Advanced One-Pot Synthesis of 3-Amino-1,2,4-Triazole Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries continuously seek efficient pathways to access nitrogen-containing heterocycles due to their profound biological significance. Patent CN113480486B introduces a groundbreaking methodology for the preparation of 3-amino-1,2,4-triazole derivatives, a class of compounds renowned for their anticonvulsant, anticancer, antifungal, and antiviral activities. This innovation addresses critical bottlenecks in existing synthetic routes by employing a streamlined one-pot tandem cyclization reaction. By integrating amidine compounds, isothiocyanate compounds, and hydrazine compounds in the presence of a base and polar organic solvent, the process achieves direct construction of the triazole core. This technical advancement is pivotal for R&D directors seeking reliable pharmaceutical intermediate suppliers who can deliver complex scaffolds with superior impurity profiles and consistent quality.
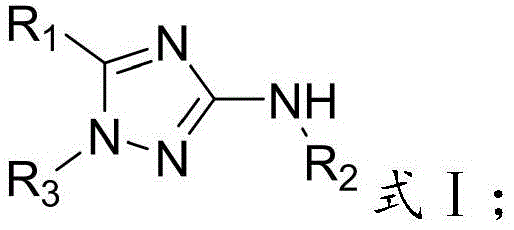
The structural versatility of these derivatives is extensive, allowing for substitution at multiple positions with phenyl, alkyl, alkoxy, halophenyl, and heteroaryl groups. Such flexibility is essential for medicinal chemists optimizing structure-activity relationships (SAR) during drug discovery phases. The ability to synthesize these diverse analogs through a unified, robust protocol represents a significant leap forward in process chemistry, ensuring that supply chains for high-purity pharmaceutical intermediates remain resilient and adaptable to varying project requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-amino-1,2,4-triazoles has been plagued by operational complexity and inefficiency. Prior art, such as the methods disclosed in academic literature, often relies on multi-step sequences that involve harsh reaction conditions and difficult purification protocols. These conventional routes typically require the isolation of unstable intermediates, leading to cumulative yield losses and increased waste generation. Furthermore, the use of specialized or hazardous reagents in traditional methods poses significant safety risks and environmental compliance challenges for manufacturing facilities.
From a supply chain perspective, these legacy processes are fragile. The necessity for multiple discrete reaction steps increases the lead time for production and amplifies the risk of batch-to-batch variability. For procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing, the low atom economy and high solvent consumption of these older methods translate into inflated raw material costs and expensive waste disposal fees. Consequently, there is an urgent industry demand for a consolidated synthetic strategy that mitigates these operational burdens while maintaining high product integrity.
The Novel Approach
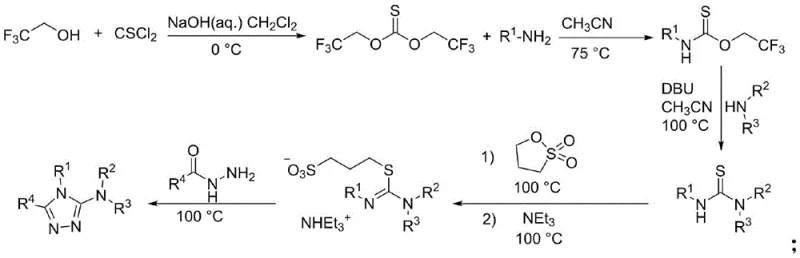
The methodology outlined in patent CN113480486B fundamentally reshapes the production landscape by condensing the synthesis into a single, efficient operation. This novel approach leverages a tandem cyclization mechanism where the amidine, isothiocyanate, and hydrazine components react sequentially in the same vessel. This one-pot design eliminates the need for intermediate isolation, drastically reducing solvent usage and processing time. The reaction proceeds under mild thermal conditions, typically between 20°C and 80°C, which lowers energy consumption and enhances process safety.
Moreover, the new method demonstrates exceptional tolerance to a wide range of functional groups, enabling the synthesis of derivatives with diverse substituents without compromising yield or purity. The operational simplicity allows for easier scale-up, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. By removing the need for highly toxic catalysts and simplifying the workup procedure to standard column chromatography, this technology offers a sustainable and economically viable alternative that aligns perfectly with modern green chemistry principles and stringent regulatory standards.
Mechanistic Insights into Tandem Cyclization Reaction
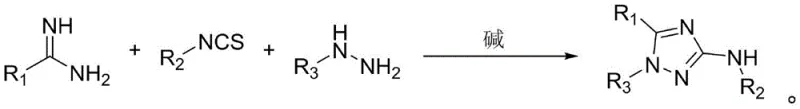
The core of this technological breakthrough lies in the intricate tandem cyclization mechanism that drives the formation of the 1,2,4-triazole ring. The reaction initiates with the nucleophilic attack of the hydrazine compound on the electrophilic carbon of the isothiocyanate group, forming a thiosemicarbazide intermediate in situ. Subsequently, the amidine component participates in a condensation reaction, facilitating the closure of the triazole ring through the elimination of small molecules such as hydrogen sulfide or ammonia derivatives, depending on the specific pathway. This cascade occurs seamlessly within the polar organic solvent medium, promoted by the presence of alkaline reagents like potassium carbonate or sodium hydroxide.
