Advanced Liquid-Phase Synthesis of 2,2'-Dithiosalicylic Acid for Commercial Scale-Up
Advanced Liquid-Phase Synthesis of 2,2'-Dithiosalicylic Acid for Commercial Scale-Up
The global demand for high-performance sulfur-containing intermediates continues to surge, driven by the expanding needs of the pharmaceutical, agrochemical, and polymer additive sectors. At the forefront of this innovation is the synthesis of 2,2'-dithiosalicylic acid (DSTA), a critical building block for fungicides like BIT and photoinitiators such as ITX. A pivotal advancement in this field is detailed in Chinese Patent CN101805277B, which discloses a robust preparation method utilizing liquid sulfur dioxide. This technology represents a paradigm shift from traditional gas-liquid processes to a more efficient homogeneous liquid-phase reaction system. By leveraging specific metal catalysts and precise temperature controls ranging from cryogenic conditions to elevated thermal treatments, this method achieves exceptional reaction yields and product purity. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this patented route is essential for securing a competitive supply chain. The structural integrity and functional versatility of the target molecule, as illustrated below, underscore its importance in modern chemical manufacturing.
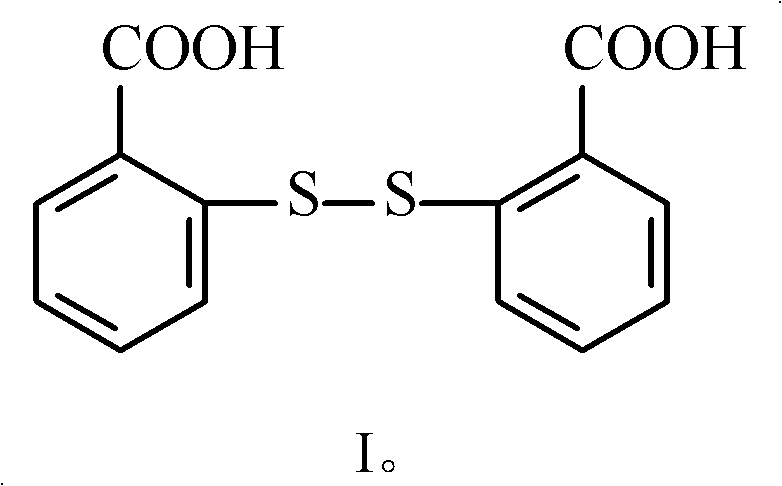
The molecular architecture of 2,2'-dithiosalicylic acid features a symmetric disulfide linkage connecting two salicylic acid moieties, a configuration that imparts unique stability and reactivity profiles suitable for diverse applications. The patent highlights that this compound, with a molecular weight of 306.06 and a melting point of 288-290°C, serves as a vital precursor in the synthesis of biocides and UV-curing agents. The ability to produce this compound with minimal impurities is paramount for downstream applications where trace contaminants could compromise the efficacy of the final formulation. Consequently, the methodology described in CN101805277B offers a strategic advantage by integrating rigorous purification steps directly into the reaction workflow, ensuring that the final isolated product meets the stringent specifications required by top-tier multinational corporations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dithiodibenzoic acid derivatives has been plagued by significant engineering and environmental challenges, primarily stemming from the use of gaseous sulfur dioxide in heterogeneous reaction systems. As noted in prior art, such as the methods reported by He Xianzhang, reacting diazonium solutions with SO2 gas results in poor gas-liquid phase contact. This inefficiency leads to a substantial portion of the sulfur dioxide escaping into the tail gas system rather than participating in the desired chemical transformation. The consequences of this low utilization rate are twofold: firstly, it necessitates the consumption of excessive raw materials to drive the reaction to completion, inflating production costs; secondly, it creates a severe environmental burden. The escaped SO2 must be neutralized using large quantities of alkali in scrubbing systems, which generates voluminous amounts of sulfur-containing wastewater and solid waste residues. Furthermore, traditional polysulfide routes, while yielding around 90%, often involve complex separation procedures and generate difficult-to-treat effluent streams, making them increasingly untenable under modern environmental regulations.
The Novel Approach
In stark contrast, the innovative process outlined in CN101805277B circumvents these issues by employing liquid sulfur dioxide as the reactant, thereby establishing a homogeneous liquid-phase reaction environment. This fundamental change ensures that the diazonium intermediate and the sulfur source are in intimate contact throughout the reaction vessel, drastically improving mass transfer kinetics. The protocol dictates that the diazonium solution, prepared from anthranilic acid, is introduced into a reactor containing liquid SO2, catalysts, and cocatalysts at temperatures between -20°C and -10°C. This controlled低温 environment stabilizes the reactive intermediates and prevents premature decomposition. Following the initial coupling, the mixture is heated to 80-100°C to facilitate the formation of the disulfide bond and complete the cyclization or coupling process. The result is a streamlined workflow that not only boosts reaction yields to impressive levels, often exceeding 95%, but also significantly simplifies the post-reaction workup, requiring merely cooling and filtration to isolate the high-purity product.
Mechanistic Insights into Catalytic Liquid-Phase Sulfonation
The success of this synthesis hinges on a sophisticated catalytic system designed to mediate the radical or ionic coupling of the diazonium species with sulfur dioxide. The patent specifies the use of transition metal salts, including copper, iron, cobalt, or nickel variants such as copper sulfate, iron sulfate, or cobalt acetate. These metal centers likely function by facilitating electron transfer processes that stabilize the sulfur-centered radicals generated from the liquid SO2, promoting their attack on the aromatic ring of the diazonium salt. Crucially, the inclusion of halide promoters—specifically iodides and bromides like potassium iodide, sodium bromide, or cuprous iodide—acts as a synergistic cocatalyst. These halides are known to enhance the reactivity of diazonium salts in Sandmeyer-type reactions, potentially by forming transient organometallic complexes that lower the activation energy for the sulfur insertion step. The precise stoichiometry is equally critical, with the patent recommending a molar ratio of anthranilic acid to sodium nitrite to HCl of approximately 1.0:1.1-1.2:3.0-3.5. This slight excess of nitrite and acid ensures complete diazotization while minimizing the formation of azo-coupling byproducts, which are common impurities in such systems.
