Scalable Electro-Synthesis of Phosphorylated Dihydroisoquinolone Derivatives for Advanced Pharma Applications
Scalable Electro-Synthesis of Phosphorylated Dihydroisoquinolone Derivatives for Advanced Pharma Applications
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways to access complex heterocyclic scaffolds essential for drug discovery. Patent CN116041394A discloses a groundbreaking methodology for the preparation of phosphorylated dihydroisoquinolone derivatives, a class of compounds known for their significant biological activities including anti-tumor and anti-inflammatory properties. This technology represents a paradigm shift from traditional transition-metal catalysis to sustainable electro-organic synthesis. By leveraging renewable electrical energy to drive radical cascade reactions, this method avoids the use of stoichiometric chemical oxidants and expensive noble metal catalysts. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: a cleaner, faster, and potentially more cost-effective route to high-value pharmaceutical intermediates. The ability to synthesize these structures under mild conditions with high atom economy addresses critical pain points in modern API manufacturing, particularly regarding impurity profiles and heavy metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydroisoquinolone frameworks has relied heavily on transition-metal catalysis, often involving precious metals such as rhodium or silver. For instance, prior art describes rhodium(III)-catalyzed C-H activation or silver-catalyzed radical cascades to achieve similar transformations. While effective, these conventional approaches suffer from inherent economic and environmental drawbacks. The reliance on noble metals introduces significant raw material costs and necessitates rigorous downstream purification to meet stringent regulatory limits on residual metals in drug substances. Furthermore, these reactions often require harsh chemical oxidants or prolonged reaction times, sometimes exceeding 18 hours, which limits throughput and increases energy consumption. The generation of metal-containing waste streams also poses a burden on environmental compliance and waste treatment infrastructure, complicating the path to commercialization for process chemists aiming for green chemistry metrics.
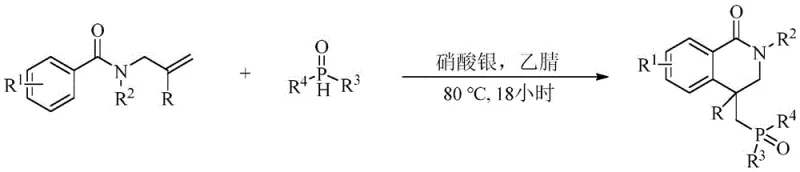
The Novel Approach
In stark contrast, the disclosed electrochemical methodology offers a streamlined and sustainable alternative. The core innovation lies in the use of constant current electrolysis to generate reactive radical species in situ, facilitating a cascade addition-cyclization sequence without external oxidants. As illustrated in the general reaction scheme, the process utilizes a simple undivided cell setup with a graphite anode and a platinum cathode. This setup allows for the precise control of oxidation potential, minimizing side reactions and over-oxidation. The reaction proceeds efficiently in dimethyl sulfoxide (DMSO) using tetrabutylammonium tetrafluoroborate as a supporting electrolyte. By replacing chemical oxidants with electrons, the process not only reduces the E-factor (environmental factor) but also simplifies the workup procedure. The mild conditions (80°C) and relatively short reaction times (1-6 hours) demonstrate superior efficiency compared to thermal counterparts, making it an attractive candidate for process intensification.
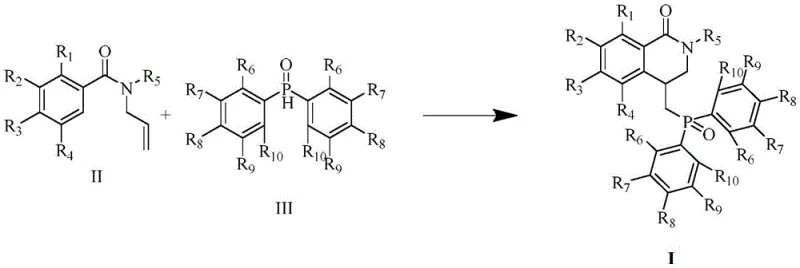
Mechanistic Insights into Electro-Oxidative Radical Cascade Cyclization
The mechanistic pathway of this transformation involves an anodic oxidation event that initiates a radical cascade. At the graphite anode, the phosphine oxide or the amide substrate undergoes single-electron transfer (SET) to generate a reactive radical intermediate. This radical species then adds across the alkene moiety of the N-allyl group, forming a new carbon-carbon bond and a carbon-centered radical. Subsequent intramolecular cyclization onto the aromatic ring, followed by rearomatization via further oxidation and deprotonation, yields the fused dihydroisoquinolone core. The beauty of this electrochemical approach lies in its tunability; by adjusting the current density and electrode potential, chemists can selectively access specific radical intermediates that might be difficult to generate using traditional chemical initiators. This level of control is crucial for managing selectivity and ensuring the formation of the desired regioisomer, particularly when dealing with substrates bearing diverse electronic substituents on the aromatic rings.
From an impurity control perspective, the absence of metal catalysts significantly simplifies the impurity profile. Traditional metal-catalyzed routes often suffer from metal-ligand complexation byproducts or homocoupling side reactions induced by the metal center. In this electrochemical system, the primary byproducts are derived from solvent oxidation or minor over-oxidation of the substrate, which are generally easier to separate via standard chromatographic techniques. The use of a platinum cathode ensures efficient reduction of protons to hydrogen gas, maintaining charge balance without introducing reducing agents that could interfere with the oxidative cycle. This clean reaction profile translates directly to higher purity crude products, reducing the burden on downstream purification units and increasing the overall yield of the isolated active pharmaceutical ingredient (API) intermediate.