Understanding this mechanism is crucial for controlling the impurity profile of the final product. The choice of base and solvent plays a pivotal role in directing the reaction trajectory towards the desired triazole scaffold while suppressing side reactions such as polymerization or hydrolysis. The patent specifies that bases like potassium carbonate and solvents like toluene or dimethylformamide create an optimal environment for this transformation. For R&D teams, this mechanistic clarity ensures that the process can be finely tuned to maximize the formation of the target isomer, thereby reducing lead time for high-purity pharmaceutical intermediates by minimizing downstream purification efforts.
Furthermore, the compatibility of various substituents (R1, R2, R3) with this mechanism underscores its robustness. Whether introducing electron-withdrawing groups like cyano or halogens, or electron-donating groups like methoxy or alkyl chains, the cyclization proceeds efficiently. This broad substrate scope indicates that the electronic and steric properties of the starting materials do not significantly hinder the cyclization kinetics, providing a reliable platform for generating diverse chemical libraries for biological screening.
How to Synthesize 3-Amino-1,2,4-Triazole Derivatives Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to ensure optimal outcomes. The protocol dictates mixing the amidine, isothiocyanate, and hydrazine precursors with a base in a polar solvent, followed by heating and stirring. Detailed operational guidelines regarding molar ratios, specifically ranging from 1:1.0-1.8:1.5-2.5:2.0-4.0 for the respective components, are critical for reproducibility. The following section outlines the standardized steps derived from the patent examples to facilitate immediate adoption in laboratory or pilot plant settings.
- Mix amidine compounds, isothiocyanate compounds, hydrazine compounds, and an alkali reagent in a polar organic solvent.
- Conduct the tandem cyclization reaction at temperatures between 20°C and 80°C for a duration of 6 to 24 hours.
- Purify the resulting reaction mixture via column chromatography using petroleum ether and ethyl acetate to obtain high-purity derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers substantial strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the manufacturing workflow. By consolidating multiple reaction steps into a single pot, the process inherently reduces the requirement for intermediate storage, handling, and quality control testing. This streamlining directly correlates to lower operational expenditures and a reduced carbon footprint, addressing both economic and environmental mandates faced by modern chemical enterprises.
- Cost Reduction in Manufacturing: The elimination of toxic catalysts and the reduction in solvent volume significantly lower the cost of goods sold (COGS). Traditional methods often rely on expensive transition metals or hazardous reagents that require specialized disposal protocols, adding hidden costs to the final product. In contrast, this new method utilizes inexpensive, commercially available inorganic bases and common organic solvents. The high atom economy of the tandem reaction ensures that a greater proportion of raw materials are converted into the final product, minimizing waste generation and maximizing resource efficiency without compromising on yield.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as benzamidines, phenyl isothiocyanates, and phenylhydrazines ensures a stable supply chain. These precursors are commodity chemicals produced by numerous global suppliers, reducing the risk of raw material shortages that can plague specialized synthetic routes. Furthermore, the robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or mixing rates, ensuring consistent batch quality and reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction temperatures (20-80°C) and atmospheric pressure conditions make this process highly scalable from gram to ton quantities without requiring expensive high-pressure reactors or cryogenic cooling systems. Additionally, the absence of heavy metal catalysts simplifies the purification process and ensures that the final product meets stringent residual metal specifications required by regulatory bodies like the FDA and EMA. This ease of compliance accelerates the regulatory approval timeline for new drug applications utilizing these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-amino-1,2,4-triazole derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity for partners evaluating this technology for integration into their supply chains.
Q: What are the key advantages of this new synthesis method over conventional routes?
A: The novel method described in patent CN113480486B utilizes a one-pot tandem cyclization strategy, eliminating the cumbersome multi-step procedures and complex operations associated with traditional synthesis. This significantly simplifies the workflow, reduces processing time, and enhances suitability for industrial-scale production without requiring highly toxic catalysts.
Q: What represents the typical purity and yield profile for these derivatives?
A: Experimental data indicates that the process consistently yields 3-amino-1,2,4-triazole derivatives with high-performance liquid chromatography (HPLC) purity ranging from 98.5% to 99.9%. Isolated yields vary based on substituent groups but generally demonstrate robust efficiency suitable for commercial intermediate supply.
Q: Is this process scalable for large-volume pharmaceutical intermediate manufacturing?
A: Yes, the protocol is explicitly designed for industrial applicability. It employs commercially available raw materials, operates under mild temperature conditions (20-80°C), and avoids hazardous catalytic systems, making it highly adaptable for scaling from laboratory grams to multi-ton commercial production runs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-1,2,4-Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis technology described in patent CN113480486B for the pharmaceutical industry. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative one-pot methodology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-amino-1,2,4-triazole derivatives meets the highest international standards.
We invite you to collaborate with us to leverage this advanced synthetic route for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing solutions.
