Revolutionizing Dabigatran Intermediate Production via Enzymatic Catalysis and Commercial Scale-Up
The pharmaceutical landscape for anticoagulant therapies has been significantly transformed by the introduction of direct thrombin inhibitors, with Dabigatran Etexilate standing as a cornerstone molecule in modern cardiovascular treatment. Patent CN103710406A discloses a groundbreaking method for preparing the main intermediate of Dabigatran Etexilate through a sophisticated enzymatic reaction, marking a pivotal shift from traditional chemical synthesis to biocatalytic processes. This innovation addresses critical bottlenecks in the manufacturing of high-purity pharmaceutical intermediates by utilizing immobilized enzyme Novozym 435 to facilitate amide condensation under remarkably mild conditions. The strategic implementation of this technology not only enhances the purity profile of the final product but also streamlines the production workflow, making it an attractive proposition for reliable pharmaceutical intermediate supplier networks seeking to optimize their portfolios. By leveraging the specificity of biocatalysts, this method circumvents the formation of complex impurity profiles often associated with harsh chemical reagents, thereby ensuring a robust supply of critical medication precursors.
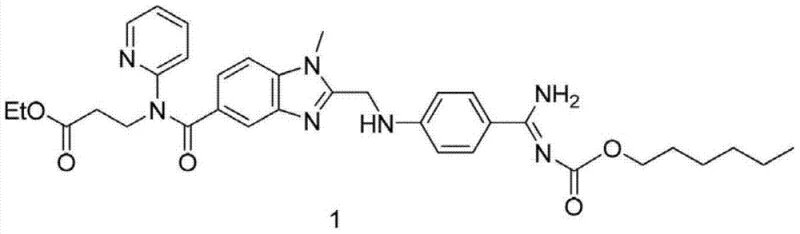
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key Dabigatran intermediates has relied heavily on chemical coupling agents such as 1,1'-Carbonyldiimidazole (CDI) or EDCI to drive amide bond formation, a process fraught with significant operational and economic challenges. As illustrated in prior art patents like WO2012152855 and WO2010045900, these conventional routes necessitate strict anhydrous conditions and low-temperature environments to prevent the hydrolysis of sensitive coupling reagents, which drastically increases energy consumption and operational complexity. Furthermore, the use of CDI introduces substantial cost burdens due to the high price of the reagent itself and the generation of stoichiometric amounts of imidazole by-products that require rigorous and costly purification steps to remove. The sensitivity of these chemical catalysts to moisture often leads to batch-to-batch variability and reduced yields, creating instability in the supply chain for high-purity pharmaceutical intermediates. Additionally, the disposal of chemical waste generated from these coupling reactions poses environmental compliance challenges, further complicating the commercial viability of these legacy manufacturing processes.
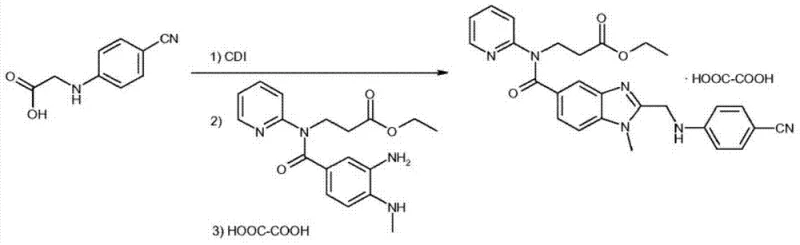
The Novel Approach
In stark contrast to these cumbersome chemical methodologies, the novel approach detailed in CN103710406A employs immobilized lipase Novozym 435 as a robust and highly selective biocatalyst to achieve the same transformation with superior efficiency. This enzymatic strategy operates effectively at room temperature or slightly elevated temperatures ranging from 10°C to 60°C, eliminating the need for cryogenic cooling and significantly reducing the thermal energy footprint of the manufacturing process. The immobilized nature of the enzyme allows for easy separation from the reaction mixture via simple filtration, enabling the potential for catalyst reuse and drastically simplifying the downstream processing workflow. By replacing expensive and hazardous chemical coupling agents with a biocatalytic system, this method achieves cost reduction in API manufacturing through both raw material savings and simplified waste management protocols. The result is a streamlined synthesis pathway that delivers high-purity products with minimal impurity generation, aligning perfectly with the stringent quality standards required for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Novozym 435-Catalyzed Amidation and Cyclization
