Advanced Catalytic Transfer Hydrogenation for High-Purity Imatinib Intermediates: A Commercial Scale-Up Perspective
Introduction to Next-Generation Imatinib Intermediate Synthesis
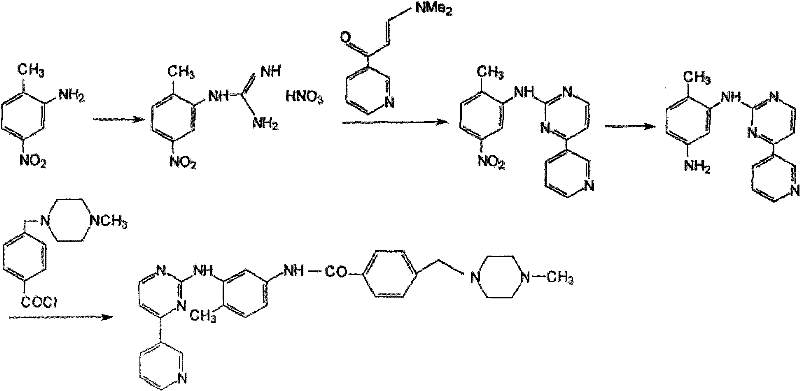
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for producing critical oncology intermediates. Patent CN102199146A introduces a transformative preparation method for N-(5-amino-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine, a pivotal building block in the synthesis of the blockbuster antitumor drug Imatinib Mesylate. This innovation addresses long-standing challenges in the reduction of nitro-groups within complex heterocyclic systems, moving away from hazardous high-pressure hydrogenation and toxic hydrazine protocols. By leveraging catalytic transfer hydrogenation with ammonium formate, the disclosed technology achieves yields exceeding 90% and HPLC purities greater than 99.5%, setting a new benchmark for quality in pharmaceutical intermediates manufacturing. For global supply chains, this represents a shift towards safer, more predictable production capabilities that align with modern Green Chemistry principles while maintaining rigorous quality standards required for GMP environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of N-(5-nitro-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine has been plagued by significant technical and operational hurdles. Traditional methods utilizing palladium on carbon with high-pressure hydrogen gas often suffer from poor selectivity, leading to the formation of difficult-to-remove over-reduction byproducts, specifically identified as Formula 1 and Formula 2 in the prior art. These impurities, often generated in amounts exceeding 1%, necessitate complex and yield-eroding purification steps such as repeated recrystallization or column chromatography. Furthermore, alternative routes employing tin dichloride generate massive quantities of toxic tin-containing waste sludge, creating severe environmental compliance burdens and escalating waste disposal costs. The use of hydrazine hydrate, another common reductant, introduces extreme safety risks due to its toxicity and potential explosiveness, requiring specialized containment infrastructure that increases capital expenditure for manufacturing facilities.
The Novel Approach
The methodology outlined in CN102199146A circumvents these pitfalls through a sophisticated application of transfer hydrogenation. By substituting gaseous hydrogen or toxic hydrazine with ammonium formate in an ester solvent system, the reaction proceeds under atmospheric pressure and moderate temperatures (60-85°C). This mild environment kinetically favors the selective reduction of the nitro group to the amine while effectively suppressing the formation of the problematic over-reduced species shown in 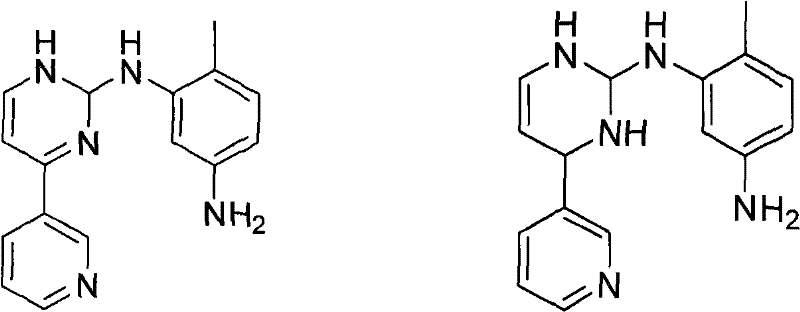 . The inclusion of anhydrous sodium sulfate further optimizes the reaction kinetics, shortening reaction times and enhancing conversion efficiency. This approach not only simplifies the downstream processing by eliminating heavy metal salt filtration but also ensures a consistent, high-purity product profile that is critical for downstream API synthesis, thereby offering a reliable pharmaceutical intermediates supplier solution for large-scale production.
