Advanced Photocatalytic Synthesis of Loxanol Derivatives for High-Potency Anticancer Drug Development
Introduction to Novel Anticancer Scaffold Technology
The pharmaceutical industry is constantly seeking novel scaffolds that offer high potency with reduced toxicity, particularly in the realm of oncology. Patent CN113149942A introduces a groundbreaking series of loxanol phenolic hydroxyl derivatives, characterized by the general structural formula (I), which demonstrate exceptional nanomolar activity against colorectal cancer and leukemia. These compounds represent a significant advancement over existing chemotherapy agents, which often suffer from narrow therapeutic windows and severe side effects such as bone marrow suppression. The core innovation lies in the specific substitution patterns at the phenolic hydroxyl group, where R can vary from simple alkyl chains to complex acyl or sulfonyl groups, allowing for fine-tuning of pharmacokinetic properties. 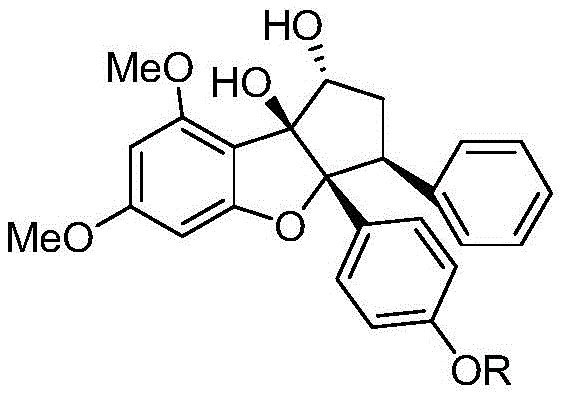 This structural flexibility enables the development of a diverse library of candidates, with specific embodiments like compound 11k showing profound efficacy in inhibiting the MAPK signaling pathway. For R&D directors and procurement specialists, understanding the synthetic accessibility of these complex molecules is crucial for evaluating their potential as viable drug candidates or high-value intermediates.
This structural flexibility enables the development of a diverse library of candidates, with specific embodiments like compound 11k showing profound efficacy in inhibiting the MAPK signaling pathway. For R&D directors and procurement specialists, understanding the synthetic accessibility of these complex molecules is crucial for evaluating their potential as viable drug candidates or high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of rocaglate-type natural products and their analogues often relies on lengthy linear sequences involving multiple protection and deprotection steps, which drastically reduce overall yield and increase waste generation. Conventional routes frequently utilize harsh acidic or basic conditions that can compromise sensitive functional groups, leading to complex impurity profiles that are difficult to purify on a commercial scale. Furthermore, many established methods depend on expensive transition metal catalysts that require rigorous removal processes to meet stringent pharmaceutical purity standards, adding significant cost and time to the manufacturing timeline. The construction of the characteristic cyclopenta[b]benzofuran core, essential for biological activity, has historically been a bottleneck, often requiring low-temperature cryogenic conditions or specialized reagents that are not suitable for large-scale production. These inefficiencies create substantial barriers to entry for generic manufacturers and limit the availability of high-quality intermediates for clinical research.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by employing a visible-light mediated photocatalytic [3+2] cycloaddition strategy to construct the core skeleton efficiently. This approach bypasses the need for cryogenic conditions and expensive chiral catalysts in the initial ring-forming steps, utilizing instead a xenon lamp to drive the reaction between a flavone intermediate and trans-methyl cinnamate. The process integrates a pinacol-like rearrangement subsequent to the photocycloaddition, which elegantly establishes the necessary stereochemistry and quaternary carbon centers in a single operational sequence. By streamlining the synthesis into fewer steps with higher atom economy, this novel route significantly reduces the environmental footprint and operational complexity associated with traditional methods. The ability to generate diverse derivatives through simple alkylation or acylation of the phenolic hydroxyl group further enhances the versatility of this platform, making it an ideal candidate for rapid structure-activity relationship (SAR) studies and commercial scale-up.
Mechanistic Insights into Photocatalytic [3+2] Cycloaddition
The heart of this synthetic innovation is the photochemical transformation of intermediate 4 into the tautomeric intermediates 5 and 6. Under the irradiation of a xenon lamp, the excited state of the flavone derivative undergoes a concerted [3+2] cycloaddition with the alkene moiety of trans-methyl cinnamate. This pericyclic reaction is highly stereoselective, dictated by the orbital symmetry and the steric environment of the substituents, resulting in the formation of a complex fused ring system with high fidelity. ![Photocatalytic [3+2] cycloaddition reaction scheme converting intermediate 4 to 5/6](/insights/img/loxanol-derivatives-photocatalytic-synthesis-pharma-supplier-20260308032831-04.webp) The mechanism avoids the use of external photosensitizers, relying on the intrinsic photophysical properties of the substrate, which simplifies the reaction mixture and downstream purification. Following the cycloaddition, the treatment with sodium methoxide induces a skeletal rearrangement akin to a pinacol shift, migrating the carbonyl group and establishing the critical ketone functionality found in intermediate 7. This cascade of events demonstrates a sophisticated command of physical organic chemistry, leveraging light energy to overcome activation barriers that would otherwise require thermal conditions detrimental to the molecule's stability.
The mechanism avoids the use of external photosensitizers, relying on the intrinsic photophysical properties of the substrate, which simplifies the reaction mixture and downstream purification. Following the cycloaddition, the treatment with sodium methoxide induces a skeletal rearrangement akin to a pinacol shift, migrating the carbonyl group and establishing the critical ketone functionality found in intermediate 7. This cascade of events demonstrates a sophisticated command of physical organic chemistry, leveraging light energy to overcome activation barriers that would otherwise require thermal conditions detrimental to the molecule's stability.
