Advanced Solvent-Free Synthesis of 2,6-Dichlorobenzonitrile for Scalable Agrochemical Production
The global demand for high-performance agrochemical intermediates continues to drive innovation in synthetic methodology, particularly for key building blocks like 2,6-dichlorobenzonitrile. Patent CN113038835B introduces a transformative approach to producing this critical herbicide intermediate through a highly selective denitrochlorination process. This technology addresses long-standing inefficiencies in traditional manufacturing by utilizing a solvent-free protocol that maximizes atom economy while minimizing environmental impact. By leveraging specific reaction parameters, the method achieves exceptional control over side reactions, ensuring that the final product meets the rigorous purity standards required by modern pharmaceutical and agricultural sectors. The strategic shift from expensive starting materials to value-added waste utilization represents a paradigm shift in how we approach industrial organic synthesis today.
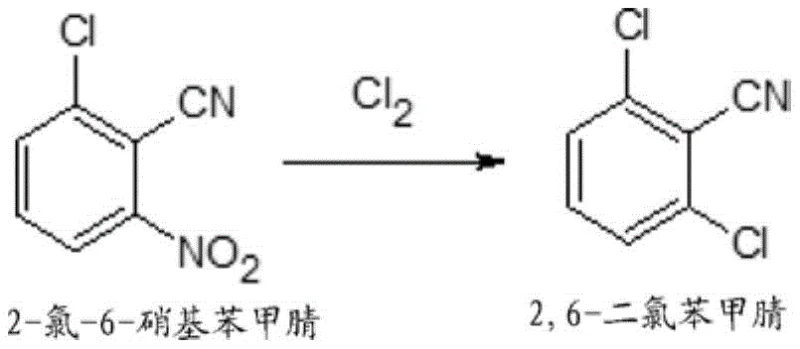
For R&D directors and process chemists, understanding the limitations of legacy methods is crucial for evaluating new technological adoptions. Conventional industrial routes often rely on the gas-phase catalytic ammoxidation of 2,6-dichlorotoluene, a process that demands substantial capital investment in specialized high-temperature reactors and corrosion-resistant equipment. Furthermore, the raw material 2,6-dichlorotoluene itself is often costly and subject to supply chain volatility, creating bottlenecks in production planning. Alternative historical methods involving lithium chloride in aprotic solvents have proven industrially unfavorable due to the high cost of reagents and the complexity of downstream processing. These traditional pathways frequently struggle with selectivity issues, leading to complex impurity profiles that require extensive and yield-eroding purification steps to resolve.
In stark contrast, the novel approach detailed in the patent utilizes 2-chloro-6-nitrobenzonitrile as a versatile precursor, which can be economically sourced from the cyanation of 2,3-dichloronitrobenzene, a common byproduct in other chlorination processes. 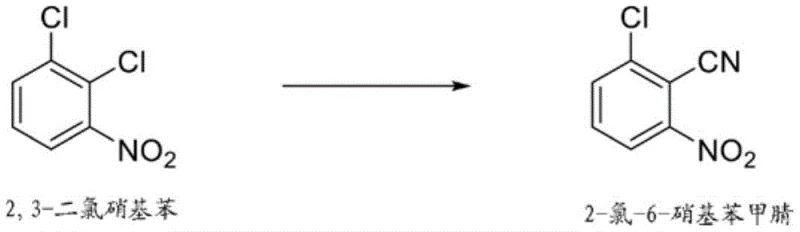 This strategy not only valorizes waste streams but also simplifies the supply chain by relying on more abundant feedstocks. The core innovation lies in the solvent-free treatment of this nitrile with chlorine gas at elevated temperatures, typically between 150°C and 200°C. By eliminating the need for bulk solvents, the process drastically reduces the volume of waste liquid generated and lowers the energy consumption associated with solvent recovery and distillation. This streamlined workflow enhances the overall process mass intensity (PMI), making it a superior candidate for green chemistry initiatives and sustainable manufacturing goals.
This strategy not only valorizes waste streams but also simplifies the supply chain by relying on more abundant feedstocks. The core innovation lies in the solvent-free treatment of this nitrile with chlorine gas at elevated temperatures, typically between 150°C and 200°C. By eliminating the need for bulk solvents, the process drastically reduces the volume of waste liquid generated and lowers the energy consumption associated with solvent recovery and distillation. This streamlined workflow enhances the overall process mass intensity (PMI), making it a superior candidate for green chemistry initiatives and sustainable manufacturing goals.