Impurity control is further achieved through the meticulous management of reaction thermodynamics and phase behavior. By maintaining the initial reaction at -20 to -10°C, the process suppresses side reactions such as hydrolysis of the diazonium group to phenols or thermal decomposition to chlorobenzenes. The subsequent heating phase at 80-100°C is carefully timed (0.5 to 2 hours) to ensure full conversion without degrading the sensitive disulfide linkage. The use of liquid SO2 also means that the reaction medium itself acts as a solvent and reactant, reducing the need for additional organic solvents that would later require removal and recycling. This "solvent-free" or "self-solventing" characteristic contributes to the high purity observed in the examples, where HPLC analysis consistently shows purity levels above 97%, with some embodiments reaching 98.8%. Such high fidelity in the crude product reduces the burden on downstream purification units, directly translating to cost reduction in fine chemical manufacturing.
How to Synthesize 2,2'-Dithiosalicylic Acid Efficiently
Implementing this synthesis route requires strict adherence to the operational parameters defined in the patent to ensure safety and reproducibility. The process begins with the preparation of the diazonium salt, where anthranilic acid is dissolved in dilute hydrochloric acid and cooled to 0-10°C before the slow addition of aqueous sodium nitrite. Once the diazotization is confirmed, typically via starch-iodide paper testing, the solution is transferred to a second reactor pre-charged with the catalyst system and liquid sulfur dioxide. The critical step involves maintaining the cryogenic temperature during the addition to manage the exotherm and prevent runaway reactions.
- Diazotization: Dissolve anthranilic acid in hydrochloric acid and water, then add sodium nitrite solution at 0-10°C to form the diazonium salt.
- Coupling Reaction: Mix the diazonium solution with liquid sulfur dioxide at -20 to -10°C using copper or iron catalysts and halide promoters.
- Thermal Treatment & Isolation: Heat the mixture to 80-100°C to complete the reaction, then cool and filter to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this liquid-phase technology offers compelling strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the production workflow, which eliminates the need for complex gas-handling infrastructure and extensive tail gas treatment facilities. By switching from a gas-liquid to a liquid-liquid system, manufacturers can significantly reduce capital expenditure on specialized reactors and scrubbing towers, leading to substantial cost savings in facility maintenance and operation. Moreover, the high atom economy of the reaction, driven by the efficient utilization of sulfur dioxide, means that raw material consumption is optimized. This efficiency buffers the supply chain against volatility in raw material pricing, providing a more stable cost base for long-term contracts. The simplified isolation procedure, which often requires only filtration and drying, further reduces energy consumption associated with solvent recovery and distillation, aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive gas-phase processing equipment and the reduction in alkali consumption for tail gas neutralization directly lower the variable costs per kilogram of product. Additionally, the high yield minimizes the loss of valuable anthranilic acid starting material, ensuring that every unit of input generates maximum output value. The process also avoids the use of costly organic solvents in the reaction step, further driving down the bill of materials.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including anthranilic acid, sodium nitrite, and liquid sulfur dioxide, are commodity chemicals with robust global supply networks. This availability reduces the risk of supply disruptions compared to processes relying on exotic or specialized reagents. Furthermore, the scalability of the liquid-phase reaction allows for flexible production scheduling, enabling manufacturers to respond rapidly to fluctuations in market demand without lengthy changeover times.
- Scalability and Environmental Compliance: The process generates significantly less wastewater and solid waste compared to traditional polysulfide or gas-phase methods, simplifying compliance with increasingly stringent environmental regulations. The reduced environmental footprint facilitates easier permitting for capacity expansion and lowers the costs associated with waste disposal and treatment. This "green chemistry" profile enhances the brand reputation of the supplier and meets the ESG criteria of major multinational buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,2'-dithiosalicylic acid, derived directly from the proprietary data and experimental examples provided in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own supply chains.
Q: Why is liquid sulfur dioxide preferred over gaseous SO2 in this synthesis?
A: Using liquid sulfur dioxide creates a homogeneous liquid-phase reaction with the diazonium solution, ensuring sufficient contact between reactants. This significantly improves SO2 utilization efficiency compared to gas-liquid reactions, reduces tail gas emissions, and minimizes the need for alkali neutralization, thereby lowering wastewater generation.
Q: What represents the optimal catalyst system for maximizing yield?
A: The patent indicates that metal salts such as copper sulfate, iron sulfate, cobalt acetate, or nickel acetate, combined with halide promoters like potassium iodide or sodium bromide, provide superior results. Specifically, optimized ratios can achieve yields exceeding 95% with purity levels above 97%.
Q: How does this method address environmental concerns in sulfur chemistry?
A: Traditional methods often generate large amounts of sulfur-containing wastewater and require extensive tail gas treatment. This novel process reduces sulfur dioxide emissions due to higher reaction efficiency and simplifies separation, resulting in substantially less production waste water and废渣 (solid waste), making it more environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Dithiosalicylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics seen in patent examples are faithfully reproduced at scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Whether your application requires DSTA for fungicide synthesis or as a photoinitiator precursor, our capability to deliver consistent, high-quality material is unmatched in the region.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain visibility into the potential economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
