Advanced Synthesis of Potent Thioglycoside SGLT2 Inhibitors for Commercial Scale-up
Advanced Synthesis of Potent Thioglycoside SGLT2 Inhibitors for Commercial Scale-up
The pharmaceutical landscape for Type 2 diabetes treatment is continuously evolving, driven by the need for agents that offer superior efficacy and metabolic stability. Patent CN114933619A introduces a groundbreaking class of thioglycoside analogs designed as potent SGLT2 inhibitors. Unlike earlier generations of glucose-lowering agents that relied on oxygen-linked glycosidic bonds prone to enzymatic degradation, this innovation leverages a robust sulfur linkage. This structural modification not only enhances resistance to beta-glucosidase hydrolysis but also maintains nanomolar inhibitory potency comparable to leading commercial drugs. For R&D directors and procurement specialists, this patent represents a significant opportunity to access high-purity pharmaceutical intermediates with a streamlined synthetic route that minimizes processing complexity while maximizing therapeutic potential.
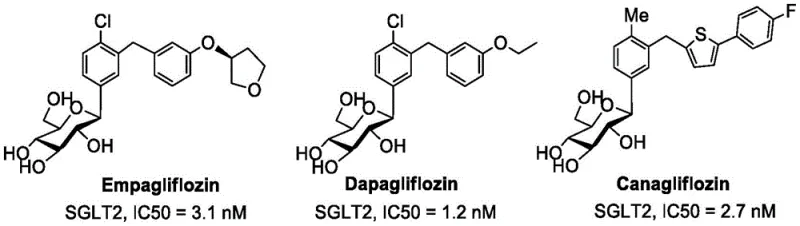
The current market is dominated by gliflozin-class drugs such as Empagliflozin, Dapagliflozin, and Canagliflozin, which function by inhibiting glucose reabsorption in the kidneys. While effective, the synthesis of these carbon-linked or oxygen-linked analogs often involves multi-step sequences with stringent stereochemical control. The technology disclosed in CN114933619A addresses these challenges by proposing a novel thioglycoside scaffold. This approach is particularly relevant for supply chain heads looking for reliable SGLT2 inhibitor intermediate suppliers, as the described methodology simplifies the connection between the sugar moiety and the aglycone, potentially reducing lead time for high-purity pharmaceutical intermediates and lowering overall production costs through improved yield efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of SGLT2 inhibitors faced a critical hurdle regarding metabolic stability. Early O-glycoside derivatives, modeled after natural phlorizin, were rapidly hydrolyzed by beta-glucosidase enzymes in the gastrointestinal tract, rendering them ineffective as oral medications. To overcome this, the industry shifted towards C-glycosides, which are stable but often require harsh reaction conditions and expensive organometallic reagents for the carbon-carbon bond formation. Furthermore, initial attempts to synthesize thioglycoside analogs, such as the meta-substituted structures designated as Analogues A, B, and C in prior art, resulted in disappointing biological activity. These earlier candidates exhibited inhibition rates of only 50-60% even at high concentrations of 100 μM, failing to meet the potency thresholds required for clinical viability. This lack of efficacy, combined with the synthetic challenges of traditional glycosylation, created a bottleneck in the cost reduction in pharmaceutical manufacturing for next-generation diabetes therapies.
The Novel Approach
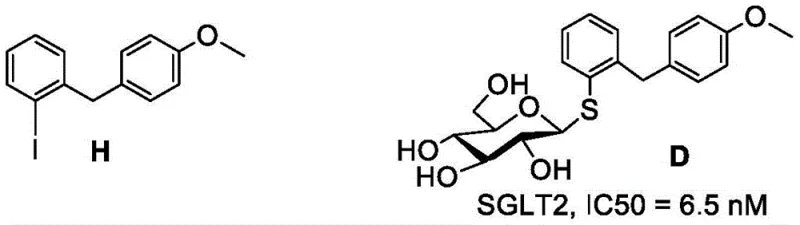
The invention disclosed in CN114933619A circumvents these limitations through a strategic ortho-substitution design. By linking the glucose 1-thiol to the ortho-position of the benzene ring alkyl of the aglycone, the inventors have achieved a dramatic improvement in biological activity. The resulting analogues, specifically compounds D, E, F, and G, demonstrate nearly 100% inhibition of SGLT2 at 100 μM concentrations, with IC50 values ranging from 3.4 nM to 6.5 nM. This potency is on par with, and in some cases exceeds, that of the reference drug Canagliflozin. The structural integrity of these molecules is further bolstered by the sulfur linkage, which confers exceptional stability against enzymatic hydrolysis without the need for complex carbon-carbon bond forming reactions. This novel approach not only solves the stability issue inherent in O-glycosides but also offers a more direct and efficient synthetic pathway compared to C-glycosides, positioning it as a superior candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed C-S Bond Formation
The core of this synthetic innovation lies in the efficient construction of the C-S glycosidic bond using a palladium-catalyzed cross-coupling reaction. This transformation is achieved under remarkably mild conditions, utilizing tetraacetyl glucose 1-thiol and specific iodoaryl derivatives as key building blocks. The reaction proceeds in tetrahydrofuran (THF) at room temperature, facilitated by a specialized palladium catalyst system and triethylamine as a base. The use of room temperature conditions is a significant advantage, as it eliminates the energy costs associated with heating and reduces the risk of thermal degradation of sensitive carbohydrate substrates. The catalyst, often a Xantphos-based palladium complex, ensures high regioselectivity and conversion rates, driving the reaction to completion within 1 to 4 hours. This mechanistic efficiency is crucial for maintaining the stereochemical integrity of the sugar moiety, which is essential for the biological activity of the final SGLT2 inhibitor.