Mechanistic Insights into Selective Denitrochlorination
The mechanistic pathway of this transformation involves a complex interplay of electrophilic substitution and radical mechanisms driven by the thermal activation of chlorine gas. Under the optimized conditions of 185°C to 195°C, the chlorine molecule acts as both a chlorinating agent and an oxidant, facilitating the displacement of the nitro group while simultaneously introducing a chlorine atom at the ortho position. The absence of solvent plays a critical role here, as it prevents the stabilization of unwanted radical intermediates that could lead to polymerization or excessive degradation of the nitrile functionality. Careful regulation of the chlorine purge rate is essential to maintain the reaction within the kinetic window that favors the desired substitution over competitive oxidation pathways. This precise control ensures that the electron-withdrawing nature of the nitrile group directs the incoming electrophile to the correct position on the aromatic ring with high fidelity.
A major challenge in halogenation chemistry is the suppression of over-chlorination and the formation of toxic byproducts, which this patent addresses through rigorous parameter control. 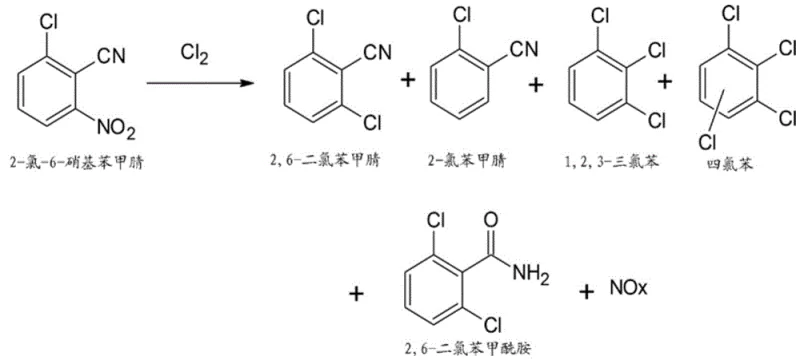 As illustrated in the reaction scheme, potential impurities such as 1,2,3-trichlorobenzene (TCB), 2-chlorobenzonitrile, and 2,6-dichlorobenzamide can form if the reaction environment is not strictly managed. The patented method limits the formation of these species to less than 2%, and often below 0.1% after purification, by optimizing the stoichiometric ratio of chlorine to substrate between 1:1 and 1:5. Additionally, the continuous removal of nitrogen oxides (NOx) prevents their re-entry into the reaction cycle, which could otherwise catalyze unwanted nitration or oxidation side reactions. This level of impurity control is vital for downstream applications where trace contaminants can affect the efficacy or safety of the final agrochemical formulation.
As illustrated in the reaction scheme, potential impurities such as 1,2,3-trichlorobenzene (TCB), 2-chlorobenzonitrile, and 2,6-dichlorobenzamide can form if the reaction environment is not strictly managed. The patented method limits the formation of these species to less than 2%, and often below 0.1% after purification, by optimizing the stoichiometric ratio of chlorine to substrate between 1:1 and 1:5. Additionally, the continuous removal of nitrogen oxides (NOx) prevents their re-entry into the reaction cycle, which could otherwise catalyze unwanted nitration or oxidation side reactions. This level of impurity control is vital for downstream applications where trace contaminants can affect the efficacy or safety of the final agrochemical formulation.
How to Synthesize 2,6-Dichlorobenzonitrile Efficiently
Implementing this synthesis route requires careful attention to thermal management and gas handling systems to ensure both safety and reproducibility. The process begins with the preparation of high-quality 2-chloro-6-nitrobenzonitrile, followed by its introduction into a reactor capable of withstanding corrosive chlorine environments at high temperatures. Operators must monitor the evolution of gases closely to adjust the chlorine feed rate dynamically, ensuring that the reaction remains exothermic but controlled. Detailed standard operating procedures regarding temperature ramping and quenching protocols are essential to prevent thermal runaways and ensure consistent batch-to-batch quality. For a comprehensive breakdown of the specific molar ratios, heating profiles, and work-up procedures, please refer to the standardized synthesis guide below.
- Preparation of the precursor 2-chloro-6-nitrobenzonitrile via cyanation of 2,3-dichloronitrobenzene using metal cyanides and copper catalysts in DMF.
- Charging the purified 2-chloro-6-nitrobenzonitrile into a reactor equipped with a chlorine gas inlet and acid scrubber system.
- Heating the reaction mass to 185-195°C and introducing chlorine gas at a controlled rate to effect selective denitrochlorination without solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers a compelling value proposition by decoupling production costs from the volatile pricing of specialty toluene derivatives. The ability to utilize 2,3-dichloronitrobenzene, often available as a surplus byproduct from other chlorination campaigns, creates a robust and cost-effective supply chain foundation. By eliminating the need for expensive solvents and reducing the reliance on precious metal catalysts found in alternative cyanation routes, the overall variable cost of goods sold is significantly reduced. This economic efficiency allows manufacturers to offer more competitive pricing structures to their clients while maintaining healthy margins, a critical factor in the highly price-sensitive agrochemical market. Furthermore, the simplified equipment requirements mean that production can be scaled up in existing facilities without the need for prohibitive capital expenditures on new infrastructure.
