Revolutionizing OLED Displays with High-Efficiency Blue Light Cuprous Halide Complexes
Revolutionizing OLED Displays with High-Efficiency Blue Light Cuprous Halide Complexes
The rapid evolution of organic light-emitting diode (OLED) technology has created an urgent demand for efficient, stable, and cost-effective electroluminescent materials, particularly in the blue spectrum which has historically been challenging to optimize. Patent CN109320530B introduces a groundbreaking class of four-coordinate mononuclear cuprous halide complexes, specifically designed to address these challenges through the innovative use of dimethylthiophene bisphosphine (dpmt) and triphenylphosphine ligands. This technology represents a significant leap forward for any reliable OLED material supplier seeking to diversify their portfolio beyond traditional noble metal emitters. The disclosed compounds, with the general formula [CuX(dpmt)(PPh3)], exhibit exceptional photophysical properties, including strong blue light emission at room temperature with maximum wavelengths ranging from 447 to 460 nm. Furthermore, the absolute quantum efficiency reaches impressive levels between 0.26 and 0.45, demonstrating that earth-abundant copper can effectively compete with expensive iridium and platinum counterparts in high-performance display applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of high-efficiency phosphorescent OLED materials has been dominated by third-row transition metals such as iridium and platinum. While these materials offer excellent performance, they suffer from severe economic and supply chain constraints due to the scarcity and exorbitant cost of the raw metals. Additionally, achieving deep blue emission with high color purity using these traditional complexes often requires complex molecular engineering that can compromise stability or increase synthetic difficulty. Many existing green-light cuprous complexes have been reported, but efficient blue-light thermally activated delayed fluorescence (TADF) cuprous complexes remain rare in the prior art. The reliance on scarce resources creates a bottleneck for cost reduction in electronic chemical manufacturing, forcing manufacturers to seek alternative chemistries that do not sacrifice performance for affordability. Furthermore, conventional synthesis routes for high-performance emitters often involve harsh conditions or multi-step processes that reduce overall yield and increase waste generation.
The Novel Approach
The invention detailed in CN109320530B overcomes these barriers by utilizing a rigid bisphosphine ligand derived from thiophene, a rich-electron heterocycle, combined with triphenylphosphine. This strategic ligand design effectively raises the LUMO energy level, resulting in the desired blue shift in emission wavelength without the need for expensive noble metals. The synthesis method is remarkably straightforward, operating under mild conditions that do not require specialized or expensive instrumentation, which is highly beneficial for industrialization. By replacing the benzene backbone found in previous iterations with a thiophene backbone, the inventors have successfully tuned the electronic properties to achieve emission in the critical 400-460 nm range. This approach not only lowers the raw material costs significantly but also simplifies the purification process, as evidenced by the high yields of 81% to 87% reported in the examples. This novel pathway offers a robust solution for producing high-purity OLED material at a fraction of the cost of traditional methods.
Mechanistic Insights into Cu-Based TADF Emission
The core of this technology lies in the unique coordination geometry and electronic structure of the [CuX(dpmt)(PPh3)] complex. The central copper atom adopts a highly distorted tetrahedral configuration, coordinated by three phosphorus atoms from the ligands and one halogen atom. This specific geometry is crucial for facilitating the thermally activated delayed fluorescence (TADF) mechanism. Detailed analysis reveals that the emission primarily originates from metal-to-ligand charge transfer (MLCT), halogen-to-ligand charge transfer (XLCT), and intra-ligand charge transitions. The introduction of the electron-donating thiophene group plays a pivotal role in modulating these energy levels. As shown in the structural analysis, the rigid framework provided by the dpmt ligand helps to minimize non-radiative decay pathways, thereby enhancing the quantum efficiency.
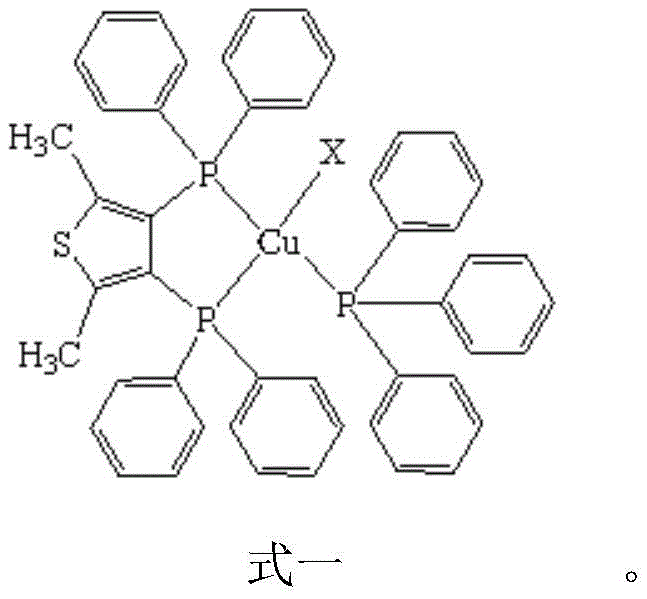
Further mechanistic studies using TDDFT calculations indicate that the HOMO orbitals are mainly distributed over the copper, halogen, and phosphorus atoms, while the LUMO orbitals are localized on the benzene and thiophene rings. This spatial separation of frontier molecular orbitals contributes to a small energy gap between the singlet (S1) and triplet (T1) excited states, calculated to be between 0.0646 and 0.1980 eV. Such a small ΔE(S1-T1) is the hallmark of TADF materials, allowing for the efficient up-conversion of triplet excitons to singlet states, which then radiatively decay to emit light. This mechanism effectively harvests both singlet and triplet excitons, theoretically enabling 100% internal quantum efficiency, a critical factor for the commercial viability of next-generation display technologies.
