Advanced Synthetic Route for 3-Fluoro-2-Methyl-4-Trifluoromethylaniline Hydrochloride: Scalable Manufacturing Insights
The pharmaceutical and agrochemical industries continuously demand advanced fluorinated building blocks due to their unique metabolic stability and lipophilicity profiles. Patent CN110885290A introduces a groundbreaking synthetic methodology for producing 3-fluoro-2-methyl-4-trifluoromethylaniline hydrochloride, a critical intermediate for next-generation bioactive molecules. This invention addresses a significant gap in the literature by providing the first reported, robust route to this specific substitution pattern, leveraging modern cross-coupling technologies alongside classical organolithium chemistry. For R&D directors and procurement strategists, this patent represents a pivotal shift from unpredictable, low-yield exploratory chemistry to a defined, scalable industrial process. The methodology not only ensures high chemical purity but also simplifies the operational complexity typically associated with poly-fluorinated aromatic systems, thereby offering a reliable foundation for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, synthesizing specifically substituted trifluoromethyl anilines has been fraught with challenges regarding regioselectivity and yield. Conventional electrophilic aromatic substitution often fails to distinguish between similar positions on the benzene ring when multiple electron-withdrawing groups like fluorine and trifluoromethyl are present, leading to difficult-to-separate isomeric mixtures that drastically reduce overall process efficiency. Furthermore, older amination techniques frequently required harsh conditions, such as high-pressure ammonia reactions or the use of unstable copper salts, which posed significant safety risks and environmental burdens in a manufacturing setting. These legacy methods often resulted in substantial waste generation and inconsistent product quality, making them unsuitable for the rigorous purity specifications demanded by modern regulatory bodies for active pharmaceutical ingredients (APIs). The inability to precisely install the methyl group ortho to the fluorine atom without affecting other sensitive functionalities was a persistent bottleneck that limited the availability of this valuable chemical scaffold.
The Novel Approach
The patented process overcomes these historical hurdles by integrating a highly selective directed ortho-metalation (DoM) strategy with a sophisticated palladium-catalyzed Buchwald-Hartwig amination. This dual-strategy approach allows for the precise construction of the carbon skeleton before introducing the nitrogen functionality, ensuring that the sensitive amine group is protected throughout the harsher synthetic steps. By utilizing tert-butyl carbamate (BocNH2) as the nitrogen source, the method avoids the formation of secondary amine byproducts and simplifies the purification workflow. The reaction conditions are remarkably mild, operating effectively between 60°C and 100°C in standard solvents like dioxane, which facilitates easier heat management and reactor compatibility compared to extreme temperature processes. This novel route transforms a previously elusive target into a commercially viable commodity, streamlining the path from raw materials to the final hydrochloride salt with exceptional efficiency.
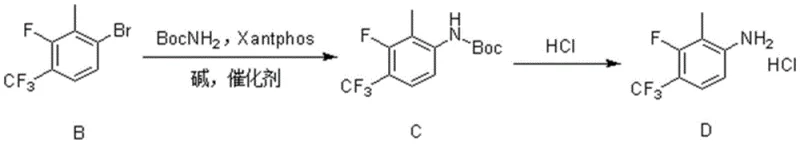
Mechanistic Insights into Pd-Catalyzed Amination and Ortho-Lithiation
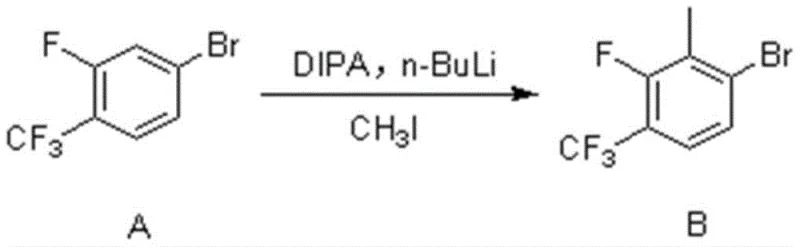
The core of this synthesis lies in the meticulous control of reactivity through mechanistic understanding, particularly in the formation of Compound B and its subsequent conversion to Compound C. The initial step involves the generation of a lithiated species from 4-bromo-2-fluorobenzotrifluoride using a base mixture of diisopropylamine (DIPA) and n-butyllithium at cryogenic temperatures (-78°C). This low-temperature environment is crucial for kinetic control, preventing side reactions such as halogen-metal exchange at the bromine position, which would lead to polymerization or decomposition. The fluorine atom acts as a directing group, guiding the bulky lithium base to the adjacent ortho-position, where it abstracts a proton to form a stable aryl lithium intermediate. Upon warming and treatment with methyl iodide, this intermediate undergoes nucleophilic substitution to install the methyl group with high fidelity, establishing the correct substitution pattern required for the final API structure.
Following the construction of the carbon framework, the transformation of the aryl bromide (Compound B) to the protected aniline (Compound C) utilizes a state-of-the-art palladium catalytic cycle. The catalyst system employs tris(dibenzylideneacetone)dipalladium [Pd2(dba)3] paired with Xantphos, a bidentate ligand known for its large bite angle. This specific ligand geometry accelerates the reductive elimination step, which is often the rate-limiting factor in the amination of sterically hindered aryl halides. The presence of cesium carbonate as a base facilitates the deprotonation of the carbamate, generating the nucleophilic nitrogen species that attacks the palladium-aryl complex. This mechanism ensures that even with the electron-withdrawing trifluoromethyl and fluorine groups deactivating the ring towards oxidative addition, the reaction proceeds smoothly to high conversion. The result is a clean transformation that minimizes the formation of homocoupling byproducts, directly contributing to the high purity of the isolated intermediate.