The core of this innovative methodology lies in the deployment of immobilized lipase Novozym 435, which serves as a highly selective biocatalyst to facilitate the amide condensation step between 3-[4-methylamino-3-amino-N-(2-pyridyl)-benzamido]-ethyl acrylate and 2-(4-cyano aniline) acetic acid. The enzyme's active site promotes the nucleophilic attack of the amine group on the carboxylic acid functionality with exceptional stereo- and chemoselectivity, ensuring that the reaction proceeds without affecting other sensitive functional groups present in the molecular structure. This specificity is crucial for maintaining the integrity of the pyridine and benzimidazole moieties, which are essential for the biological activity of the final anticoagulant drug. The reaction mechanism benefits from the unique microenvironment provided by the immobilization support, which stabilizes the enzyme structure and enhances its tolerance to organic solvents, allowing the reaction to proceed in media such as methylene chloride or tetrahydrofuran. Furthermore, the patent highlights the synergistic effect of adding ionic liquids like [bmim]PF6 or [bmim]BF4 to the reaction mixture, which improves the solubility of the polar reactants and enhances mass transfer rates, leading to accelerated reaction kinetics and higher overall conversion yields.
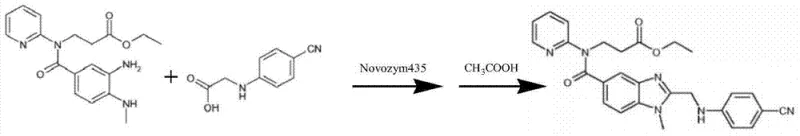
Following the enzymatic amidation, the process incorporates a critical cyclization step mediated by glacial acetic acid under reflux conditions to form the benzimidazole ring system, a transformation that is pivotal for the pharmacological efficacy of the intermediate. The control of impurities during this phase is managed through the precise regulation of temperature and reaction time, typically maintaining the reflux at 110°C to 120°C for approximately 2 hours to ensure complete conversion without degradation. The enzymatic precursor step significantly reduces the burden on this cyclization phase by providing a cleaner starting material with fewer side-reaction by-products compared to chemically catalyzed routes. This two-stage process, combining biocatalytic selectivity with thermal cyclization, creates a robust impurity control mechanism that consistently delivers products with purity levels exceeding 95% as demonstrated in the patent examples. The ability to achieve such high purity without extensive chromatographic purification is a major advantage for reducing lead time for high-purity pharmaceutical intermediates, ensuring that the manufacturing process remains both economically and technically viable for large-scale production.
How to Synthesize Dabigatran Intermediate Efficiently
The synthesis of this critical anticoagulant precursor is achieved through a meticulously optimized sequence that balances reaction efficiency with operational simplicity, making it highly suitable for industrial adoption. The process begins with the dissolution of the amine and acid reactants in a solvent system that may include ionic liquids to enhance solubility, followed by the addition of the immobilized enzyme to initiate the amidation. Detailed standard operating procedures regarding specific stoichiometric ratios, solvent volumes, and filtration techniques are essential for replicating the high yields reported in the patent data. The following guide outlines the standardized synthesis steps derived directly from the technical disclosures to ensure reproducibility and quality control.
- Dissolve 3-[4-methylamino-3-amino-N-(2-pyridyl)-benzamido]-ethyl acrylate and 2-(4-cyano aniline) acetic acid in a mixed solvent of methylene chloride and ionic liquid, then add immobilized enzyme Novozym 435.
- Stir the reaction mixture at room temperature for 10 to 24 hours, then filter to remove the immobilized enzyme and evaporate the organic solvent to obtain the oily intermediate.
