Revolutionizing Chiral Synthesis with Dehydroabietine Thiourea Catalysts for Scalable Pharma Production
Introduction to Advanced Organocatalytic Technologies
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the need for more sustainable and efficient methods to produce high-purity chiral intermediates. Patent CN101318146A introduces a groundbreaking class of dehydroabietine-chiral thiourea catalysts that address critical limitations in current organocatalytic methodologies. This technology leverages naturally derived chiral sources to create robust catalysts capable of high-efficiency dual-handed synthesis, meaning they can effectively produce both (R) and (S) enantiomers of gamma-nitro-aromatic ketones and heteroketones with exceptional optical purity. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a pivotal shift towards more reliable and cost-effective manufacturing pathways for complex API intermediates. The ability to access both enantiomeric forms with high selectivity using a unified catalyst platform significantly streamlines process development and reduces the dependency on multiple distinct catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric organocatalysis has faced substantial challenges regarding catalyst versatility and configurational control. Traditional bifunctional thiourea catalysts, such as those derived from cinchona alkaloids or sugar rings, often exhibit a pronounced bias where they excel at producing one specific enantiomer but fail dramatically when attempting to synthesize the opposite configuration. For instance, prior art indicates that certain sugar-derived thiourea catalysts might achieve high yields for the (S)-configuration but suffer from poor reactivity and significantly reduced enantioselectivity when targeting the (R)-configuration, sometimes yielding less than half the desired product quality. This inconsistency forces manufacturers to maintain separate inventory lines for different catalyst types, complicating supply chain logistics and increasing the overall cost of goods sold. Furthermore, many conventional methods rely on harsh reaction conditions or expensive metal additives that introduce contamination risks and necessitate rigorous purification steps to meet stringent pharmaceutical impurity profiles.
The Novel Approach
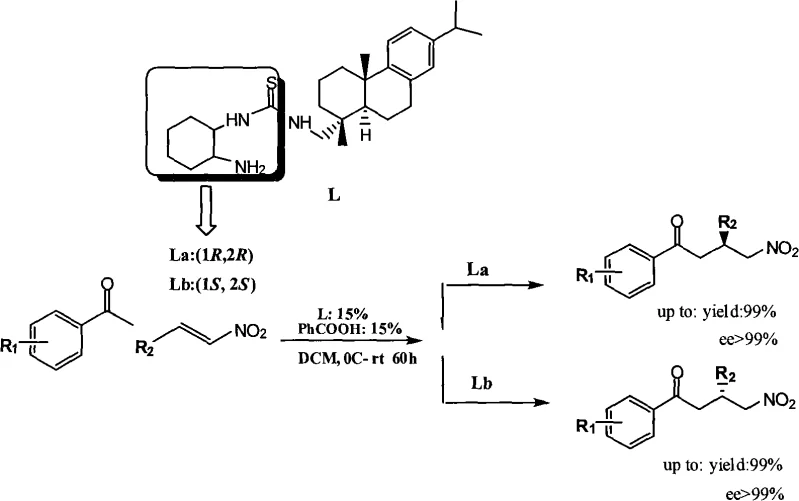
The innovation disclosed in patent CN101318146A overcomes these historical barriers by utilizing a dehydroabietine backbone, a rigid and bulky natural product derivative, to support the chiral thiourea moiety. This structural design creates a highly defined chiral environment that maintains high catalytic activity regardless of whether the (1R,2R) or (1S,2S) cyclohexanediamine component is employed. Consequently, manufacturers can achieve up to 99% yield and greater than 99% enantiomeric excess (ee) for both target configurations using essentially the same synthetic protocol. This dual-handed capability is a rare and valuable trait in industrial catalysis, allowing for flexible production scheduling and rapid response to changing market demands for specific enantiomers. The catalyst is also noted for its ease of preparation, separation, and recyclability, which aligns perfectly with green chemistry principles and reduces the environmental footprint of large-scale chemical manufacturing processes.
Mechanistic Insights into Dehydroabietine-Thiourea Catalyzed Michael Addition
The core of this technology lies in the bifunctional nature of the dehydroabietine-thiourea catalyst, which operates through a sophisticated hydrogen-bonding network to activate both the nucleophile and the electrophile simultaneously. In the asymmetric Michael addition of ketones to nitroalkenes, the thiourea moiety acts as a double hydrogen bond donor to activate the nitro group of the alkene, while the amine functionality assists in enolizing the ketone substrate. This cooperative activation lowers the energy barrier for the carbon-carbon bond formation while strictly controlling the spatial approach of the reactants. The bulky dehydroabietine skeleton provides significant steric hindrance that shields one face of the reactive intermediate, thereby enforcing high stereoselectivity. Unlike flexible catalyst backbones that may adopt multiple conformations leading to mixed stereochemical outcomes, the rigid tricyclic structure of dehydroabietine locks the catalyst into a single, highly effective conformation that ensures consistent chirality transfer throughout the reaction cycle.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical manufacturing. The high specificity of the catalyst minimizes the formation of diastereomers and regioisomers, which are often difficult to separate and can pose toxicological risks in final drug products. The reaction proceeds efficiently in dichloromethane with a benzoic acid additive, which further fine-tunes the acidity of the system to optimize reaction rates without promoting side reactions. The mild conditions, operating between 0°C and room temperature, prevent thermal degradation of sensitive functional groups often present in complex drug intermediates. This level of control ensures that the resulting gamma-nitro-aromatic ketones and heteroketones meet the rigorous purity specifications required for downstream processing into active pharmaceutical ingredients, reducing the burden on quality control laboratories and minimizing batch rejection rates.
