Advanced Biocatalytic Synthesis of Chiral Amino Alcohols for Pharmaceutical Manufacturing
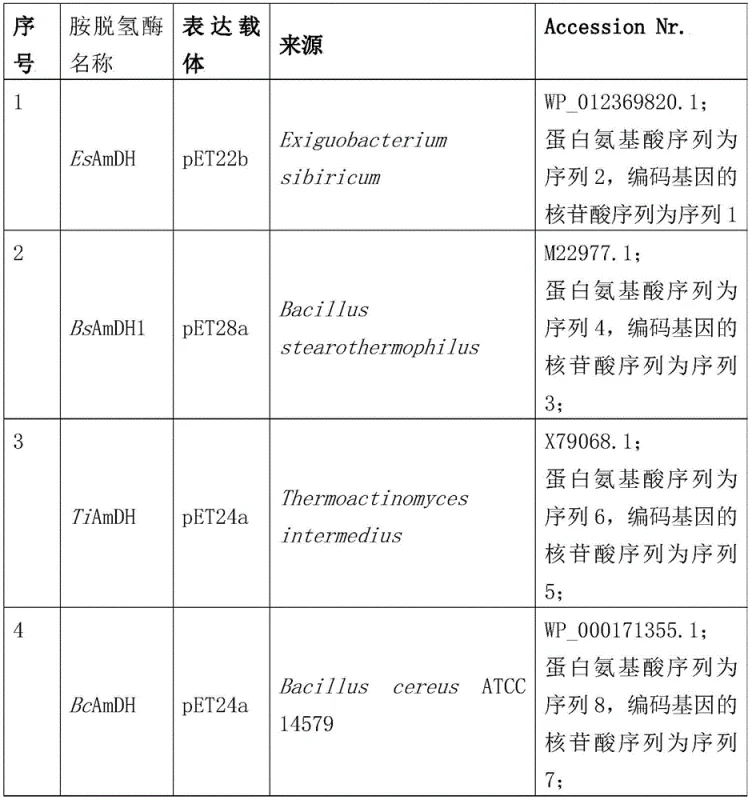
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access critical chiral building blocks, particularly amino alcohols which serve as pivotal intermediates for drugs like Ethambutol and Dolutegravir. A significant breakthrough in this domain is detailed in the Chinese patent CN112779232B, which discloses a robust synthetic method for chiral amine alcohol compounds utilizing specific amine dehydrogenases. This technology represents a paradigm shift from traditional harsh chemical syntheses to highly selective biocatalytic processes, offering a reliable pharmaceutical intermediate supplier with the capability to deliver high-purity products. The patent highlights the application of biological materials, including specific amine dehydrogenases derived from organisms such as Bacillus stearothermophilus and Exiguobacterium sibiricum, to catalyze the asymmetric reductive amination of hydroxyketone substrates. By leveraging these engineered enzymes, manufacturers can achieve exceptional stereocontrol and yield, addressing the long-standing challenges of cost and environmental impact associated with producing optically pure amines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral amino alcohols like (S)-2-amino-1-butanol and (R)-3-amino-1-butanol has relied heavily on chemical synthesis routes that are fraught with inefficiencies and safety concerns. Traditional methods often involve the reduction of amino acids using hazardous reagents such as lithium aluminium hydride or high-pressure hydrogenation over nickel catalysts, which require extreme temperatures and pressures that pose significant operational risks. Furthermore, these chemical pathways frequently result in racemic mixtures, necessitating complex and costly resolution steps using chiral acids like dibenzoyl tartaric acid to isolate the desired enantiomer, which inherently limits the maximum theoretical yield to 50 percent unless dynamic kinetic resolution is employed. The reliance on transition metal catalysts also introduces the risk of heavy metal contamination in the final product, requiring rigorous and expensive purification protocols to meet the stringent purity specifications demanded by regulatory bodies for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel biocatalytic approach outlined in the patent data utilizes engineered amine dehydrogenases to directly convert inexpensive hydroxyketone substrates into chiral amino alcohols with remarkable precision. This method operates under mild aqueous conditions, typically at temperatures ranging from 20°C to 70°C and near-neutral pH levels, drastically reducing energy consumption and equipment stress compared to high-pressure chemical reactors. The enzymatic route exhibits intrinsic stereoselectivity, consistently delivering products with an enantiomeric excess (ee) of greater than 99 percent, thereby eliminating the need for downstream chiral resolution and effectively doubling the potential yield compared to traditional resolution methods. By employing whole-cell catalysis or crude enzyme preparations, the process simplifies the production workflow, allowing for cost reduction in pharmaceutical intermediate manufacturing through reduced solvent usage and simplified waste treatment protocols.
Mechanistic Insights into Amine Dehydrogenase Catalyzed Reductive Amination
The core of this technological advancement lies in the specific catalytic mechanism of the amine dehydrogenase (AmDH), which facilitates the direct asymmetric reductive amination of ketones using ammonia as the nitrogen source. The enzyme binds the hydroxyketone substrate, such as 1-hydroxy-2-butanone or 4-hydroxy-2-butanone, within its active site, where it orchestrates the transfer of a hydride ion from the reduced cofactor NADH to the carbonyl carbon while simultaneously incorporating an ammonium ion to form the chiral amine. This concerted mechanism ensures that the hydride attack occurs from a specific face of the planar carbonyl group, dictating the absolute configuration of the resulting alcohol-amine product, whether it be the (S)-configuration for 2-amino-1-butanol or the (R)-configuration for 3-amino-1-butanol depending on the specific enzyme variant employed.
