By utilizing Palladium on Carbon instead of more expensive platinum-based catalysts, the initial capital expenditure on catalytic materials is minimized. More importantly, the patented method includes a specific protocol for recovering Triphenylphosphine from the reaction mother liquor. Since Triphenylphosphine is a costly reagent, its ability to be recycled with high efficiency means that the effective cost per kilogram of the final API is significantly lower than in processes where this reagent is consumed stoichiometrically and discarded. Additionally, the use of common solvents like methanol, ethanol, and dichloromethane facilitates easy solvent recovery and reuse, further driving down operational expenses.
- Enhanced Supply Chain Reliability: Supply chain resilience is dramatically improved by the robustness of the synthetic pathway. Unlike direct plant extraction, which is susceptible to crop failures and seasonal downtime, this chemical synthesis can operate continuously throughout the year, provided there is a steady feed of Tabersonine. The process tolerances are wide enough to accommodate minor variations in raw material quality without affecting the final output, reducing the rate of batch rejections. This reliability allows supply chain heads to maintain lower safety stock levels while still meeting delivery commitments, optimizing working capital and reducing inventory holding costs associated with perishable botanical raw materials.
- Scalability and Environmental Compliance: The transition from laboratory to industrial scale is facilitated by the simplicity of the unit operations involved, which primarily consist of hydrogenation, filtration, crystallization, and distillation. These are standard operations in any fine chemical facility, meaning no exotic or specialized equipment is required, accelerating the timeline for technology transfer. From an environmental perspective, the process minimizes waste generation through efficient solvent recycling and the recovery of phosphorus byproducts. The use of a cation exchange resin in the final step also reduces the load of acidic wastewater, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations governing API manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Vinpocetine synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what potential partners and licensees can expect regarding performance and scalability. Understanding these nuances is vital for making informed decisions about technology adoption and vendor selection in the competitive landscape of cerebrovascular therapeutics.
Q: What are the primary advantages of the Tabersonine route over direct plant extraction?
A: The semi-synthetic route starting from Tabersonine extracted from Voacanga seeds overcomes the geographical and seasonal limitations of harvesting Vinca minor plants. It offers a more consistent supply chain, higher overall yields, and eliminates the variability associated with direct botanical extraction of Vincamine.
Q: How does this process address cost efficiency in large-scale manufacturing?
A: The process incorporates a critical recovery loop for Triphenylphosphine, a key reagent in the oxidation-reduction step. By recycling this expensive reagent with high efficiency (>98% recovery), the overall raw material cost is significantly reduced. Additionally, the use of heterogeneous catalysts like Pd/C simplifies downstream processing.
Q: Is the final esterification step compliant with green chemistry principles?
A: Yes, the final step utilizes a cation exchange resin in conjunction with sulfuric acid. This heterogeneous catalysis approach minimizes corrosive waste and simplifies product isolation compared to traditional homogeneous acid catalysis, aligning with modern environmental compliance standards for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinpocetine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent data to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical yields described in patents like CN102040606A are realized in actual manufacturing campaigns. We understand that for cerebral vasodilators, consistency is key, which is why our facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels. Our commitment to stringent purity specifications ensures that every batch of Vinpocetine we produce meets the highest international standards, ready for immediate formulation into finished dosage forms.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this semi-synthetic supply chain. We encourage you to request specific COA data and route feasibility assessments to validate our capabilities against your internal benchmarks. Let us be your strategic partner in securing a stable, high-quality, and cost-effective supply of Vinpocetine for the global market.
