Scalable Synthesis of Symmetric DiFmoc-Lys Plus Linker for Commercial MAPS Polypeptide Production
Scalable Synthesis of Symmetric DiFmoc-Lys Plus Linker for Commercial MAPS Polypeptide Production
The landscape of synthetic vaccine development is undergoing a significant transformation driven by the need for higher purity and more robust immunogenic carriers. Patent CN111233714B introduces a groundbreaking advancement in the field of Multiple Antigen Peptide Systems (MAPS) by disclosing a novel unnatural amino acid linker known as DiFmoc-Lys (plus). This innovation directly addresses the longstanding bottleneck in branched peptide synthesis where traditional lysine cores suffer from structural asymmetry. By engineering a linker where both bifurcate arms are completely consistent in length and chemical structure, this technology ensures that subsequent amino acid coupling reactions proceed with uniform efficiency. For R&D directors and procurement strategists, this represents a pivotal shift towards more predictable manufacturing outcomes, drastically reducing the formation of deletion sequences that typically complicate downstream purification. The implementation of this symmetric core is not merely a chemical curiosity but a fundamental process improvement that enhances the overall success rate of MAPS polypeptide production.
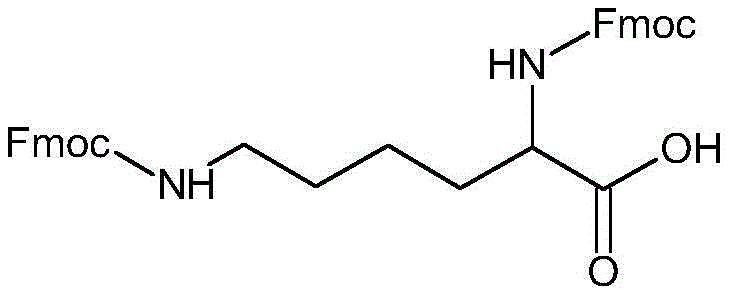
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the traditional synthesis of Multiple Antigen Peptides, the industry has relied heavily on the natural amino acid Lysine as a branching core. However, this approach is inherently flawed due to the distinct chemical environments of the alpha and epsilon amino groups. The alpha-amino group is adjacent to the carboxyl functionality, creating significant steric hindrance and electronic differences compared to the epsilon-amino group located at the end of the side chain. Consequently, when antigen peptides are coupled to these sites, the reaction kinetics are uneven; the shorter, more hindered arm reacts slower than the longer, more accessible arm. This disparity leads to a heterogeneous mixture of products where some branches are fully elongated while others are truncated. Such heterogeneity creates a nightmare for quality control, necessitating rigorous and costly HPLC purification steps to isolate the desired octameric structure from a sea of impurities. Furthermore, previous attempts to mitigate this by adding beta-alanine spacers introduced amide bonds that, while helpful, did not achieve perfect structural uniformity, leaving residual efficiency gaps in the synthesis cycle.
The Novel Approach
The methodology outlined in CN111233714B circumvents these intrinsic limitations by constructing a completely synthetic, symmetric lysine analogue. Instead of relying on the natural asymmetry of lysine, the inventors have designed DiFmoc-Lys (plus) to possess two identical branching arms, effectively mirroring the structure on both sides of the central alpha-carbon. This structural symmetry ensures that the steric environment around both reactive amino groups is identical, thereby normalizing the coupling efficiency for every subsequent amino acid addition. From a process chemistry perspective, this eliminates the kinetic variance that plagues conventional MAPS synthesis. The result is a crude product with significantly higher homogeneity, meaning the burden on downstream purification is substantially reduced. For a reliable MAPS polypeptide supplier, this translates to a more streamlined workflow where the focus shifts from salvaging yield through complex purification to optimizing the initial coupling efficiency. This approach not only improves the final purity specifications but also enhances the reproducibility of the synthesis across different batches, a critical factor for regulatory compliance in vaccine manufacturing.