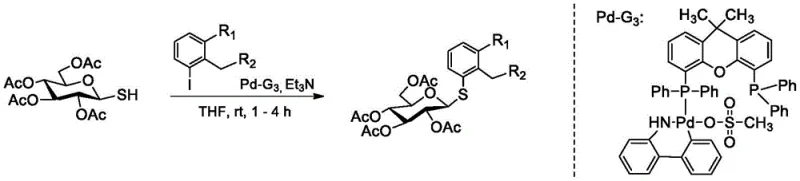
Following the coupling step, the synthesis involves a straightforward deprotection sequence to reveal the free hydroxyl groups necessary for biological interaction. The tetraacetyl-protected intermediate is treated with a sodium hydroxide solution in methanol, a standard saponification protocol that cleaves the ester bonds without affecting the robust C-S linkage. This step typically proceeds with near-quantitative yields, often exceeding 97%, highlighting the chemical robustness of the thioglycoside core. The combination of a high-yielding coupling reaction followed by an efficient deprotection step results in an overall process that is both atom-economical and operationally simple. For process chemists, this translates to fewer purification burdens and a cleaner impurity profile, which is vital for meeting the stringent purity specifications required for API intermediates. The ability to perform these transformations without cryogenic conditions or exotic reagents significantly lowers the barrier for entry in terms of equipment and safety requirements.
How to Synthesize Thioglycoside Analogs Efficiently
The synthesis of these high-potency thioglycoside analogs is designed for operational simplicity and scalability. The process begins with the preparation of the protected intermediate via the palladium-catalyzed coupling described above, followed by a basic hydrolysis to yield the final active compound. The detailed standardized synthesis steps, including precise molar ratios, solvent volumes, and workup procedures, are outlined below to ensure reproducibility and quality control for manufacturing teams.
- Perform a palladium-catalyzed cross-coupling reaction between tetraacetyl glucose 1-thiol and an iodoaryl derivative in THF with triethylamine at room temperature.
- Purify the resulting tetraacetyl-protected intermediate using column chromatography.
- Deprotect the acetyl groups using sodium hydroxide in methanol to yield the final thioglycoside analog.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this thioglycoside synthesis route offers tangible economic and logistical benefits. The primary advantage lies in the significant simplification of the manufacturing process. By avoiding the harsh conditions and multiple steps often required for C-glycoside synthesis, manufacturers can achieve substantial cost savings in raw material consumption and energy usage. The high yields reported in the patent examples, particularly the near-quantitative recovery in the deprotection step, minimize waste generation and maximize the throughput of valuable intermediates. This efficiency directly contributes to cost reduction in pharmaceutical manufacturing, allowing for more competitive pricing of the final API. Furthermore, the use of commercially available starting materials, such as tetraacetyl glucose thiol and various iodoaryl derivatives, ensures a stable and reliable supply chain, reducing the risk of bottlenecks associated with custom-synthesized reagents.
- Cost Reduction in Manufacturing: The elimination of energy-intensive heating steps and the use of mild room temperature reactions significantly lower utility costs. Additionally, the high conversion rates reduce the need for extensive recycling of unreacted starting materials, streamlining the production budget and enhancing overall process economics.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard catalytic systems means that sourcing is less vulnerable to geopolitical or logistical disruptions. The robustness of the reaction conditions also allows for flexibility in manufacturing locations, enabling regional production hubs to maintain consistent supply continuity for global markets.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional glycosylation methods that might require heavy metal promoters or strong Lewis acids. The aqueous workup and standard organic solvents used are compatible with existing waste treatment infrastructure, facilitating easier regulatory compliance and smoother scale-up from pilot plant to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the thioglycoside analogs described in patent CN114933619A. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the stability, potency, and manufacturability of these novel compounds for potential licensees and partners.
Q: How does the stability of these thioglycoside analogs compare to traditional O-glycosides?
A: Unlike traditional O-glycosides which are susceptible to hydrolysis by beta-glucosidase in the small intestine, these thioglycoside analogs feature a sulfur linkage that provides exceptional resistance to enzymatic hydrolysis, ensuring better bioavailability and metabolic stability.
Q: What is the inhibitory potency of the new analogs compared to marketed drugs?
A: The novel analogs (specifically compounds D, E, F, and G) exhibit IC50 values in the low nanomolar range (3.4 nM to 6.5 nM), which is comparable to or better than established drugs like Canagliflozin (2.7 nM), whereas previous meta-substituted attempts showed poor activity.
Q: Is the synthesis process suitable for large-scale manufacturing?
A: Yes, the process utilizes mild reaction conditions (room temperature), readily available starting materials, and achieves high yields (up to 99% in deprotection steps), making it highly amenable to commercial scale-up with reduced energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioglycoside Analogs Supplier
As the demand for next-generation antidiabetic agents grows, partnering with an experienced CDMO is essential for translating patent innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless. Our facility is equipped with rigorous QC labs capable of verifying the stringent purity specifications required for SGLT2 inhibitor intermediates, guaranteeing that every batch meets the highest international standards for safety and efficacy. We understand the critical nature of supply chain continuity and are committed to delivering high-quality intermediates that support your drug development timelines.
We invite you to collaborate with us to leverage this advanced thioglycoside technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your path to market.