To maintain catalytic turnover, the process integrates a cofactor regeneration system, typically utilizing glucose dehydrogenase (GDH) to oxidize glucose and regenerate NADH from the oxidized NAD+ produced during the amination reaction. This coupling creates a self-sustaining cycle where the expensive cofactor is used in catalytic rather than stoichiometric amounts, significantly lowering the material cost of the reaction. The patent data indicates that optimal activity is achieved within a specific pH range of 8 to 10 and temperatures up to 55°C for thermophilic variants like BsAmDH1, demonstrating the enzyme's robustness under process-relevant conditions. Understanding this mechanistic interplay between the primary amination enzyme and the regeneration system is crucial for R&D teams aiming to scale this technology, as it allows for the precise tuning of substrate feeding rates and cofactor concentrations to maximize space-time yield.
How to Synthesize Chiral Amino Alcohols Efficiently
Implementing this biocatalytic route requires a structured approach to enzyme expression and reaction engineering to ensure consistent quality and high throughput. The patent provides a comprehensive framework for generating the necessary biocatalysts, starting with the cloning of amine dehydrogenase genes into expression vectors like pET-24a and transforming them into host cells such as E. coli BL21(DE3). Following induction with IPTG, the resulting recombinant cells can be used directly as whole-cell catalysts or processed into crude enzyme powders, offering flexibility in how the biocatalyst is deployed in the reactor. The detailed standardized synthesis steps see the guide below outline the precise conditions for maximizing conversion rates and maintaining enzyme stability throughout the production campaign.
- Construct recombinant vectors (e.g., pET-24a) containing the gene encoding the specific amine dehydrogenase (such as BsAmDH1 or EsAmDH) and transform into E. coli BL21(DE3).
- Induce protein expression using IPTG at optimized temperatures (20-30°C) to obtain whole cells or purified enzyme preparations.
- Perform asymmetric reductive amination in a buffer system (pH 8.5-9.0) containing the hydroxyketone substrate, ammonium source, and a coenzyme regeneration system (GDH/Glucose).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond simple yield improvements. The shift from chemical to biological synthesis fundamentally alters the cost structure of producing chiral amino alcohols by removing the dependency on volatile organic solvents and precious metal catalysts, which are subject to significant market price fluctuations and supply chain disruptions. By utilizing renewable feedstocks like glucose for cofactor regeneration and operating in aqueous buffers, the process aligns with green chemistry principles, potentially reducing the environmental compliance costs associated with hazardous waste disposal and solvent recovery systems.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and transition metal catalysts leads to substantial cost savings in raw material procurement. Since the enzymatic process achieves high stereoselectivity inherently, the loss of material associated with discarding the unwanted enantiomer during resolution is completely avoided, effectively improving the overall mass balance and reducing the cost per kilogram of the final active pharmaceutical ingredient. Furthermore, the ability to use whole cells as catalysts negates the need for costly enzyme purification steps, allowing for a more economical deployment of biocatalytic power in large-scale reactors.
- Enhanced Supply Chain Reliability: Relying on fermentation-derived enzymes and readily available hydroxyketone substrates diversifies the supply base away from specialized chemical intermediates that may have single-source bottlenecks. The robustness of the described amine dehydrogenases, particularly those from thermophilic sources, ensures consistent performance even under varying operational conditions, reducing the risk of batch failures that can disrupt downstream drug manufacturing schedules. This reliability is critical for maintaining continuous production lines and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed for scalability, with data supporting high substrate loading concentrations up to 100 mmol/L and the use of high-density whole-cell suspensions, facilitating the transition from laboratory benchtop to commercial tonnage production. The aqueous nature of the reaction mixture simplifies downstream processing and wastewater treatment, as it avoids the generation of halogenated solvent waste streams common in traditional chemical synthesis. This environmental advantage not only reduces disposal costs but also future-proofs the manufacturing site against increasingly stringent global environmental regulations regarding volatile organic compound emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amine dehydrogenase technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this synthesis route for their own supply chains. Understanding these specifics helps in assessing the feasibility of integrating this biocatalytic method into existing manufacturing infrastructure.
Q: What are the key advantages of using amine dehydrogenase over traditional chemical synthesis for chiral amino alcohols?
A: The biocatalytic method described in patent CN112779232B offers superior stereoselectivity (>99% ee) under mild reaction conditions (20-70°C), eliminating the need for high-pressure hydrogenation and expensive chiral resolving agents required in conventional chemical routes.
Q: Which specific enzymes demonstrated the highest conversion rates for (S)-2-amino-1-butanol synthesis?
A: Among the screened library, the amine dehydrogenase BsAmDH1 derived from Bacillus stearothermophilus showed the best performance, achieving a conversion rate of 99% for the reduction of 1-hydroxy-2-butanone to (S)-2-amino-1-butanol.
Q: How is the cofactor NADH regenerated in this industrial process?
A: The process utilizes a coupled enzyme system where glucose dehydrogenase (GDH) regenerates NADH from NAD+ using glucose as a sacrificial electron donor, ensuring the reaction proceeds efficiently without the need for stoichiometric amounts of expensive cofactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amino Alcohol Supplier
As the demand for high-purity chiral intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge biocatalytic technologies backed by rigorous quality control. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and compliant with international standards. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the >99% ee and high conversion rates promised by this novel enzymatic route, guaranteeing that every batch meets the exacting requirements of the global pharmaceutical market.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this efficient and sustainable biocatalytic process into your supply chain strategy.