. The inclusion of anhydrous sodium sulfate further optimizes the reaction kinetics, shortening reaction times and enhancing conversion efficiency. This approach not only simplifies the downstream processing by eliminating heavy metal salt filtration but also ensures a consistent, high-purity product profile that is critical for downstream API synthesis, thereby offering a reliable pharmaceutical intermediates supplier solution for large-scale production.
Mechanistic Insights into Pd-C Catalyzed Transfer Hydrogenation
The core of this technological advancement lies in the mechanistic elegance of using ammonium formate as a hydrogen donor in the presence of a heterogeneous palladium catalyst. Unlike direct hydrogenation where H2 gas adsorption can be aggressive and non-selective, the decomposition of ammonium formate on the Pd surface generates atomic hydrogen in situ at a controlled rate. This "slow-release" mechanism prevents the local excess of reactive hydrogen species that typically drives the over-reduction of the pyrimidine or pyridine rings, or the formation of hydroxylamine intermediates that condense into azo-compounds. The ester solvent, particularly ethyl acetate, plays a dual role as both a reaction medium and a stabilizer, ensuring the substrate remains in solution while facilitating the interaction between the solid catalyst and the liquid phase reagents. This precise control over the hydrogen flux is what allows the process to achieve such exceptional chemoselectivity, preserving the integrity of the sensitive heterocyclic core essential for Imatinib's biological activity.
From an impurity control perspective, the addition of anhydrous sodium sulfate acts as a crucial process parameter that modulates the reaction environment. While the exact mechanistic role may involve water scavenging to drive the equilibrium of formate decomposition forward, empirically it correlates with a drastic reduction in reaction time from roughly 6 hours down to 2-3 hours. Shorter residence times at elevated temperatures inherently reduce the opportunity for thermal degradation or secondary side reactions. Furthermore, the ability to recycle the 10% Pd-C catalyst mechanically up to 10 times suggests that the catalyst surface remains active and resistant to poisoning by the nitrogen-rich substrate or products. This stability is vital for maintaining consistent batch-to-batch quality, ensuring that the high-purity pharmaceutical intermediates produced meet the stringent specifications required by regulatory bodies for oncology drug substances.
How to Synthesize N-(5-Amino-2-Methylphenyl)-4-(3-Pyridyl)-2-Pyrimidinamine Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the transfer hydrogenation pathway. The process begins by suspending the nitro-substrate in an ester solvent, followed by the addition of the palladium catalyst and the hydrogen source. The reaction is then heated to reflux, allowing the ammonium formate to decompose and effect the reduction. Post-reaction workup is notably straightforward, involving simple filtration to recover the catalyst and an aqueous wash to remove residual salts, followed by crystallization.
- Charge the reactor with N-(5-nitro-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine substrate, 10% Pd-C catalyst (2-10% w/w), and anhydrous sodium sulfate in an ester solvent like ethyl acetate.
- Add ammonium formate (3-4.5 molar equivalents) as the hydrogen donor and heat the mixture to reflux (60-85°C) for 2-6 hours under stirring.
