Scalable Metal-Free Synthesis of Phosphonomethylene Five-Membered Rings for Advanced Pharmaceutical Intermediates
Introduction to Next-Generation Organophosphorus Synthesis
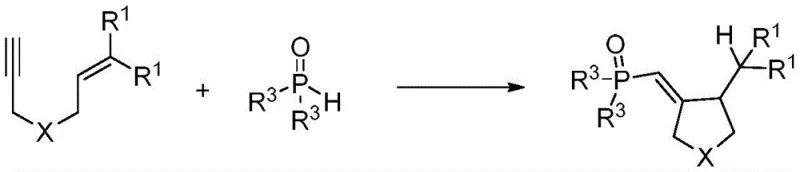
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the production of complex heterocyclic scaffolds essential for modern drug discovery. Patent CN111072720B introduces a groundbreaking approach for the synthesis of phosphonomethylene-substituted five-membered cyclic compounds, a structural motif frequently encountered in bioactive molecules exhibiting antitumor, antibacterial, and anti-inflammatory properties. This innovation departs radically from conventional reliance on transition metal catalysis, instead leveraging the power of visible-light photocatalysis mediated by inexpensive organic dyes. By utilizing 1,6-enynes and diarylphosphine oxides as readily available starting materials, this technology enables the construction of valuable phosphono-pyrrolidine derivatives under exceptionally mild conditions. The significance of this development extends beyond academic interest; it offers a tangible solution to the persistent challenges of metal contamination and harsh reaction environments that plague current manufacturing protocols. For pharmaceutical developers and chemical procurement specialists, this patent represents a critical opportunity to access high-purity intermediates through a streamlined, environmentally benign process that aligns perfectly with modern green chemistry principles and regulatory demands for cleaner production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of phosphorus-containing heterocycles has been dominated by transition metal-catalyzed pathways involving elements such as nickel, copper, and silver. While effective in forming carbon-phosphorus bonds, these traditional methods suffer from inherent drawbacks that complicate their industrial application. The primary concern is the inevitable introduction of heavy metal residues into the reaction mixture, which necessitates rigorous and costly purification steps to meet the stringent impurity profiles required for Active Pharmaceutical Ingredients (APIs). Furthermore, these processes often demand harsh reaction conditions, including elevated temperatures and the use of strong oxidants or complex additives, which not only increase energy consumption but also pose significant safety risks during scale-up. The reliance on precious or toxic metals also introduces supply chain volatility, as the availability and price of these catalysts can fluctuate wildly. Additionally, the generation of metal-laden waste streams creates substantial environmental liabilities, forcing manufacturers to invest heavily in waste treatment infrastructure to comply with increasingly strict environmental regulations. These cumulative factors result in a manufacturing process that is economically inefficient, operationally complex, and environmentally unsustainable.
The Novel Approach
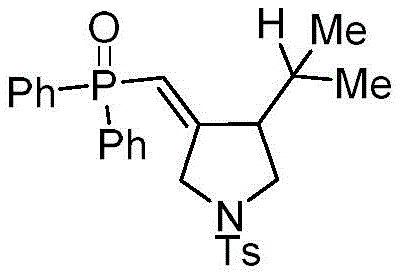
In stark contrast to the limitations of metal catalysis, the methodology disclosed in CN111072720B utilizes a metal-free photocatalytic system driven by visible light. This innovative route employs organic dyes, specifically Eosin Y, as potent photosensitizers to initiate a radical cascade reaction between 1,6-enynes and diarylphosphine oxides. The reaction proceeds smoothly at room temperature under nitrogen protection, eliminating the need for external heating or hazardous oxidants. By replacing transition metals with an organic dye, the process inherently avoids heavy metal contamination, drastically simplifying the downstream workup and purification stages. The use of common solvents like tetrahydrofuran (THF) and household-grade fluorescent lamps further underscores the practicality and accessibility of this method. This approach not only enhances the safety profile of the synthesis but also significantly reduces the operational complexity, allowing for a more straightforward translation from laboratory bench to commercial production. The ability to achieve high yields, such as the reported 83% in specific embodiments, without the baggage of metal toxicity marks a substantial advancement in the field of organophosphorus chemistry.
Mechanistic Insights into Visible-Light Mediated Radical Cyclization
The core of this synthetic breakthrough lies in the efficient generation of phosphorus-centered radicals via a photoredox mechanism. Upon irradiation with visible light, the Eosin Y photocatalyst absorbs photons and transitions to an excited state, possessing sufficient redox potential to interact with the diarylphosphine oxide substrate. This interaction facilitates the homolytic cleavage of the P-H bond, generating a reactive phosphorus radical species without the need for stoichiometric oxidants or metal mediators. This phosphorus radical then undergoes a highly selective intermolecular addition to the alkene moiety of the 1,6-enyne substrate, initiating a cascade sequence. The resulting carbon-centered radical subsequently engages in an intramolecular cyclization with the alkyne group, closing the five-membered ring and forming the stable phosphonomethylene-substituted pyrrolidine skeleton. This radical pathway is remarkably tolerant of various functional groups, allowing for the synthesis of diverse derivatives simply by modifying the substituents on the enyne or the phosphine oxide. The mechanistic elegance ensures that side reactions are minimized, leading to clean reaction profiles and high atom economy. Understanding this mechanism is crucial for process chemists, as it highlights the robustness of the reaction and its potential for broad substrate scope, enabling the rapid generation of chemical libraries for drug screening programs.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity landscape of the final product. In traditional metal-catalyzed reactions, metal-ligand complexes and metal-induced side products often co-elute with the desired compound, requiring specialized scavengers or chromatography to remove. In this photocatalytic system, the primary byproducts are derived from the organic dye or unreacted starting materials, which are generally easier to separate using standard silica gel chromatography. The mild reaction conditions also prevent thermal degradation of sensitive functional groups, preserving the integrity of complex molecular architectures. This results in a superior purity profile right out of the reactor, reducing the burden on quality control laboratories. For R&D directors, this means faster iteration cycles during process development, as less time is spent troubleshooting metal removal strategies. The predictability of the radical mechanism allows for precise tuning of reaction parameters, such as light intensity and catalyst loading, to further optimize the impurity profile and maximize the yield of the target phosphono-heterocycle.