How to Synthesize Phosphorylated Dihydroisoquinolone Efficiently
The practical implementation of this synthesis is straightforward and relies on standard electrochemical equipment available in most process development laboratories. The protocol involves dissolving the N-allyl-N-methoxybenzamide substrate and the diphenylphosphine oxide coupling partner in DMSO, along with the electrolyte. The mixture is then subjected to constant current electrolysis using inexpensive graphite and platinum electrodes. The detailed operational parameters, including current density, temperature, and stoichiometry, have been optimized to maximize yield and minimize energy consumption. For process chemists looking to replicate or scale this reaction, the robustness of the method across various substituted substrates provides a wide scope for library synthesis. The following section outlines the standardized procedural steps derived from the patent examples to ensure reproducibility and safety during scale-up operations.
- Dissolve N-allyl-N-methoxybenzamide and diphenylphosphine oxide in dimethyl sulfoxide (DMSO) with tetrabutylammonium tetrafluoroborate as the electrolyte.
- Set up an undivided cell with a graphite anode and a platinum sheet cathode, applying a constant current of 8 mA at 80°C.
- After 3 hours, cool the mixture, remove the solvent, and purify the crude product via column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical protocol offers tangible strategic benefits beyond mere technical novelty. The elimination of noble metal catalysts like silver nitrate or rhodium complexes removes a volatile cost component from the bill of materials. Precious metal prices are subject to significant market fluctuations and geopolitical supply risks; by substituting these with electricity and carbon-based electrodes, the manufacturing process becomes more resilient to external market shocks. Furthermore, the simplified purification workflow reduces the consumption of silica gel and solvents during chromatography, contributing to substantial cost savings in consumables. The shorter reaction times also imply higher asset utilization rates, allowing manufacturing facilities to produce more batches per year without capital investment in new reactors, thereby improving the overall return on invested capital (ROIC) for the production line.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the removal of expensive catalytic systems. Traditional methods often require loadings of 5-10 mol% of silver or rhodium salts, which represent a significant portion of the raw material cost. By utilizing electrons as the reagent, the variable cost per kilogram of product is drastically lowered. Additionally, the mild reaction conditions reduce energy demands for heating and cooling compared to high-temperature thermal processes. The use of commodity chemicals like DMSO and tetrabutylammonium salts, which are readily available in bulk quantities, further stabilizes the supply chain and prevents bottlenecks associated with specialized reagents. This economic efficiency makes the process highly competitive for the production of generic drug intermediates where margin pressure is intense.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry. This electrochemical method relies on starting materials that are structurally simple and commercially abundant, such as benzoyl chlorides and allylamines. Unlike complex ligands or specialized oxidants that may have single-source suppliers, the reagents for this process can be sourced from multiple global vendors, mitigating the risk of supply disruption. The robustness of the reaction across a wide range of substituents (halogens, alkyl groups, alkoxy groups) means that a single platform technology can be used to manufacture a diverse portfolio of intermediates. This flexibility allows manufacturers to respond quickly to changes in demand for different drug candidates without retooling the entire production line, ensuring a steady flow of materials to downstream formulation sites.
- Scalability and Environmental Compliance: Scaling electrochemical reactions has historically been a challenge, but modern flow electrochemistry solutions have made this increasingly viable. The undivided cell configuration described in the patent is inherently simpler to engineer for continuous flow than complex multiphase catalytic systems. From an environmental standpoint, the process aligns perfectly with green chemistry principles by avoiding toxic heavy metals and stoichiometric oxidants. This reduces the volume of hazardous waste requiring disposal and lowers the cost of environmental compliance and wastewater treatment. For multinational corporations with strict sustainability goals, adopting this technology demonstrates a commitment to reducing the carbon footprint of the supply chain, enhancing the corporate social responsibility (CSR) profile of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these nuances is critical for process engineers evaluating the feasibility of technology transfer. The answers highlight the operational simplicity and the distinct advantages over legacy methods, providing clarity for decision-makers assessing the risk-reward profile of adopting this new synthetic route for their specific pipeline assets.
Q: Does this electrochemical method require expensive noble metal catalysts?
A: No, the disclosed process eliminates the need for noble metal catalysts like silver or rhodium, utilizing electricity as the primary oxidant, which significantly reduces raw material costs and metal contamination risks.
Q: What is the typical reaction time compared to traditional thermal methods?
A: The electrochemical protocol achieves high yields within approximately 3 hours, whereas traditional silver-catalyzed methods often require over 18 hours, offering a substantial improvement in throughput efficiency.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the use of simple electrode materials like graphite and platinum, combined with mild reaction conditions and commercially available solvents like DMSO, makes the process highly amenable to scale-up in flow electrochemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorylated Dihydroisoquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electro-organic synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate innovative academic and patent technologies like CN116041394A into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of phosphorylated dihydroisoquinolone derivative meets the highest quality standards required by global regulatory agencies. Our commitment to quality assurance ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Whether you are in the early stages of drug discovery or preparing for commercial launch, our technical procurement team is ready to assist. We encourage you to request a Customized Cost-Saving Analysis to evaluate how switching to this electrochemical route can impact your overall project economics. Contact us today to discuss your specific requirements, and let us provide you with specific COA data and route feasibility assessments tailored to your project timeline. Together, we can accelerate the delivery of life-saving medicines to patients while adhering to the highest standards of sustainability and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
