Advanced Catalytic Resolution of Quinoline Axial Chiral Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the construction of axially chiral scaffolds, which serve as critical backbones in numerous bioactive molecules and functional materials. Patent CN107522585B introduces a groundbreaking hydrogenation resolution method specifically designed for quinoline axial chiral compounds, leveraging a sophisticated transfer hydrogenation system. This technology utilizes a chiral phosphoric acid catalyst in conjunction with a Hantzsch ester hydrogen donor to achieve exceptional stereocontrol without the need for harsh reducing agents. The significance of this innovation lies in its ability to produce quinoline-based axial chiral compounds with enantiomeric excess values reaching up to 99%, alongside tetrahydroquinoline derivatives with up to 97% ee. For a reliable quinoline intermediate supplier, mastering such high-selectivity transformations is paramount to delivering materials that meet the stringent purity requirements of modern drug discovery pipelines. The process boasts a remarkable resolution coefficient (S) of up to 209, indicating a highly efficient kinetic resolution that maximizes the utility of the starting racemic material while minimizing waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axial chiral compounds has relied heavily on conventional chemical resolution, cross-coupling reactions, or oxidative coupling strategies that often present significant logistical and economic challenges for large-scale manufacturing. Traditional kinetic resolution methods frequently employ bromination, carbon-hydrogen bond activation, or reductive amination processes that may require stoichiometric amounts of chiral auxiliaries or expensive transition metal catalysts. These legacy approaches often suffer from moderate enantioselectivity, necessitating repetitive recrystallization steps that drastically reduce overall yield and increase production costs. Furthermore, many conventional hydrogenation techniques rely on high-pressure hydrogen gas and precious metal catalysts, introducing safety hazards and potential heavy metal contamination issues that are increasingly unacceptable in the production of high-purity pharmaceutical intermediates. The reliance on such resource-intensive methods creates bottlenecks in the supply chain, leading to extended lead times and inconsistent batch-to-batch quality that can jeopardize downstream clinical development timelines.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a metal-free organocatalytic system that fundamentally shifts the paradigm towards greener and more sustainable chemical manufacturing. By employing chiral phosphoric acid as the catalyst and Hantzsch ester as the hydrogen source, this method achieves resolution under mild conditions, typically at normal pressure and temperatures ranging from 30°C to 60°C. This transition from metal-catalyzed to organocatalyzed processes eliminates the need for rigorous heavy metal scavenging steps, thereby streamlining the purification workflow and significantly reducing the environmental footprint of the synthesis. The operational simplicity of this transfer hydrogenation system allows for easier handling and scalability, as it avoids the specialized equipment required for high-pressure hydrogenation. For procurement teams focused on cost reduction in pharmaceutical intermediate manufacturing, this methodology offers a compelling value proposition by reducing raw material complexity and energy consumption while maintaining superior stereochemical outcomes.
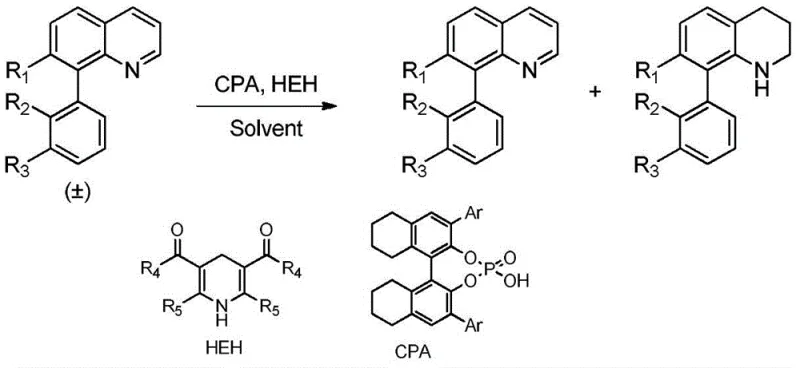
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Transfer Hydrogenation
The core of this technological advancement lies in the precise mechanistic interaction between the chiral phosphoric acid catalyst and the quinoline substrate within the transfer hydrogenation cycle. The chiral phosphoric acid acts as a bifunctional catalyst, simultaneously activating the imine nitrogen of the quinoline ring through hydrogen bonding interactions while orienting the Hantzsch ester for hydride delivery. This dual activation creates a highly organized chiral environment that differentiates between the enantiomers of the racemic substrate with exceptional fidelity. The hydride transfer from the Hantzsch ester to the protonated quinoline intermediate occurs through a well-defined transition state, where the bulky substituents on the phosphoric acid backbone sterically hinder one face of the substrate, forcing the reaction to proceed with high enantioselectivity. Understanding this mechanism is crucial for R&D directors evaluating the feasibility of adapting this route for novel analogs, as it highlights the tunability of the catalyst structure to accommodate various steric and electronic demands of different substrates.
