Advanced Melt-Acetylation Strategy for High-Purity Isosorbide Mononitrate Impurities and Commercial Scalability
Introduction to Next-Generation Impurity Synthesis
The rigorous control of impurities in active pharmaceutical ingredients (APIs) is a cornerstone of modern drug safety and regulatory compliance. Patent CN110590799B introduces a transformative preparation method for critical isomers, specifically 2-O-acetyl-5-isosorbide dinitrate and 5-O-acetyl-2-isosorbide dinitrate, which serve as essential reference standards for Isosorbide Mononitrate (5-ISMN). This patent details a robust synthetic strategy that bypasses the limitations of traditional enzymatic or harsh chemical methods by utilizing a solvent-free melt acetylation technique. For R&D directors and quality control managers, the ability to access these specific isomers with high purity is vital for establishing accurate impurity profiles, thereby ensuring the therapeutic efficacy and safety of anti-anginal medications. The technology represents a significant leap forward in the manufacturing of pharmaceutical intermediates, offering a pathway that is not only chemically efficient but also aligns with green chemistry principles by minimizing solvent usage during the reaction phase.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these specific isosorbide derivatives has been plagued by inefficiencies and environmental concerns that hinder scalable production. Prior art, such as US Patent 5614643A, relies heavily on enzymatic hydrolysis using lipase from Pseudomonas fluorescens, a process that requires strict pH control via automatic titrators and extended reaction times exceeding 24 hours. Furthermore, subsequent nitration steps often involve hazardous nitrating mixtures at low temperatures, leading to complex workups and variable yields. Alternative chemical routes disclosed in European patent WO2018/172496A utilize organic bases like triethylamine and catalysts like 4-dimethylaminopyridine (DMAP) in dichloromethane. While these methods shorten the route, they generate substantial amounts of acid-containing and alkali-containing wastewater, creating significant disposal costs and environmental liabilities. The reliance on expensive biocatalysts or toxic amine catalysts makes these conventional processes economically unattractive for large-scale supply chains seeking cost-effective solutions.
The Novel Approach
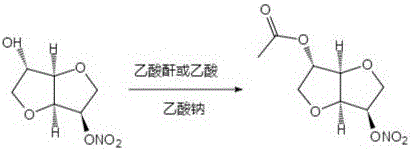
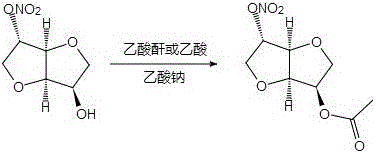
In stark contrast, the methodology outlined in CN110590799B employs a direct acetylation strategy under melt conditions, fundamentally simplifying the reaction engineering. By mixing 5-isosorbide dinitrate or 2-isosorbide dinitrate with a simple sodium acetate catalyst and heating the mixture to 100-105°C, the reactants melt and react directly upon the addition of acetic anhydride or acetic acid. This solvent-free initial state drastically reduces the volume of reaction mass, enhancing thermal efficiency and reaction kinetics. The process operates under reflux for a concise period of 0.5 to 3 hours, achieving completion much faster than enzymatic alternatives. Post-reaction processing involves standard extraction and crystallization techniques, avoiding the need for chromatographic purification which is often a bottleneck in impurity synthesis. This approach not only streamlines the operational workflow but also significantly lowers the E-factor (environmental factor) of the process, making it a superior choice for sustainable manufacturing.
Mechanistic Insights into Sodium Acetate-Catalyzed Melt Acetylation
The core of this technological breakthrough lies in the utilization of sodium acetate as a mild, non-nucleophilic base catalyst within a high-temperature melt matrix. In traditional solution-phase acetylations, the reaction rate is often diffusion-limited by the solvent cage, but in this melt system, the reactants are in a quasi-liquid state with high local concentrations, promoting rapid collision frequency between the hydroxyl groups of the isosorbide nitrate and the acetic anhydride. The sodium acetate facilitates the deprotonation of the hydroxyl group, generating a more nucleophilic alkoxide intermediate in situ without the need for strong, hazardous bases like sodium hydride. This mechanistic pathway ensures high regioselectivity, preferentially acetylating the specific hydroxyl position (either 2-OH or 5-OH depending on the starting isomer) while leaving the nitrate ester groups intact. The stability of the nitrate moiety under these mildly basic, anhydrous conditions is crucial, as stronger bases or higher temperatures could lead to denitration or decomposition, which are common failure modes in nitrate chemistry.
From an impurity control perspective, this mechanism offers distinct advantages in minimizing side reactions. The absence of water during the initial melt phase prevents hydrolysis of the acetic anhydride, ensuring that the acetylating agent is utilized efficiently for the target transformation. Furthermore, the use of sodium acetate avoids the introduction of tertiary amines, which can form difficult-to-remove salts or act as nucleophilic impurities themselves. The subsequent purification via recrystallization from solvents like chloroform or ethanol leverages the distinct solubility profiles of the acetylated products versus unreacted starting materials and di-acetylated byproducts. This precise control over the reaction environment allows for the production of reference standards with purity levels exceeding 99%, which is critical for HPLC method validation in pharmaceutical quality control laboratories. The robustness of this catalytic cycle ensures batch-to-batch consistency, a key requirement for regulatory submissions.