How to Synthesize Chiral Gamma-Nitro Ketones Efficiently
Implementing this catalytic system in a production environment involves a straightforward protocol that balances high performance with operational simplicity. The process begins with the preparation of the specific enantiomer of the dehydroabietine-thiourea catalyst required for the target product configuration, followed by the setup of the Michael addition reaction in a standard reactor. The methodology is designed to be robust against minor variations in moisture or oxygen levels, making it more forgiving than many transition-metal catalyzed alternatives. Detailed standardized synthetic steps for the preparation of the catalyst and the subsequent conversion of substrates into high-purity chiral intermediates are outlined in the guide below, ensuring reproducibility across different scales of operation.
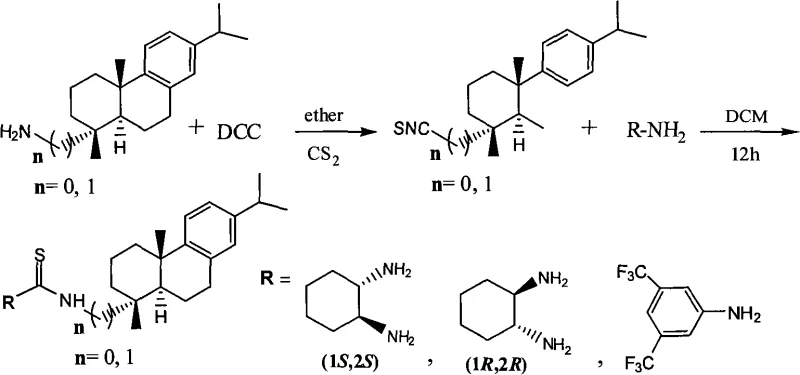
- Prepare the catalyst by reacting dehydroabietamine with carbon disulfide and DCC to form an isothiocyanate intermediate, followed by coupling with chiral cyclohexanediamine.
- Dissolve the nitroalkene substrate, the dehydroabietine-thiourea catalyst (15 mol%), and benzoic acid (15 mol%) in dichloromethane.
- Add the aromatic ketone at 0°C, allow the reaction to proceed to room temperature for 60-72 hours, and purify via column chromatography to obtain high-purity chiral products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of dehydroabietine-thiourea catalysts presents a compelling value proposition centered around risk mitigation and long-term cost efficiency. The shift towards organocatalysis eliminates the reliance on precious metals such as palladium, rhodium, or iridium, which are subject to volatile market pricing and geopolitical supply constraints. By removing these heavy metals from the process entirely, manufacturers can bypass expensive scavenging steps and reduce the complexity of waste treatment protocols, leading to substantial operational savings. Furthermore, the ability to produce both enantiomers with high efficiency using a single catalyst platform simplifies inventory management, allowing companies to maintain a leaner stock of catalytic materials while retaining the flexibility to switch production between different chiral targets as needed.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts directly translates to lower raw material costs and reduced expenditure on metal removal technologies. Since the catalyst is derived from dehydroabietic acid, a readily available natural product, the starting materials are abundant and cost-stable compared to synthetic chiral ligands. The high turnover and recyclability of the catalyst mean that less material is consumed per kilogram of product, driving down the variable cost of production. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling, contributing to a lower overall carbon footprint and utility costs for the facility.
- Enhanced Supply Chain Reliability: Sourcing natural product-derived catalysts mitigates the risk of supply disruptions often associated with complex synthetic reagents. The robustness of the reaction conditions allows for manufacturing in facilities with standard equipment capabilities, reducing the need for specialized infrastructure. High yields and selectivity mean that less starting material is wasted, improving the overall mass balance and ensuring that supply commitments can be met consistently. The simplified purification process also shortens the manufacturing cycle time, enabling faster turnaround from raw material intake to finished intermediate delivery.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical effluents. Scaling up organocatalytic reactions is generally more straightforward than metal-catalyzed ones due to the absence of exothermic risks associated with metal activation. The use of common solvents like dichloromethane, which can be recovered and recycled, further enhances the sustainability profile. This compliance advantage reduces regulatory hurdles and potential fines, securing the long-term viability of the manufacturing site and protecting the company's reputation for environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of dehydroabietine-thiourea catalysts in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this technology into existing production lines.
Q: What is the primary advantage of dehydroabietine thiourea catalysts over traditional sugar-derived catalysts?
A: Unlike traditional catalysts that often exhibit poor reactivity for one specific enantiomer configuration, dehydroabietine thiourea catalysts enable high-yield and high-enantioselectivity synthesis for both (R) and (S) configurations using analogous catalyst structures, providing true dual-handed control.
Q: Are metal reagents required for this asymmetric Michael addition process?
A: No, the process described in patent CN101318146A is a metal-free organocatalytic method. It utilizes a bifunctional thiourea catalyst and benzoic acid additive, eliminating the need for expensive transition metals and simplifying downstream purification.
Q: What are the typical reaction conditions for synthesizing gamma-nitro-aromatic ketones?
A: The reaction typically proceeds in dichloromethane (DCM) starting at 0°C and warming to room temperature over 60 to 72 hours. It tolerates mild conditions without the need for strict anhydrous or oxygen-free environments, making it suitable for scalable operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic technologies like the dehydroabietine-thiourea system in accelerating drug development timelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of chiral intermediates meets the exacting standards required by global regulatory bodies. We are equipped to handle the nuances of asymmetric synthesis, providing our clients with a secure and efficient supply chain for their most critical pharmaceutical building blocks.
We invite you to collaborate with us to explore how this cutting-edge catalytic technology can optimize your specific manufacturing requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's unique needs, demonstrating exactly how switching to this metal-free pathway can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you navigate the complexities of chiral synthesis with confidence and precision.