- Add glacial acetic acid to the oily substance and reflux at 110°C for 2 hours to complete cyclization, followed by extraction and drying to yield the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route presents a compelling value proposition centered around stability, cost efficiency, and operational flexibility. The elimination of moisture-sensitive coupling reagents like CDI removes a significant source of supply chain volatility, as these chemicals often require specialized storage and handling conditions that increase logistical overhead. By shifting to a robust enzymatic catalyst that can be stored under standard conditions and potentially reused across multiple batches, manufacturers can achieve substantial cost savings in raw material procurement and inventory management. Furthermore, the mild reaction conditions reduce the dependency on specialized cryogenic equipment and high-energy heating systems, leading to a lower total cost of ownership for the production facility. This transition not only optimizes the direct manufacturing costs but also enhances the overall resilience of the supply chain by minimizing the risk of batch failures due to reagent degradation or environmental fluctuations.
- Cost Reduction in Manufacturing: The replacement of expensive stoichiometric coupling agents with a catalytic amount of immobilized enzyme fundamentally alters the cost structure of the synthesis, driving down the variable cost per kilogram of the intermediate. Since the enzyme acts as a true catalyst and can be recovered via filtration, the consumption of high-value reagents is drastically minimized compared to traditional chemical methods that generate stoichiometric waste. Additionally, the simplified work-up procedure, which avoids complex quenching and extraction steps associated with chemical coupling by-products, reduces the consumption of solvents and utilities. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediate, allowing buyers to secure high-purity pharmaceutical intermediates at a more sustainable price point without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the Novozym 435 catalyst ensures consistent reaction performance across different batches, mitigating the risk of supply disruptions caused by failed reactions or off-spec material. Unlike chemical catalysts that may degrade rapidly upon exposure to ambient moisture, the immobilized enzyme maintains its activity over extended periods, providing a stable and predictable production schedule. This reliability is critical for maintaining continuous supply to downstream API manufacturers, ensuring that production timelines are met without unexpected delays. The ability to source stable and long-lasting catalysts also simplifies vendor management, reducing the frequency of reordering and the administrative burden associated with managing hazardous chemical inventories.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, as the filtration-based separation of the catalyst is easily adaptable to large-scale industrial reactors. The reduction in hazardous chemical waste and the use of milder solvents align with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for manufacturing sites. The lower energy requirements for temperature control further contribute to a reduced carbon footprint, supporting corporate sustainability goals. This combination of scalability and environmental stewardship makes the technology a future-proof investment for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis technology, providing clarity on its operational feasibility and strategic benefits. These insights are derived from the specific technical parameters and advantageous effects disclosed in the patent documentation, offering a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the integration of this route into existing manufacturing frameworks.
Q: What are the primary advantages of using Novozym 435 over traditional chemical coupling agents like CDI?
A: The use of Novozym 435 eliminates the need for moisture-sensitive and expensive coupling reagents like CDI, significantly reducing raw material costs and simplifying the reaction conditions to ambient temperatures while maintaining high selectivity.
Q: How does the addition of ionic liquids impact the reaction yield and scalability?
A: Ionic liquids such as [bmim]PF6 improve the solubility of reactants and enhance mass transfer within the reaction mixture, leading to higher yields and shorter reaction times, which is critical for efficient commercial scale-up.
Q: Is this enzymatic process suitable for large-scale industrial production of anticoagulant intermediates?
A: Yes, the process is highly suitable for industrial amplification because the immobilized enzyme can be easily removed by filtration and potentially reused, and the mild reaction conditions reduce energy consumption and safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market, and we are fully equipped to leverage this enzymatic pathway for your production needs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Dabigatran intermediate meets the highest international standards for safety and efficacy. Our commitment to technological innovation allows us to offer solutions that not only meet current regulatory requirements but also anticipate future industry shifts towards greener and more efficient manufacturing practices.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs through the implementation of this superior synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with us, you gain access to a reliable supply of high-quality intermediates backed by deep technical expertise and a commitment to long-term partnership.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
