Scalable Synthesis of 2-Carboxyphenylboronic Acid Salts for High-Volume API Production
Scalable Synthesis of 2-Carboxyphenylboronic Acid Salts for High-Volume API Production
The pharmaceutical industry constantly seeks robust and scalable synthetic routes for critical intermediates, particularly those used in the manufacture of blockbuster drugs like Telmisartan. Patent CN101113153A introduces a significant technological advancement in the preparation of 2-carboxyphenylboronic acid and its derivatives, addressing long-standing challenges in purity and isolation. This patent describes a novel process involving the metallation of 4,4-dimethyl-2-phenyl-oxazoline followed by boronation, offering a superior alternative to traditional oxidation methods. For R&D directors and procurement specialists, understanding this chemistry is vital for securing a reliable supply chain of high-purity pharmaceutical intermediates. The ability to isolate the product as a stable sodium salt represents a key breakthrough in process chemistry, enabling efficient large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-carboxyphenylboronic acid has relied on the oxidation of 2-tolylboronic acid using strong oxidizing agents such as potassium permanganate. While conceptually straightforward, this approach suffers from severe drawbacks when attempted on a technical or industrial scale. The reaction mixture becomes incredibly complex, making the separation of the desired acid from manganese byproducts and unreacted starting materials extremely difficult. Consequently, the yield is often compromised, and the resulting product lacks the purity required for sensitive cross-coupling reactions used in API synthesis. Furthermore, the free acid form is prone to degradation and difficult to handle, posing significant risks for transportation and long-term storage, which creates bottlenecks for supply chain managers seeking consistency.
The Novel Approach
In stark contrast, the method disclosed in CN101113153A utilizes a directed ortho-metallation strategy that fundamentally changes the purification landscape. By employing 4,4-dimethyl-2-phenyl-oxazoline as a substrate, the process leverages the coordinating ability of the nitrogen and oxygen atoms to direct a Grignard reagent specifically to the ortho-position. This precision eliminates the formation of regioisomers and simplifies the reaction profile. The subsequent conversion to a boronic ester and finally to the carboxylate salt allows for a purification strategy based on crystallization rather than difficult chromatographic separations. This shift from oxidation to metallation-boronation not only improves chemical efficiency but also drastically enhances the operational safety and scalability of the manufacturing process.
Mechanistic Insights into Oxazoline-Directed Metallation and Boronation
The core of this innovative synthesis lies in the formation of the organomagnesium intermediate. The reaction begins with the treatment of 4,4-dimethyl-2-phenyl-oxazoline with a Grignard reagent, such as cyclohexylmagnesium chloride, in an aprotic solvent like tetrahydrofuran (THF). The oxazoline ring acts as a potent directing group, coordinating with the magnesium species to facilitate the deprotonation of the aromatic ring at the position ortho to the heterocycle. This results in the formation of the stable magnesiated species shown below, which serves as the nucleophile for the subsequent borylation step. The choice of solvent and temperature control during this phase is critical to prevent side reactions and ensure complete conversion of the starting material.
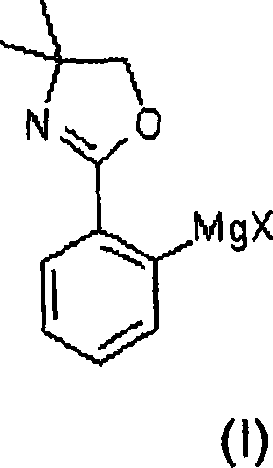
Following the metallation, the reactive intermediate is treated with a borate ester, typically trimethyl borate, to install the boron functionality. This step converts the carbon-magnesium bond into a carbon-boron bond, yielding a protected boronic ester derivative. The protection of the boronic acid as an ester is a strategic move that prevents premature protodeboronation or oligomerization, which are common pitfalls in boronic acid chemistry. The resulting intermediate retains the oxazoline protecting group on the carboxylic acid precursor, allowing for a controlled hydrolysis later in the sequence. This multi-step protection-deprotection strategy ensures that the sensitive functional groups remain intact until the final isolation stage, maximizing the overall yield of the process.