How to Synthesize High-Efficiency Blue Light Cuprous Complexes Efficiently
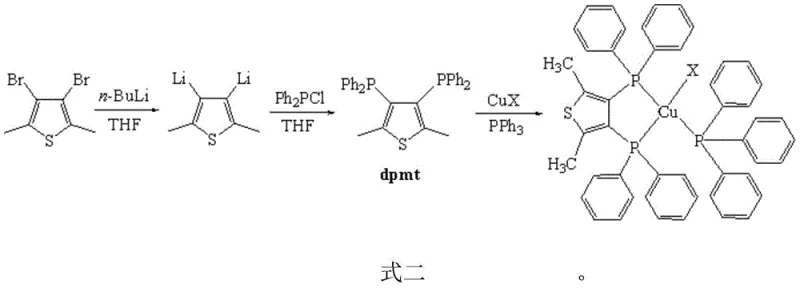
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating these advanced materials, making it accessible for R&D teams aiming to replicate or scale the process. The procedure begins with the preparation of the key dpmt ligand via a lithiation-phosphination sequence, followed by a simple coordination reaction with cuprous halides and triphenylphosphine. The entire process is designed to be operationally simple, avoiding the need for extreme pressures or temperatures that often complicate scale-up. The use of common solvents like tetrahydrofuran (THF) and dichloromethane ensures that the process can be easily integrated into existing chemical manufacturing infrastructure. For a detailed breakdown of the specific stoichiometric ratios, reaction times, and purification techniques required to achieve the reported high yields, please refer to the standardized synthesis guide below.
- Synthesize the dpmt ligand by reacting 2,5-dimethyl-3,4-dibromothiophene with n-butyllithium at -78°C in THF, followed by quenching with chlorodiphenylphosphine.
- Mix the purified dpmt ligand with cuprous halide (CuX where X=I, Br, or Cl) in dichloromethane under nitrogen atmosphere.
- Add triphenylphosphine (PPh3) to the mixture, stir overnight at room temperature, and purify via recrystallization to obtain the final complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to copper-based emitters offers substantial strategic benefits beyond mere technical performance. The primary advantage is the drastic reduction in raw material volatility; unlike iridium, which is subject to geopolitical supply risks and price spikes, copper is an abundant base metal with a stable and transparent global market. This shift fundamentally alters the cost structure of OLED material production, enabling significant cost savings that can be passed down the value chain or reinvested into R&D. Moreover, the simplified synthesis route reduces the number of unit operations and energy consumption, further driving down the cost of goods sold (COGS). The high yields reported (81-87%) mean less waste and higher throughput per batch, optimizing capital utilization in manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts like iridium or platinum removes a major cost driver from the bill of materials. Since copper salts are inexpensive and widely available, the direct material cost is significantly lowered. Additionally, the synthesis does not require expensive instrumentation or harsh reaction conditions, which reduces capital expenditure on specialized reactors and lowers utility costs associated with heating or cooling. The high purity of the final product achieved through simple recrystallization minimizes the need for complex chromatographic purification steps, streamlining the production workflow and reducing solvent consumption.
- Enhanced Supply Chain Reliability: Sourcing copper and phosphine ligands is far more reliable than sourcing specialty noble metal precursors, which often have long lead times and limited suppliers. The reagents used in this process, such as n-butyllithium, chlorodiphenylphosphine, and triphenylphosphine, are commodity chemicals available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the stability of the intermediates and the final complexes ensures that inventory can be managed effectively without rapid degradation, supporting a more resilient just-in-time manufacturing model.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram or ton-scale commercial production without fundamental changes to the chemistry. The use of standard organic solvents allows for established recovery and recycling protocols, aligning with increasingly stringent environmental regulations. The absence of toxic heavy metals like iridium simplifies waste disposal and regulatory compliance, reducing the administrative burden and liability associated with hazardous material handling. This environmental friendliness enhances the corporate sustainability profile, a key metric for modern supply chain evaluations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating these materials into their product lines, we have compiled answers to common questions regarding the performance and handling of these cuprous complexes. These insights are derived directly from the experimental data and characterization results presented in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding the thermal stability and emission characteristics is crucial for device engineers designing OLED stacks.
Q: What are the key advantages of these cuprous complexes over traditional iridium-based emitters?
A: Unlike expensive third-row transition metals like iridium and platinum, copper is abundant and significantly cheaper. These complexes offer high quantum efficiency (0.26-0.45) and strong blue emission (447-460nm) without the prohibitive cost associated with noble metals.
Q: How does the dpmt ligand contribute to the blue-shifted emission?
A: The introduction of the electron-rich thiophene ring in the dpmt ligand raises the LUMO energy level compared to benzene-based analogs. This electronic modification results in a significant blue shift in the maximum emission wavelength, making it ideal for full-color displays.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis involves standard organic reactions like lithiation and coordination in common solvents like THF and dichloromethane. The process operates at mild temperatures (room temperature for complexation) and achieves high yields (81-87%), indicating excellent potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cuprous Halide Complex Supplier
As the demand for high-performance blue emitters continues to surge, partnering with an experienced CDMO is essential for translating patented laboratory success into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of cuprous halide complex meets the exacting standards required for OLED fabrication. We understand the critical nature of electronic materials and operate with the discipline necessary to maintain supply continuity for global display manufacturers.
We invite you to engage with our technical procurement team to discuss how this technology can be adapted to your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to these copper-based emitters. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and profitability in your OLED production lines.
