Advanced Refining Technology for High-Purity Indocyanine Green: Ensuring Injectable Grade Quality and Commercial Scalability
The pharmaceutical industry continuously demands higher purity standards for diagnostic agents, particularly for injectable formulations where safety margins are non-negotiable. Patent CN112638873A introduces a groundbreaking refining method for indocyanine green, a critical near-infrared fluorescent dye used extensively in medical imaging and liver function testing. This technology addresses the persistent challenge of removing inorganic salt impurities, specifically sodium iodide, and organic impurities like triethylamine salts, which are notoriously difficult to separate due to their structural similarities and solubility profiles. By shifting away from traditional extraction methods that compromise yield, this patent outlines a precise recrystallization protocol using specific solvent pairs. For R&D directors and procurement specialists, this represents a significant leap forward in producing reliable pharmaceutical intermediate supplier materials that meet stringent pharmacopeia requirements without sacrificing production efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of indocyanine green has been plagued by a trade-off between purity and yield, creating substantial bottlenecks for cost reduction in pharmaceutical intermediates manufacturing. Conventional techniques, such as those described in prior art like CN104130178, often rely on washing crude products with acetone. However, sodium iodide has limited solubility in acetone, leading to entrapped impurities where residual sodium iodide levels remain unacceptably high, often exceeding 5%, which violates injectable standards. Alternatively, methods utilizing water-organic solvent extraction, as seen in US2009069573, attempt to leverage solubility differences but fail because indocyanine green itself is highly water-soluble. This results in catastrophic product loss during the aqueous wash steps, with documented yields dropping to approximately 56%, rendering the process economically unviable for large-scale commercial scale-up of complex diagnostic agents.
The Novel Approach
The innovative strategy presented in this patent circumvents these issues by employing a controlled recrystallization technique rather than simple washing or extraction. The core of this method involves dissolving the crude indocyanine green in a 'good' solvent, such as methanol or ethanol, followed by the slow, dropwise addition of a 'poor' solvent like ethyl acetate, acetone, or methyl tert-butyl ether. This specific solvent pairing creates a thermodynamic environment where the target indocyanine green sodium salt precipitates efficiently, while impurities like sodium iodide and the elusive indocyanine green triethylamine salt remain in the supernatant. This approach allows for the simultaneous removal of both inorganic and organic impurities, achieving residual sodium iodide levels below 2.5% and triethylamine content below 0.2%, all while maintaining impressive yields consistently above 85%, thereby solving the long-standing purity-yield paradox.
Mechanistic Insights into Solvent Pair Recrystallization
Understanding the molecular interactions in this refining process is crucial for R&D teams aiming to replicate or scale this technology. The mechanism relies heavily on the differential solubility parameters of the target molecule versus its impurities in mixed solvent systems. Indocyanine green exists primarily as a sodium salt in the final medicinal product, but during synthesis, it can form a triethylamine salt complex due to the presence of sulfonate groups which readily bind with cationized amines. This triethylamine salt impurity is structurally analogous to the target product, making separation via standard chromatography or simple precipitation extremely difficult. The chosen 'good' solvent effectively solvates the polar sulfonate groups of both the target and the impurity, ensuring a homogeneous starting solution. Upon the introduction of the 'poor' solvent, the dielectric constant of the medium shifts, reducing the solubility of the larger, more hydrophobic indocyanine green sodium salt lattice, prompting it to crystallize out selectively.
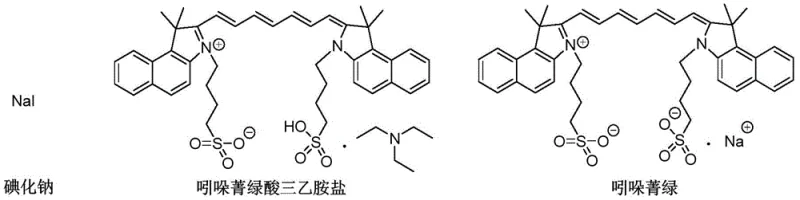
Furthermore, the kinetics of crystallization play a vital role in impurity exclusion. By slowly adding the poor solvent and potentially introducing seed crystals, the process encourages the growth of large, pure crystals of the indocyanine green sodium salt. This slow growth minimizes the occlusion of mother liquor, which contains the dissolved sodium iodide and triethylamine complexes. The structural distinction shown in the visual data highlights the ionic nature of the sodium salt versus the bulkier organic triethylamine salt. The solvent system is tuned to keep these ionic and bulky organic impurities in solution while the target molecule precipitates. This selective precipitation is the key to achieving high-purity indocyanine green suitable for injection, effectively breaking the association between the dye and the triethylamine cation which conventional gas chromatography often fails to detect accurately.