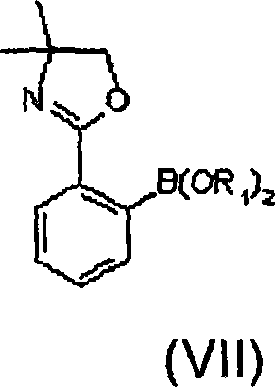
How to Synthesize 2-Carboxyphenylboronic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable intermediate with high fidelity. The process involves three distinct phases: the generation of the Grignard adduct, the borylation reaction, and the final hydrolysis and salt formation. Each step is optimized for industrial feasibility, utilizing common solvents and reagents that are readily available in the global chemical market. The detailed standard operating procedures for temperature control, addition rates, and workup conditions are essential for replicating the high purity levels reported in the patent data. Manufacturers aiming to adopt this route should pay close attention to the stoichiometry of the Grignard reagent and the specific hydrolysis conditions to ensure optimal crystal formation of the final salt.
- Metallation of 4,4-dimethyl-2-phenyl-oxazoline using a Grignard reagent in THF to form the ortho-magnesiated intermediate.
- Reaction of the magnesiated intermediate with a borate ester (e.g., trimethyl borate) to generate the protected boronic ester derivative.
- Hydrolysis of the ester and oxazoline ring followed by neutralization and crystallization to isolate the high-purity sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metallation-based process offers tangible benefits that extend beyond simple chemical yield. The primary advantage lies in the isolation of the product as a sodium salt, which possesses superior physical properties compared to the free acid. This solid form is non-hygroscopic, stable, and easy to handle, significantly reducing the risks associated with storage and logistics. By eliminating the need for complex purification steps like column chromatography or extensive recrystallization of the free acid, the process reduces the consumption of solvents and consumables, leading to substantial cost savings in manufacturing. Furthermore, the high purity achievable through simple filtration ensures that downstream coupling reactions proceed with minimal interference from impurities.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous oxidizing agents like potassium permanganate removes the need for costly waste treatment processes associated with heavy metal disposal. Additionally, the ability to isolate the product via crystallization rather than extraction or chromatography significantly lowers the operational expenditure related to solvent recovery and labor. The use of standard Grignard reagents and borate esters, which are commodity chemicals, further stabilizes the raw material costs, providing a predictable pricing structure for long-term contracts.
- Enhanced Supply Chain Reliability: The robust nature of the sodium salt form ensures that the material can be stockpiled without degradation, acting as a buffer against supply disruptions. The simplified purification process reduces the batch cycle time, allowing manufacturers to respond more quickly to fluctuations in demand. By avoiding the technical difficulties associated with the oxidation route, suppliers can guarantee consistent quality and availability, which is critical for maintaining the production schedules of downstream API manufacturers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: This process is inherently scalable, as demonstrated by the patent examples which utilize standard reactor configurations and agitation methods. The absence of heavy metal contaminants in the final product simplifies the regulatory compliance burden, as there is no need for rigorous testing and removal of trace metals like manganese. The waste stream is primarily composed of organic salts and solvents that can be managed through standard incineration or recycling protocols, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-carboxyphenylboronic acid salts. These insights are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these nuances helps stakeholders make informed decisions regarding vendor qualification and process adoption. The answers reflect the consensus on best practices for handling boronic acid intermediates in a GMP environment.
Q: Why is the sodium salt form preferred over the free acid for this intermediate?
A: The patent highlights that the free acid is difficult to separate from reaction byproducts on a technical scale. Converting it to the sodium salt allows for high-purity isolation (>99.5%) via crystallization, significantly improving stability and handling for transport and storage.
Q: What is the role of the oxazoline ring in this synthesis?
A: The 4,4-dimethyl-2-phenyl-oxazoline acts as a powerful directing group. It facilitates the regioselective metallation at the ortho-position of the phenyl ring by the Grignard reagent, ensuring the boron atom is installed exactly where needed for the subsequent coupling reactions.
Q: How does this process improve supply chain reliability compared to oxidation methods?
A: Traditional oxidation methods using potassium permanganate often result in complex mixtures that are hard to purify. This metallation-boronation route yields a crystalline salt that can be easily filtered and washed, removing magnesium salts and amine byproducts, thus ensuring a consistent and reliable supply of high-quality material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Carboxyphenylboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team has extensively analyzed the process described in CN101113153A and possesses the expertise to implement this advanced synthesis route on a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify the polymorphic form and water content of the sodium salt, guaranteeing that every batch performs predictably in your synthesis campaigns.
We invite you to collaborate with us to optimize your supply chain for Telmisartan and other related APIs. By leveraging our manufacturing capabilities, you can achieve significant efficiencies and reduce the risks associated with intermediate sourcing. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can add value to your operations.