Impurity control in this process is managed through careful optimization of the irradiation time and the stoichiometry of the alkene partner. The patent details specific workup procedures, such as quenching with saturated ammonium chloride and utilizing flash column chromatography with chloroform-acetone mixtures, to isolate the desired diastereomers effectively. The subsequent reduction of the ketone in intermediate 8 using sodium triacetoxyborohydride is chemoselective, reducing the carbonyl without affecting the benzyl ether protecting groups or the aromatic rings. This selectivity is paramount for maintaining the integrity of the molecule and ensuring that the final product, such as the 4'-demethoxy-loxanol precursor (10), meets the high purity specifications required for biological testing. The robust nature of these reactions suggests that impurity profiles can be tightly controlled, a key consideration for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Loxanol Derivatives Efficiently
The synthesis of these high-value anticancer intermediates follows a logical progression from readily available starting materials to the final functionalized derivatives. The process begins with a Claisen-Schmidt condensation to form the chalcone backbone, followed by oxidative cyclization to generate the flavone core. The critical photocycloaddition step then builds molecular complexity, which is subsequently refined through rearrangement, reduction, and deprotection. This modular approach allows for the late-stage diversification of the phenolic hydroxyl group, enabling the rapid generation of a library of analogues for screening purposes.
- Condense 2-hydroxy-4,6-dimethoxyacetophenone with 4-benzyloxybenzaldehyde using potassium hydroxide in ethanol, followed by iodine-mediated cyclization in pyridine to form the flavone intermediate.
- Perform a photocatalytic [3+2] cycloaddition between the flavone intermediate and trans-methyl cinnamate under xenon lamp irradiation to construct the complex cyclopentane-fused core.
- Execute pinacol-like rearrangement using sodium methoxide, followed by demethylation with lithium chloride and reduction with sodium triacetoxyborohydride to yield the active precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical implications of this synthetic route are highly favorable. The reliance on commodity chemicals such as acetophenones and benzaldehydes ensures a stable and cost-effective supply of raw materials, mitigating the risks associated with sourcing exotic or proprietary reagents. The elimination of precious metal catalysts in the key bond-forming steps removes the need for expensive scavenging resins and extensive metal analysis, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the use of standard solvents like ethanol, acetonitrile, and dichloromethane aligns with existing infrastructure in most chemical production facilities, facilitating seamless technology transfer and scale-up without the need for specialized equipment investments.
- Cost Reduction in Manufacturing: The streamlined synthetic sequence significantly lowers the cost of goods sold (COGS) by reducing the total number of unit operations and minimizing solvent consumption. By avoiding the use of stoichiometric amounts of expensive chiral auxiliaries or rare earth metals, the process achieves substantial cost savings while maintaining high stereochemical integrity. The high yields reported in the patent examples, such as the 76.9% yield in the rearrangement step, indicate a robust process that maximizes material throughput and minimizes waste disposal costs. Additionally, the ability to perform the derivatization step under mild conditions reduces energy consumption, further enhancing the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: The synthetic route is designed with supply chain resilience in mind, utilizing reagents that are globally available from multiple vendors. This redundancy prevents bottlenecks that often occur when relying on single-source suppliers for specialized catalysts. The robustness of the photocatalytic step, which does not require stringent exclusion of moisture or oxygen beyond standard inert atmosphere techniques, simplifies operational protocols and reduces the likelihood of batch failures due to environmental factors. Consequently, manufacturers can offer more reliable lead times and consistent quality, which is critical for maintaining uninterrupted clinical trial supplies and commercial production schedules.
- Scalability and Environmental Compliance: From an environmental perspective, this method offers a greener alternative to traditional synthesis by reducing the E-factor through higher atom economy and fewer purification steps. The avoidance of heavy metal waste streams simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations. The process is amenable to continuous flow processing, particularly the photochemical step, which can overcome light penetration limitations associated with large batch reactors. This scalability ensures that the production capacity can be easily expanded to meet growing market demand for these potent anticancer agents without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of loxanol derivatives. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation. Understanding these details is essential for partners looking to integrate this technology into their own development pipelines.
Q: What is the biological activity profile of the synthesized loxanol derivatives?
A: The synthesized derivatives, particularly compound 11k, exhibit nanomolar-level activity against colorectal cancer (HCT116) and leukemia (HEL) cell lines. They function by inhibiting the MAPK signaling pathway and inducing cell cycle arrest at the G1 phase.
Q: How does the photocatalytic step impact the scalability of this process?
A: While photocatalysis traditionally poses scale-up challenges, the use of a xenon lamp and standard solvents like acetonitrile allows for adaptable flow chemistry or batch reactor configurations, ensuring consistent yield without requiring exotic catalysts.
Q: Are the starting materials for this synthesis readily available?
A: Yes, the primary raw materials, including 2-hydroxy-4,6-dimethoxyacetophenone and 4-benzyloxybenzaldehyde, are commercially accessible commodity chemicals, which significantly reduces supply chain risk and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Loxanol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the loxanol scaffold in next-generation oncology therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle complex photochemical reactions and sensitive organometallic transformations, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality intermediates. We understand that time-to-market is critical in the competitive landscape of cancer drug development, and our dedicated technical team is committed to accelerating your timeline through optimized process chemistry.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific drug discovery needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance your supply chain efficiency and reduce your overall development costs. Let us be your strategic partner in bringing these promising anticancer candidates to patients worldwide.