How to Synthesize 3-Fluoro-2-Methyl-4-Trifluoromethylaniline Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for reproducing this high-value intermediate in a laboratory or pilot plant setting. The process begins with the careful preparation of the lithiated precursor under inert atmosphere, followed by the palladium-catalyzed coupling which requires precise stoichiometric control of the ligand and catalyst to maximize turnover. The final deprotection step is straightforward, utilizing common acid solutions to remove the Boc group and precipitate the product as a stable hydrochloride salt. For detailed operational parameters, including specific molar ratios, solvent volumes, and temperature ramping rates, operators should refer to the standardized synthesis steps provided below which encapsulate the optimal conditions identified in the experimental examples.
- Perform directed ortho-metalation on 4-bromo-2-fluorobenzotrifluoride using n-BuLi and DIPA at -78°C, followed by quenching with methyl iodide to form the bromo-intermediate.
- Execute a Buchwald-Hartwig coupling reaction between the bromo-intermediate and tert-butyl carbamate using a Pd/Xantphos catalyst system in dioxane at elevated temperatures.
- Deprotect the Boc-group by reacting the intermediate with hydrogen chloride in an organic solvent (methanol, ethyl acetate, or dioxane) to yield the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly impact the bottom line and supply chain resilience for downstream manufacturers. The reliance on commodity chemicals such as methyl iodide, cesium carbonate, and standard organic solvents means that the raw material supply chain is robust and less susceptible to geopolitical disruptions or scarcity issues often associated with exotic reagents. Furthermore, the elimination of complex purification steps, such as extensive chromatography, in favor of crystallization and filtration workflows significantly reduces processing time and solvent consumption. This streamlined approach translates to lower operational expenditures (OPEX) and a reduced environmental footprint, aligning with modern green chemistry initiatives that are increasingly becoming a prerequisite for vendor qualification in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The implementation of this catalytic amination strategy eliminates the need for stoichiometric amounts of expensive copper mediators or harsh nitration/reduction sequences that typically generate large volumes of hazardous waste. By achieving high yields in the coupling step (reported up to 91% in examples), the process maximizes atom economy and minimizes the loss of valuable fluorinated starting materials, which are often the most costly component of the synthesis. Additionally, the ability to recycle solvents like dioxane and ethyl acetate further drives down the variable costs per kilogram of production, offering a competitive pricing structure for bulk procurement without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of 4-bromo-2-fluorobenzotrifluoride as a starting material leverages an established supply chain for fluorinated aromatics, ensuring consistent availability of feedstock. The reaction conditions are tolerant to minor variations in temperature and mixing, which enhances batch-to-batch consistency—a critical factor for maintaining uninterrupted supply to API manufacturers. The robustness of the Pd/Xantphos catalyst system also means that the process is less prone to stalling or failure due to trace impurities in the reagents, reducing the risk of production delays and ensuring that delivery schedules can be met reliably even during periods of high demand.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is facilitated by the absence of extremely hazardous reagents like pyrophoric organometallics in the main coupling step; the lithiation is contained and quenched early in the sequence. The final product isolation involves simple acid-base chemistry and crystallization, unit operations that are easily adapted to large-scale reactors without requiring specialized equipment. Moreover, the reduction in heavy metal waste (due to low catalyst loading) and the use of recyclable solvents simplify wastewater treatment and regulatory compliance, making this a sustainable choice for long-term manufacturing partnerships focused on environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated aniline derivative. These answers are derived directly from the experimental data and technical specifications disclosed in the patent documentation, providing clarity on the feasibility and benefits of adopting this synthetic route for industrial applications. Understanding these nuances helps stakeholders make informed decisions regarding process integration and vendor selection.
Q: What is the key advantage of the lithiation step in this synthesis?
A: The use of directed ortho-metalation (DoM) with n-BuLi/DIPA allows for precise regioselective methylation at the position ortho to the fluorine atom, overcoming the limitations of traditional electrophilic substitution which often yields complex isomer mixtures.
Q: Why is the Xantphos ligand selected for the amination step?
A: Xantphos (4,5-bis(diphenylphosphino)-9,9-dimethylxanthene) possesses a large bite angle that stabilizes the palladium catalyst during the oxidative addition and reductive elimination steps, significantly enhancing the yield of the sterically hindered aryl amine formation.
Q: How does this method improve supply chain stability for fluorinated intermediates?
A: By utilizing readily available starting materials like 4-bromo-2-fluorobenzotrifluoride and avoiding exotic reagents, combined with mild reaction conditions (60-100°C) and high yields, the process ensures consistent batch-to-batch reproducibility essential for long-term commercial supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Fluoro-2-Methyl-4-Trifluoromethylaniline Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of life-saving medicines and advanced agrochemicals. Our technical team has extensively analyzed the synthetic pathway described in CN110885290A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are outfitted with state-of-the-art reactors capable of handling cryogenic lithiation and inert atmosphere coupling reactions, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for this essential building block. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can enhance your project's timeline and budget efficiency while delivering the premium quality required for global regulatory submissions.