How to Synthesize Phosphonomethylene Five-Membered Rings Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure reproducibility and optimal yield. The process begins with the precise weighing of the 1,6-enyne substrate and the diarylphosphine oxide reagent, maintaining a molar ratio that favors the phosphine oxide to drive the reaction to completion. The choice of solvent is critical, with tetrahydrofuran (THF) demonstrating superior performance compared to other polar and non-polar solvents. The addition of the Eosin Y photocatalyst must be carefully controlled, with 5 mol% identified as the sweet spot for balancing reaction rate and cost efficiency. The reaction vessel should be purged with nitrogen to create an inert atmosphere, preventing the quenching of radical intermediates by oxygen. Once the mixture is prepared, it is subjected to irradiation from a standard 40W fluorescent lamp, a light source that is both energy-efficient and widely available. Monitoring the reaction progress via thin-layer chromatography (TLC) allows operators to determine the exact endpoint, typically around 24 hours, ensuring maximum conversion before workup. The detailed standardized synthesis steps for this procedure are outlined below.
- Combine 1,6-enyne substrate and diarylphosphine oxide (3.0 equivalents) in tetrahydrofuran (THF) solvent.
- Add Eosin Y organic dye photocatalyst (5 mol%) to the reaction mixture under nitrogen protection.
- Irradiate the mixture with a 40W visible light source at room temperature for approximately 24 hours, followed by column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic technology offers compelling strategic advantages that extend far beyond simple chemical transformation. The elimination of transition metal catalysts represents a direct and significant reduction in raw material costs, as expensive salts of silver, palladium, or copper are no longer required. This shift also removes the logistical burden associated with sourcing and storing hazardous metal reagents, streamlining inventory management. Furthermore, the simplified purification process translates into reduced consumption of chromatography media and solvents during the isolation phase, driving down the overall cost of goods sold (COGS). The use of ambient temperature conditions significantly lowers energy expenditures related to heating and cooling, contributing to a smaller carbon footprint and lower utility bills. From a supply chain resilience perspective, the reliance on commodity chemicals like organic dyes and common solvents mitigates the risk of supply disruptions often associated with specialized catalytic systems. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the incidence of failed batches and the associated financial losses. Ultimately, this technology empowers organizations to build a more agile, cost-effective, and sustainable supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts eliminates a major cost driver in traditional synthesis, while the simplified workup reduces labor and material expenses associated with metal scavenging and extensive purification. The use of low-cost organic dyes and standard lighting equipment further minimizes capital expenditure and operational overhead, leading to substantial long-term savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials such as 1,6-enynes and diarylphosphine oxides, the process reduces dependency on niche suppliers of specialized reagents. The stability of the reaction conditions ensures that production schedules are not disrupted by sensitive parameter fluctuations, guaranteeing consistent delivery timelines for downstream customers and strengthening overall supply chain continuity.
- Scalability and Environmental Compliance: The mild, metal-free nature of this synthesis aligns perfectly with global environmental regulations, minimizing the generation of hazardous heavy metal waste. This compliance reduces the costs and complexities associated with waste disposal and environmental permitting. Additionally, the simplicity of the setup facilitates easy scale-up from gram to kilogram quantities, allowing for rapid response to market demand without the need for complex reactor engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production workflows. The insights provided here cover aspects ranging from catalyst selection to yield optimization, offering a comprehensive overview of the process capabilities.
Q: What are the primary advantages of this photocatalytic method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metals like nickel, copper, or silver, thereby removing the risk of heavy metal residues in the final API intermediate and simplifying downstream purification processes significantly.
Q: What represents the optimal reaction conditions for maximizing yield in this synthesis?
A: Experimental data indicates that using Tetrahydrofuran (THF) as the solvent, Eosin Y as the photocatalyst at 5 mol% loading, and a 40W fluorescent lamp light source provides the highest yields, reaching up to 83-94% depending on the substrate.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates at room temperature without high-pressure equipment or harsh oxidants, making it inherently safer and more adaptable for scale-up compared to traditional high-temperature metal-catalyzed processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphonomethylene Five-Membered Ring Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis described in CN111072720B for the production of high-value organophosphorus intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry approach are realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phosphonomethylene-substituted cyclic compounds meets the highest quality standards required by the global pharmaceutical industry. Our commitment to excellence ensures that our clients receive materials that are not only chemically pure but also fully compliant with international regulatory guidelines.
We invite procurement directors and R&D leaders to collaborate with us to optimize their supply chains using this advanced synthetic route. By leveraging our capabilities, you can achieve Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us help you navigate the transition to more sustainable and cost-effective manufacturing solutions, securing your position in the competitive landscape of fine chemical production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
