Advanced Manufacturing of 3,5-Dichloro-4-Fluorobromobenzene: A Technical Breakthrough for Global Supply Chains
The global demand for fluorinated aromatic intermediates continues to surge, driven by their critical role in the development of next-generation agrochemicals and active pharmaceutical ingredients (APIs). A pivotal advancement in this sector is detailed in patent CN112047804A, which outlines a robust and economically superior preparation method for 3,5-dichloro-4-fluorobromobenzene (CAS 17318-08-0). This compound serves as a versatile building block, where the carbon-bromine bond facilitates further functionalization into complex fluorine-containing architectures. Unlike traditional methodologies that rely on expensive and difficult-to-source aniline precursors, this novel approach utilizes 2,6-dichlorofluorobenzene as the starting material. This strategic shift in raw material selection not only drastically lowers the entry cost for synthesis but also streamlines the purification process through efficient crystallization techniques rather than energy-intensive distillation. For R&D directors and procurement specialists alike, understanding this pathway offers a clear roadmap to optimizing supply chain resilience and reducing the overall cost of goods sold (COGS) for high-value fluorinated intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dichloro-4-fluorobromobenzene has been plagued by significant technical and economic hurdles inherent to the Sandmeyer reaction pathway. Conventional processes typically commence with 3,5-dichloro-4-fluoroaniline, a raw material that is notoriously difficult to prepare and commands a premium price in the fine chemical market. The core instability arises during the diazotization step; the presence of multiple strong electron-withdrawing substituents (chlorine and fluorine) on the aromatic ring renders the resulting diazonium salt highly unstable. This instability frequently leads to premature decomposition, resulting in inconsistent reaction yields and the formation of complex impurity profiles that are challenging to remove. Furthermore, prior art methods often necessitate the use of specialized tubular reactors to manage the exothermic nature of the diazotization safely, imposing heavy capital expenditure requirements on manufacturers. The reliance on stoichiometric amounts of cuprous bromide and the generation of heavy metal waste further complicate the environmental footprint, creating substantial disposal costs and regulatory compliance burdens for large-scale production facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN112047804A introduces a paradigm shift by reversing the synthetic logic to begin with the readily available 2,6-dichlorofluorobenzene. This route bypasses the instability issues of the aniline precursor entirely by establishing the halogen substitution pattern early in the sequence through a controlled nitration. A key innovation lies in the isolation of the intermediate 2,4-dichloro-3-fluoronitrobenzene via cooling crystallization at temperatures between -5°C and 10°C. This physical separation method is far more efficient and safer than traditional vacuum distillation, significantly reducing thermal degradation and product loss. By deferring the introduction of the bromine atom to a later stage via an oxidative bromination-deamination sequence, the process achieves superior control over regioselectivity. This approach not only enhances the overall yield, with examples demonstrating outputs exceeding 90%, but also simplifies the downstream purification, ensuring a high-purity final product that meets the stringent specifications required for pharmaceutical and agrochemical applications.
Mechanistic Insights into the Multi-Step Halogenation Sequence
The chemical elegance of this synthesis lies in its precise manipulation of electronic effects and reaction conditions to achieve high regioselectivity. The initial nitration of 2,6-dichlorofluorobenzene is directed by the interplay between the fluorine and chlorine substituents. Under the influence of mixed acid (HNO3/H2SO4), the electrophilic attack occurs preferentially at the position para to the fluorine atom, yielding 2,4-dichloro-3-fluoronitrobenzene as the major isomer. Following catalytic hydrogenation using palladium on carbon (Pd/C) or platinum on carbon (Pt/C), the nitro group is reduced to an amine, setting the stage for the critical bromination step. Unlike direct electrophilic bromination which might suffer from poly-substitution issues, this protocol employs a sulfation step to form the aniline sulfate salt. This protonation moderates the nucleophilicity of the amino group, preventing unwanted side reactions while directing the incoming bromine to the desired position. The subsequent addition of hydrogen peroxide acts as a co-oxidant, facilitating the regeneration of active brominating species and allowing the reaction to proceed with only 0.5 equivalents of elemental bromine, a significant departure from the stoichiometric excesses typically required.
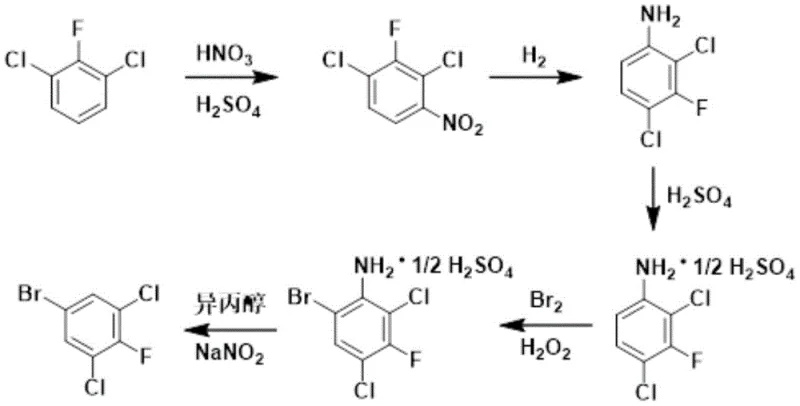
The final transformation involves a modified Sandmeyer-type deamination where the amino group is replaced by a hydrogen atom, effectively locking in the bromine substitution pattern established in the previous step. This is achieved through diazotization with sodium nitrite in the presence of isopropanol and a copper salt catalyst. Mechanistically, isopropanol serves as a reducing agent to decompose the diazonium intermediate, a greener alternative to the traditionally used sodium hypophosphite which generates phosphorous-containing waste. The use of isopropanol not only reduces environmental pollution but also improves the reaction yield by minimizing the formation of phenolic byproducts. The copper salt, preferably cuprous oxide, catalyzes the electron transfer processes necessary for the smooth evolution of nitrogen gas and the formation of the final carbon-hydrogen bond. This intricate sequence ensures that the final 3,5-dichloro-4-fluorobromobenzene is obtained with high purity, often exceeding 97%, ready for vacuum distillation and subsequent use in cross-coupling reactions.