Furthermore, the impurity control mechanism inherent in this catalytic system is driven by the high resolution coefficient, which ensures that the unreacted starting material and the reduced product possess distinct and high optical purities. The kinetic nature of the resolution means that one enantiomer reacts significantly faster than the other, allowing for the isolation of both the enriched starting material and the chiral product from a single reaction run. This dual-output capability effectively doubles the value of the input material compared to traditional resolutions where half of the material is often discarded or requires racemization. The mild reaction conditions also minimize the formation of side products such as over-reduced species or decomposition byproducts, which are common issues in high-temperature or high-pressure hydrogenation processes. Consequently, the resulting crude reaction mixtures are cleaner, facilitating simpler workup procedures and higher recovery rates of the target high-purity axial chiral compounds.
How to Synthesize Quinoline Axial Chiral Compounds Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the resolution efficiency and enantiomeric excess. The standard protocol involves conducting the reaction under an inert nitrogen atmosphere to prevent oxidation of the sensitive Hantzsch ester and ensure consistent catalytic activity throughout the process. Operators must precisely weigh the chiral phosphoric acid catalyst at 5 mol% relative to the substrate and add 1.2 equivalents of the Hantzsch ester to drive the equilibrium towards the desired reduced product without excessive waste. The choice of solvent plays a critical role, with dichloromethane demonstrating optimal performance in balancing solubility and reaction rate, although other solvents like benzene or THF can be evaluated depending on specific substrate properties. Detailed standardized synthetic steps see the guide below for exact procedural specifications regarding temperature control and reaction monitoring.
- Prepare the reaction mixture under nitrogen protection by adding the substrate quinoline racemic axis chiral compound and chiral phosphoric acid catalyst (5 mol%).
- Introduce Hantzsch ester (1.2 equivalents) as the hydrogen source and add dichloromethane solvent (3.0 ml per 0.1 mmol substrate).
- Stir the reaction at 30-60°C for 20-24 hours, then remove solvent and purify via column chromatography to isolate high-ee products.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this organocatalytic resolution technology offers substantial benefits that extend beyond mere technical performance metrics. The elimination of precious metal catalysts directly correlates to a significant reduction in raw material costs, as chiral phosphoric acids and Hantzsch esters are generally more affordable and stable than their transition metal counterparts. This shift also mitigates supply chain risks associated with the volatility of precious metal markets and the geopolitical constraints often affecting the availability of catalysts like palladium or rhodium. Additionally, the mild operating conditions reduce energy consumption and lower the safety barriers for production, allowing for manufacturing in facilities that may not be equipped for high-pressure hydrogenation. These factors collectively contribute to a more resilient and cost-effective supply chain for complex chiral building blocks.
- Cost Reduction in Manufacturing: The removal of expensive transition metals from the catalytic cycle eliminates the need for costly metal scavenging resins and extensive purification protocols required to meet strict residual metal limits. This simplification of the downstream processing workflow leads to substantial cost savings in both consumables and labor hours, while the high resolution coefficient ensures that the starting material is utilized efficiently, reducing the effective cost per gram of the chiral product. Furthermore, the commercial availability of the catalyst components ensures stable pricing and avoids the premiums often associated with specialized ligand-metal complexes.
- Enhanced Supply Chain Reliability: The use of commercially available reagents and standard organic solvents like dichloromethane ensures that the supply chain is not dependent on single-source or hard-to-procure specialty chemicals. The robustness of the reaction under normal pressure and moderate temperatures means that production is less susceptible to disruptions caused by equipment failure or safety shutdowns common in high-pressure operations. This reliability translates to more consistent delivery schedules and the ability to scale production volumes rapidly in response to fluctuating market demand without requiring significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with increasing regulatory pressures to reduce heavy metal discharge in pharmaceutical wastewater. By avoiding toxic metals, the environmental burden of waste treatment is drastically simplified, facilitating easier compliance with global environmental standards. The scalability is further enhanced by the homogeneous nature of the catalytic system, which allows for straightforward translation from laboratory bench scale to multi-ton commercial production without the mass transfer limitations often encountered in heterogeneous hydrogenation systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation resolution technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Stakeholders can use this information to assess the fit of this technology for their specific project requirements regarding purity, cost, and timeline.
Q: What is the enantiomeric excess achievable with this hydrogenation resolution method?
A: According to patent CN107522585B, this method can achieve an enantiomeric excess (ee) of up to 99% for quinoline-based axial chiral compounds and up to 97% for tetrahydroquinoline derivatives, with a resolution coefficient (S) reaching 209.
Q: Does this process require expensive transition metal catalysts?
A: No, the process utilizes a chiral phosphoric acid organocatalyst and Hantzsch ester, eliminating the need for costly precious metals like palladium or platinum, which significantly simplifies downstream purification and reduces heavy metal contamination risks.
Q: What are the typical reaction conditions for this resolution?
A: The reaction operates under mild conditions, specifically at normal pressure and temperatures between 30-60°C, using common organic solvents like dichloromethane, making it highly suitable for safe industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Axial Chiral Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates to accelerate drug development and commercialization. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN107522585B can be successfully translated into robust manufacturing processes. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of quinoline derivatives we produce. We understand that consistency is key, and our team is dedicated to maintaining the highest standards of quality management to support your long-term supply needs.
We invite you to collaborate with us to leverage this advanced resolution technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs while delivering the high-purity materials your research demands.