How to Synthesize Indocyanine Green Efficiently
Implementing this refining protocol requires precise control over solvent ratios and temperature to ensure reproducibility and optimal impurity profiles. The process begins with the dissolution of the crude material in a protic solvent like methanol at ambient temperatures, typically between 20°C and 40°C, which balances energy consumption with solubility requirements. Following dissolution, the anti-solvent is introduced gradually to prevent shock precipitation, which could trap impurities within the crystal lattice. While the general concept is straightforward, the specific mass-volume ratios of the crude product to the good solvent and poor solvent are critical parameters that determine the final purity and recovery rates. For detailed operational parameters and specific solvent combinations validated in the patent examples, please refer to the standardized synthesis guide below.
- Dissolve the crude indocyanine green product in a selected good solvent (such as methanol or ethanol) at a controlled temperature between 0°C and 80°C to ensure complete solubilization.
- Slowly add a specific poor solvent (such as acetone, ethyl acetate, or methyl tert-butyl ether) dropwise to the solution under continuous stirring to induce controlled crystallization.
- Allow the mixture to stir for sufficient time to ensure full crystal growth, then filter the precipitate and wash with the poor solvent to obtain high-purity indocyanine green.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refining technology translates directly into enhanced supply security and significant operational efficiencies. The primary economic driver here is the drastic improvement in process yield compared to legacy extraction methods. By avoiding the massive product losses associated with aqueous extractions, manufacturers can significantly reduce the cost of goods sold (COGS) per kilogram of active ingredient. This yield enhancement means that less raw material is required to produce the same amount of finished high-purity indocyanine green, effectively lowering the burden on upstream synthesis capacity and reducing the overall environmental footprint of the manufacturing process through decreased waste generation.
- Cost Reduction in Manufacturing: The elimination of inefficient extraction steps and the high recovery rate of the product lead to substantial cost savings in raw material utilization. Since the process operates effectively at ambient temperatures without the need for extreme heating or complex vacuum distillation setups, energy consumption is also minimized. Furthermore, the ability to remove impurities in a single crystallization step simplifies the workflow, reducing labor hours and equipment occupancy time, which collectively drives down the manufacturing overhead for this critical diagnostic agent.
- Enhanced Supply Chain Reliability: A robust purification method that consistently delivers yields above 85% ensures a more predictable and stable supply of indocyanine green. Unlike processes prone to variable recovery rates due to emulsion formation or solubility fluctuations in extraction, this recrystallization method offers high reproducibility. This reliability is essential for maintaining continuous production schedules for injectable drugs, reducing the risk of stockouts and ensuring that downstream formulation partners receive consistent quality batches without delays caused by re-processing or failed purity tests.
- Scalability and Environmental Compliance: The simplicity of the solvent system, utilizing common industrial solvents like methanol and ethyl acetate, facilitates easy scale-up from pilot to commercial production volumes. The process avoids the use of excessive water, thereby reducing the volume of wastewater requiring treatment, which is a significant advantage for environmental compliance. Additionally, the efficient removal of sodium iodide and triethylamine ensures that the final product meets strict regulatory standards without the need for additional, waste-generating purification cycles, aligning with green chemistry principles and sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this refining technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms traditional techniques in terms of impurity clearance and process efficiency. Understanding these nuances helps stakeholders make informed decisions about integrating this technology into their existing supply chains.
Q: Why is the removal of sodium iodide critical in indocyanine green manufacturing?
A: Sodium iodide is a byproduct of the synthesis process and can cause severe allergic reactions if injected. Pharmacopeia standards strictly limit its content to below 5%, making effective removal essential for patient safety and regulatory compliance.
Q: How does this new refining method improve yield compared to traditional extraction?
A: Traditional water-organic extraction methods often result in significant product loss due to the high water solubility of indocyanine green, yielding as low as 56%. The novel recrystallization method maintains yields above 85% while effectively removing impurities.
Q: What specific impurities does this process target besides sodium iodide?
A: In addition to inorganic salts, this method effectively removes indocyanine green triethylamine salt, a structurally similar organic impurity that is difficult to detect by gas chromatography but poses safety risks in injectable formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indocyanine Green Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN112638873A can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities like sodium iodide and triethylamine salts, guaranteeing that every batch of indocyanine green we supply meets the highest pharmacopeia standards for injectable applications.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced refining technology for your diagnostic imaging portfolios. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this high-yield purification route for your specific volume requirements. Please contact us today to obtain specific COA data from our recent batches and discuss route feasibility assessments tailored to your supply chain needs, ensuring a secure and high-quality source of this vital medical imaging agent.