How to Synthesize 3,5-Dichloro-4-Fluorobromobenzene Efficiently
Implementing this synthesis requires careful attention to temperature control and reagent addition rates to maximize safety and yield. The process begins with the nitration step where maintaining the reaction temperature between 20°C and 80°C is crucial to prevent over-nitration or oxidation. Following the isolation of the nitro intermediate via repeated cooling crystallization, the hydrogenation step should be conducted in solvents such as methanol or ethyl acetate under moderate hydrogen pressure (e.g., 2 MPa) to ensure complete reduction. The subsequent bromination and deamination steps are highly sensitive to pH and temperature; specifically, the diazotization must be performed at low temperatures (-40°C to 10°C) to maintain the stability of the diazonium salt before the addition of the copper catalyst and isopropanol. For a detailed, step-by-step operational guide including specific molar ratios and workup procedures, please refer to the standardized synthesis protocol below.
- Nitration of 2,6-dichlorofluorobenzene using mixed acid followed by cooling crystallization to isolate 2,4-dichloro-3-fluoronitrobenzene.
- Catalytic hydrogenation of the nitro compound to form 2,4-dichloro-3-fluoroaniline using Pd/C or Pt/C catalysts.
- Sulfation of the aniline followed by oxidative bromination using Br2 and H2O2, and final deamination using isopropanol and sodium nitrite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond mere technical feasibility. The primary driver for cost reduction is the substitution of the expensive 3,5-dichloro-4-fluoroaniline with 2,6-dichlorofluorobenzene, a commodity chemical with a robust global supply base and significantly lower market price. This raw material swap fundamentally alters the cost structure of the intermediate, offering substantial margin improvements for downstream API manufacturers. Furthermore, the process eliminates the need for specialized tubular reactors, allowing production to be executed in standard glass-lined or stainless steel reaction kettles found in most multipurpose fine chemical plants. This compatibility with existing infrastructure removes barriers to entry for contract manufacturing organizations (CMOs) and reduces the lead time required to qualify new production lines, ensuring a more agile and responsive supply chain capable of meeting fluctuating market demands without massive capital investment.
- Cost Reduction in Manufacturing: The economic benefits of this route are multifaceted, stemming from both raw material savings and process efficiencies. By reducing the stoichiometric requirement of elemental bromine from 1 equivalent to 0.5 equivalents through the synergistic use of hydrogen peroxide, the process significantly lowers the consumption of this hazardous and costly reagent. Additionally, the replacement of sodium hypophosphite with isopropanol in the deamination step reduces the load on wastewater treatment systems, lowering the operational expenditures associated with environmental compliance and waste disposal. The high yields achieved through crystallization purification rather than distillation also mean less material is lost to thermal degradation or column hold-up, directly improving the mass balance and overall process economy.
- Enhanced Supply Chain Reliability: Reliance on a single, difficult-to-source precursor like 3,5-dichloro-4-fluoroaniline creates a single point of failure in the supply chain. By shifting to 2,6-dichlorofluorobenzene, manufacturers can tap into a broader network of suppliers, mitigating the risk of shortages and price volatility. The mild reaction conditions and the use of common solvents like methanol and isopropanol further enhance supply security, as these materials are universally available and not subject to the same regulatory restrictions as specialized catalysts or reagents. This diversification of the supply base ensures business continuity and allows for more accurate long-term planning and inventory management.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns perfectly with modern green chemistry principles. The reduction in heavy metal usage (copper salts are used in catalytic amounts) and the minimization of bromine waste address key environmental, social, and governance (ESG) criteria increasingly demanded by multinational corporations. The ability to scale this process from kilogram to multi-ton quantities using standard equipment means that production can be ramped up quickly to meet commercial deadlines without the lengthy engineering studies required for high-pressure or high-temperature processes. This scalability ensures that the supply of high-purity 3,5-dichloro-4-fluorobromobenzene can grow in tandem with the development pipelines of our partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these nuances is essential for R&D teams evaluating process transfer and for procurement professionals assessing vendor qualifications. We encourage stakeholders to review these points to gain a comprehensive understanding of how this method compares to legacy technologies.
Q: What is the primary advantage of using 2,6-dichlorofluorobenzene over 3,5-dichloro-4-fluoroaniline?
A: 2,6-Dichlorofluorobenzene is significantly more accessible and cost-effective than 3,5-dichloro-4-fluoroaniline. Additionally, the new route avoids the instability issues associated with diazotizing highly electron-deficient anilines in traditional Sandmeyer reactions.
Q: How does the new process improve environmental compliance regarding bromine usage?
A: The introduction of hydrogen peroxide in the bromination step acts as an oxidant that regenerates bromine species, effectively reducing the stoichiometric requirement of elemental bromine from 1 equivalent to approximately 0.5 equivalents, thereby minimizing hazardous waste.
Q: Can this process be scaled for industrial production without special equipment?
A: Yes, the patent explicitly states that the method utilizes standard reaction kettles and does not require specialized tubular reactors or extreme pressure conditions, making it highly suitable for immediate commercial scale-up in existing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dichloro-4-Fluorobromobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from pilot plant to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs and analytical instruments capable of verifying stringent purity specifications, guaranteeing that every batch of 3,5-dichloro-4-fluorobromobenzene meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your long-term business goals.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality intermediates consistently. Contact us today to secure a sustainable and cost-effective supply of this critical fluorinated building block.