- Filter off the reusable catalyst, wash the filtrate with water, concentrate, and recrystallize the residue from ethyl acetate/n-hexane to obtain the high-purity amino intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages beyond mere technical feasibility. The elimination of high-pressure hydrogenation equipment significantly lowers the barrier to entry for contract manufacturing organizations (CMOs), expanding the pool of qualified vendors and enhancing supply security. By removing the need for hazardous reagents like hydrazine hydrate and toxic heavy metals like tin, the process drastically reduces the costs associated with environmental health and safety (EHS) compliance, waste treatment, and regulatory reporting. These operational efficiencies contribute to substantial cost savings in pharmaceutical intermediates manufacturing, allowing for more competitive pricing models without compromising on quality. Additionally, the robustness of the reaction conditions ensures high reproducibility, minimizing the risk of batch failures that can disrupt production schedules and delay time-to-market for critical cancer therapies.
- Cost Reduction in Manufacturing: The economic impact of this method is driven primarily by the recyclability of the palladium catalyst and the simplicity of the workup. Since the 10% Pd-C can be reused multiple times, the consumption of this precious metal is minimized, directly lowering the bill of materials. Furthermore, the avoidance of complex purification steps like column chromatography or extensive recrystallization cycles reduces solvent consumption and labor hours. The qualitative shift from a multi-step, waste-intensive process to a streamlined one-pot reduction significantly optimizes the overall cost structure, making the commercial scale-up of complex pharmaceutical intermediates more economically viable.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized reagents or equipment. This method utilizes commodity chemicals like ammonium formate and ethyl acetate, which are readily available in the global market, reducing the risk of raw material shortages. The atmospheric pressure operation means that production is not bottlenecked by the availability of high-pressure reactors, allowing for greater flexibility in manufacturing scheduling. This adaptability ensures that partners can respond more agilely to fluctuations in demand for Imatinib, securing a steady flow of high-purity pharmaceutical intermediates to the API production lines.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and waste issues, but this technology inherently mitigates those risks. The absence of explosive hydrazine and the generation of benign gaseous byproducts (nitrogen, carbon dioxide) simplify the scale-up engineering requirements. Waste streams are significantly cleaner, lacking the heavy metal contamination associated with tin reductions, which facilitates easier disposal and lowers the environmental footprint. This alignment with green chemistry principles not only future-proofs the supply chain against tightening environmental regulations but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its implementation. The following questions address common concerns regarding impurity management, catalyst lifecycle, and safety protocols inherent to this specific patent technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the process capabilities.
Q: How does the Pd-C/Ammonium Formate method improve impurity profiles compared to traditional hydrogenation?
A: Traditional high-pressure hydrogenation often leads to over-reduction byproducts (Formulas 1 and 2). The transfer hydrogenation method described in CN102199146A operates under milder conditions, suppressing these side reactions and keeping total impurities below 0.5%, often achieving >99.8% purity without complex chromatography.
Q: Is the palladium catalyst recoverable in this process?
A: Yes, a significant advantage of this protocol is the mechanical recyclability of the 10% Pd-C catalyst. The patent data indicates the catalyst can be recovered and reused up to 10 times without significant loss of activity, drastically reducing heavy metal consumption and waste disposal costs.
Q: What are the safety advantages of using ammonium formate over hydrazine hydrate?
A: Hydrazine hydrate is highly toxic and requires stringent safety measures and waste treatment. Ammonium formate is a solid, easier to handle, and decomposes into benign gases (CO2, NH3, H2), eliminating the need for high-pressure equipment and reducing the environmental footprint of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(5-Amino-2-Methylphenyl)-4-(3-Pyridyl)-2-Pyrimidinamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN102199146A are faithfully translated into your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities at ppm levels, guaranteeing that every batch of N-(5-amino-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine meets the exacting standards required for oncology drug development. Our commitment to quality assurance ensures that the cost reduction in pharmaceutical intermediates manufacturing never comes at the expense of product integrity.
We invite you to leverage our technical capabilities to optimize your Imatinib supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our advanced catalytic transfer hydrogenation platform can enhance your project's efficiency and profitability. Let us be your trusted partner in delivering high-quality solutions for the global pharmaceutical market.