Supply chain reliability is further enhanced by the process inherent safety features and environmental compliance measures, which mitigate the risk of regulatory shutdowns. 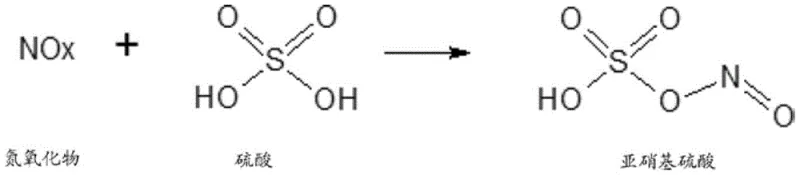 The integrated capture of NOx gases into nitrosylsulfuric acid not only solves a major waste disposal challenge but also generates a secondary revenue stream from the sale of this diazotization reagent. This circular economy approach transforms a potential environmental liability into a value-added co-product, aligning with increasingly strict global emissions standards. For supply chain heads, this means a more resilient operation that is less susceptible to fines or production halts due to environmental non-compliance. The robustness of the solvent-free system also simplifies logistics, as there is no need to procure, store, or dispose of large volumes of hazardous organic solvents, thereby reducing the overall logistical footprint and associated risks.
The integrated capture of NOx gases into nitrosylsulfuric acid not only solves a major waste disposal challenge but also generates a secondary revenue stream from the sale of this diazotization reagent. This circular economy approach transforms a potential environmental liability into a value-added co-product, aligning with increasingly strict global emissions standards. For supply chain heads, this means a more resilient operation that is less susceptible to fines or production halts due to environmental non-compliance. The robustness of the solvent-free system also simplifies logistics, as there is no need to procure, store, or dispose of large volumes of hazardous organic solvents, thereby reducing the overall logistical footprint and associated risks.
- Cost Reduction in Manufacturing: The elimination of solvents and the use of abundant feedstocks drastically lower raw material and waste treatment expenses. By avoiding the high energy costs associated with solvent recovery distillation columns, the process achieves substantial operational savings. The conversion of waste NOx into a sellable chemical further offsets production costs, creating a multi-faceted economic advantage. This holistic cost structure makes the technology highly attractive for large-scale commercial deployment where margin optimization is paramount.
- Enhanced Supply Chain Reliability: Sourcing precursors from widely available chlorination byproducts reduces dependency on single-source suppliers of specialized toluenes. The simplified reaction setup requires less maintenance and has fewer failure points compared to complex gas-phase ammoxidation units. This reliability ensures consistent delivery schedules for customers, fostering stronger long-term partnerships and trust in the supply base. The ability to pivot feedstock sources based on market availability adds a layer of strategic flexibility to procurement planning.
- Scalability and Environmental Compliance: The solvent-free nature of the reaction inherently reduces the volume of hazardous waste, simplifying the permitting process for capacity expansion. The efficient scrubbing system ensures that emissions remain well within regulatory limits, future-proofing the facility against tightening environmental laws. This scalability allows manufacturers to respond quickly to spikes in market demand without facing significant regulatory hurdles. The green chemistry credentials of the process also enhance the brand reputation of the supplier in sustainability-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this denitrochlorination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature to ensure accuracy and relevance. Understanding these details helps stakeholders make informed decisions about adopting this route for their specific manufacturing needs. The answers cover aspects ranging from impurity profiles to equipment requirements, providing a holistic view of the technology's viability.
Q: What are the primary advantages of the solvent-free denitrochlorination method over traditional ammoxidation?
A: The solvent-free denitrochlorination method eliminates the need for expensive solvents and reduces waste generation significantly. Unlike traditional ammoxidation which requires costly 2,6-dichlorotoluene and heavy equipment investment, this route utilizes readily available nitro-compounds and operates under simpler conditions, leading to lower capital expenditure and operational costs.
Q: How does the process manage the formation of hazardous NOx byproducts?
A: The process incorporates an integrated scrubbing system where evolved NOx gases are continuously removed and reacted with concentrated sulfuric acid. This converts the hazardous gases into nitrosylsulfuric acid, a valuable diazotization reagent, thereby turning a potential environmental liability into a commercially viable co-product while ensuring regulatory compliance.
Q: What level of purity can be achieved with this synthetic route?
A: By strictly controlling chlorine addition rates and reaction temperatures between 185°C and 195°C, the method achieves high selectivity. Post-reaction purification steps, including crystallization and washing, consistently yield 2,6-dichlorobenzonitrile with purity levels exceeding 99%, meeting stringent specifications for agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dichlorobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN113038835B to maintain competitiveness in the global agrochemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2,6-dichlorobenzonitrile meets the highest international standards. Our commitment to process excellence means we can deliver high-purity intermediates with consistent quality, supporting your R&D and manufacturing timelines effectively.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can drive value for your organization. Let us be your partner in achieving sustainable and cost-efficient production of complex chemical intermediates.