Mechanistic Insights into Grignard-Mediated Symmetric Core Construction
The synthesis of the DiFmoc-Lys (plus) linker is a masterclass in leveraging organometallic chemistry to solve steric problems in peptide science. The process begins with the conversion of Fmoc-butanediamine into a Grignard reagent, specifically N-Fmoc-4-bromobutylamine magnesium bromide. This step is crucial as it activates the carbon chain for nucleophilic attack. Simultaneously, the lysine derivative NH2-Lys(Fmoc)-Otbu is transformed into an electrophilic partner, I-Lys(Fmoc)-Otbu, through a diazotization-iodination sequence using sodium nitrite and iodine. The convergence of these two fragments via a Grignard coupling reaction allows for the precise construction of the extended side chain. By attaching a four-carbon chain bearing a protected amine to the alpha-position of the lysine derivative, the chemists effectively extend the shorter alpha-arm to match the length of the native epsilon-arm. This mechanistic strategy avoids the introduction of extraneous amide bonds found in beta-alanine modifications, preserving the flexibility and chemical nature of the backbone. The final deprotection step using trifluoroacetic acid (TFA) cleanly removes the tert-butyl ester, yielding the free acid form ready for resin loading without compromising the integrity of the Fmoc protecting groups on the amines.
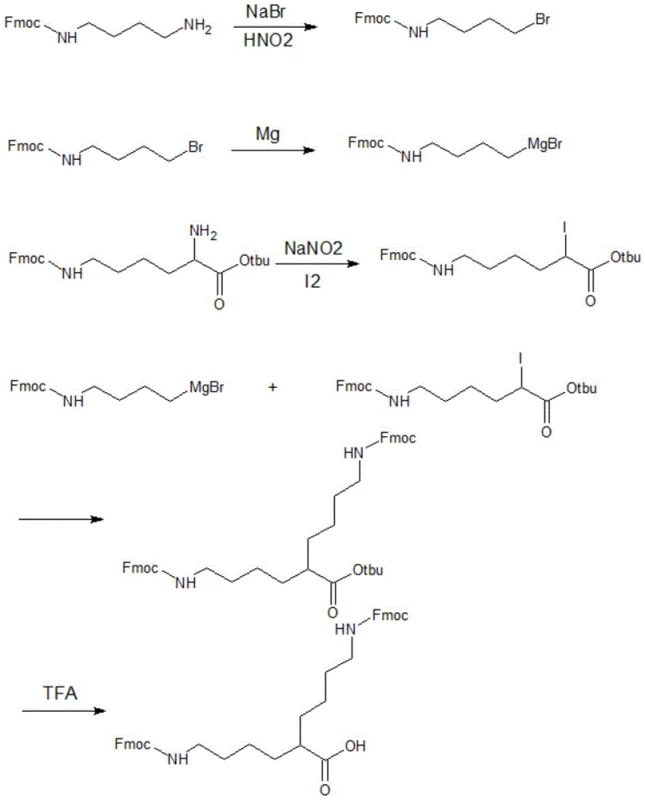
Controlling impurities during this synthesis is paramount, particularly given the sensitivity of Grignard reagents to moisture and protic solvents. The protocol specifies the use of anhydrous conditions and precise stoichiometric control to minimize side reactions such as Wurtz coupling or protonation of the organometallic species. The use of the tert-butyl ester protecting group on the lysine carboxylate is a strategic choice, as it provides orthogonality to the Fmoc amine protection, allowing for selective manipulation of the molecule. This orthogonal protection scheme ensures that the final linker can be seamlessly integrated into standard Fmoc-based solid-phase peptide synthesis (SPPS) workflows. For R&D teams, understanding this mechanism highlights the robustness of the route; it relies on well-established organic transformations that are scalable and do not require exotic catalysts. The high purity of the resulting linker is a direct consequence of this clean reaction pathway, which minimizes the formation of regioisomers or over-alkylated byproducts that could otherwise propagate errors through the entire peptide assembly line.
How to Synthesize DiFmoc-Lys (plus) Efficiently
The preparation of this novel linker involves a convergent strategy that merges a functionalized alkyl chain with a protected lysine core. The process is designed to be operationally simple while maintaining high fidelity in structural control. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility in your laboratory setting.
- Convert Fmoc-butanediamine to N-Fmoc-4-bromobutylamine using NaBr and HNO2, then react with Magnesium to form the Grignard reagent.
- Transform NH2-Lys(Fmoc)-Otbu into the iodo-derivative I-Lys(Fmoc)-Otbu using NaNO2 and Iodine catalysis.