How to Synthesize 2-O-acetyl-5-isosorbide dinitrate Efficiently
The implementation of this synthesis route in a laboratory or pilot plant setting requires careful attention to thermal management and stoichiometry to maximize yield and purity. The process begins with the precise weighing of the isosorbide mononitrate isomer and the sodium acetate catalyst, typically in a molar ratio ranging from 1:0.02 to 1:0.10. The mixture is heated in an oil bath to approximately 105°C until a homogeneous melt is observed, indicating that the solid reactants have fully liquefied and the catalyst is dispersed. At this stage, acetic anhydride is added slowly to control the exotherm, followed by heating to reflux for a duration of 1 hour to ensure complete conversion. Detailed standardized operating procedures regarding temperature ramping rates and quenching protocols are essential for safety and reproducibility. For a comprehensive step-by-step guide including specific workup parameters and crystallization conditions, please refer to the technical protocol below.
- Mix isosorbide mononitrate isomer with sodium acetate catalyst and heat to 100-105°C to melt the reactants completely.
- Add acetic anhydride or acetic acid while stirring and maintain reflux conditions for 0.5 to 3 hours to complete the acetylation.
- Cool the reaction mixture, extract with ethyl acetate or chloroform, and purify via recrystallization to obtain high-purity target isomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this melt-acetylation technology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of expensive biocatalysts like lipase and toxic organic bases like DMAP removes significant cost drivers from the bill of materials. Additionally, the reduction in solvent usage during the reaction phase lowers both raw material costs and waste disposal fees, contributing to a leaner manufacturing budget. The simplified downstream processing, which avoids column chromatography in favor of crystallization, reduces labor hours and equipment occupancy time, thereby increasing overall plant throughput. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands for high-purity pharmaceutical intermediates without the volatility associated with complex enzymatic supply chains.
- Cost Reduction in Manufacturing: The shift from enzymatic or amine-catalyzed processes to a simple sodium acetate system drastically reduces the cost of goods sold (COGS). By removing the dependency on specialized enzymes that require cold chain logistics and have limited shelf lives, manufacturers can stabilize their input costs. Furthermore, the ability to use acetic acid or anhydride in near-stoichiometric amounts minimizes reagent waste. The avoidance of halogenated solvents during the reaction step, reserving them only for extraction, aligns with solvent recovery protocols that further enhance economic efficiency. This economic model supports competitive pricing for high-value impurity standards, making it feasible for pharmaceutical companies to maintain rigorous quality control programs without prohibitive expenses.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including isosorbide mononitrate, acetic anhydride, and sodium acetate, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague specialty reagent markets. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without compromising product quality, ensures high first-pass yields and consistent delivery schedules. For supply chain planners, this predictability is invaluable, allowing for accurate inventory forecasting and reducing the need for safety stock buffers. The scalability of the melt process means that production can be ramped up quickly from kilogram to tonne scales to support clinical trial material needs or commercial API launches.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner profile that simplifies regulatory compliance. The reduction in wastewater generation, particularly the absence of amine-contaminated aqueous streams, lowers the burden on effluent treatment plants. The use of sodium acetate, a benign salt, eliminates the need for heavy metal scavenging or complex neutralization steps required by other catalytic systems. This green chemistry advantage facilitates easier permitting for new production lines and aligns with the sustainability goals of major pharmaceutical customers. The process is inherently safer due to the controlled addition of reagents in the melt phase, reducing the risk of thermal runaway compared to large-volume solution reactions. These factors collectively enhance the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isosorbide derivatives. Understanding the nuances of this synthesis helps stakeholders make informed decisions about sourcing and quality specifications. The answers provided are derived directly from the technical disclosures and experimental data within the patent literature, ensuring accuracy and relevance for industry professionals.
Q: What are the primary advantages of the melt-acetylation method over enzymatic routes?
A: The melt-acetylation method described in patent CN110590799B eliminates the need for expensive lipase enzymes and complex pH control systems, significantly reducing reaction time from days to hours and simplifying downstream purification.
Q: How does this synthesis route improve impurity control for Isosorbide Mononitrate?
A: By providing a direct and selective acetylation pathway, this method generates specific isomeric impurities with high purity (>99%), which are essential reference standards for accurately quantifying impurities in the final drug substance.
Q: Is the sodium acetate catalyst recoverable in this process?
A: Sodium acetate acts as a mild base catalyst that remains in the aqueous phase during the extraction workup, allowing for easy separation from the organic product layer without requiring complex metal scavenging steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-O-acetyl-5-isosorbide dinitrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurities play in the development and quality assurance of cardiovascular medications. Our technical team has extensively analyzed the melt-acetylation route described in CN110590799B and possesses the expertise to optimize this chemistry for industrial-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-O-acetyl-5-isosorbide dinitrate or 5-O-acetyl-2-isosorbide dinitrate meets the highest pharmacopeial standards. We are committed to being a reliable pharmaceutical intermediates supplier that supports your regulatory filings with dependable data and material.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project requirements. Whether you need custom synthesis of rare isomers or large-scale supply of established intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes quality, sustainability, and cost-efficiency, empowering your R&D and manufacturing operations to succeed in a competitive global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