- Couple the Grignard reagent with the iodo-lysine derivative, followed by TFA acidolysis to remove the tert-butyl ester and yield DiFmoc-Lys (plus).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the DiFmoc-Lys (plus) technology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing process for complex branched peptides. By ensuring uniform coupling efficiency, the process inherently reduces the consumption of expensive protected amino acids that are often wasted in conventional methods due to incomplete reactions on hindered sites. This efficiency gain translates directly into cost reduction in vaccine antigen manufacturing, as fewer raw materials are required to achieve the same theoretical yield. Furthermore, the improved homogeneity of the crude product means that purification resources, such as HPLC column life and solvent consumption, are utilized much more effectively. This leads to a significant optimization of the overall cost of goods sold (COGS), making high-quality MAPS vaccines more economically viable for large-scale immunization programs.
- Cost Reduction in Manufacturing: The elimination of asymmetric coupling inefficiencies means that the molar excess of reagents typically required to drive difficult couplings can be reduced. In traditional MAPS synthesis, large excesses of activated amino acids are often needed to overcome the steric hindrance of the alpha-branch, leading to substantial material waste. With the symmetric DiFmoc-Lys (plus) linker, reaction kinetics are normalized, allowing for more stoichiometric usage of precious building blocks. Additionally, the reduction in deletion sequences minimizes the loss of product during the purification phase, effectively increasing the overall process yield without requiring additional capital investment in equipment. This qualitative improvement in material efficiency creates a leaner manufacturing profile that is highly attractive for cost-sensitive pharmaceutical projects.
- Enhanced Supply Chain Reliability: The synthesis route for the linker relies on commodity chemicals such as magnesium, sodium bromide, and iodine, which are readily available in the global market. Unlike processes that depend on scarce transition metal catalysts or specialized enzymes, this method ensures a stable supply of the core linker. The robustness of the chemistry also implies a lower risk of batch failure due to reagent sensitivity, thereby enhancing the predictability of delivery schedules. For supply chain planners, this reliability is crucial for maintaining continuous production lines for vaccine antigens. The ability to source raw materials easily and execute the synthesis with high consistency reduces the lead time for high-purity immunogens, ensuring that downstream formulation teams receive their materials on schedule.
- Scalability and Environmental Compliance: The process is inherently scalable, moving smoothly from gram-scale laboratory optimization to kilogram-scale commercial production. The use of standard solvents like DMF and THF, combined with straightforward workup procedures involving acidolysis, fits well within existing GMP facilities. Moreover, by improving the overall yield and purity, the process generates less chemical waste per unit of active product. This reduction in waste volume simplifies effluent treatment and aligns with increasingly stringent environmental regulations. The commercial scale-up of complex peptide linkers is often hindered by heat transfer and mixing issues in heterogeneous reactions, but the solution-phase synthesis of this linker avoids many of these pitfalls, offering a clear path to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel linker technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners.
Q: Why is the DiFmoc-Lys (plus) linker superior to traditional Lysine cores for MAPS?
A: Traditional Lysine cores have inconsistent arm lengths between the alpha and epsilon amino groups, causing uneven coupling efficiency. DiFmoc-Lys (plus) features structurally identical arms, ensuring uniform reaction kinetics and higher crude purity.
Q: What is the impact of this linker on downstream purification costs?
A: By eliminating deletion sequences caused by steric hindrance differences, the process significantly reduces the burden on HPLC purification, leading to higher recovery rates of the refined polypeptide product.
Q: Is this synthesis method scalable for industrial vaccine production?
A: Yes, the method utilizes standard reagents like Magnesium, NaBr, and TFA, and is compatible with standard Fmoc solid-phase synthesis protocols, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DiFmoc-Lys (plus) Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to market launch. Our facilities are equipped with state-of-the-art reactors and rigorous QC labs capable of meeting the stringent purity specifications required for vaccine intermediates. We understand that the synthesis of branched peptides like MAPS demands precision, and our team is adept at managing the nuances of Fmoc chemistry to deliver products that consistently meet your quality targets. By leveraging our infrastructure, you can mitigate the risks associated with process scale-up and focus on your core competencies in drug development.
We invite you to engage with our technical procurement team to discuss how this innovative linker can optimize your specific vaccine pipeline. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies gained by switching to the DiFmoc-Lys (plus) architecture. Please contact us to request specific COA data from our pilot batches and comprehensive route feasibility assessments tailored to your project timelines. Let us collaborate to bring safer and more effective synthetic vaccines to the global market through superior chemical manufacturing.
