Advanced Synthesis of Selexipag Intermediates: A Technical Breakdown for Commercial Scale-Up
Advanced Synthesis of Selexipag Intermediates: A Technical Breakdown for Commercial Scale-Up
The pharmaceutical landscape for pulmonary arterial hypertension treatments relies heavily on the efficient production of complex heterocyclic intermediates. Patent CN113214169B introduces a transformative synthetic methodology for producing 4-[(5,6-diphenylpyrazin-2-yl)(isopropyl)amino]-1-butanol, a critical precursor structurally aligned with the active pharmaceutical ingredient Selexipag. This patent addresses long-standing inefficiencies in heterocyclic chemistry by replacing harsh halogenation protocols with a mild, lithium bromide-catalyzed cyclization strategy. For R&D directors and supply chain managers, this represents a pivotal shift towards greener, more cost-effective manufacturing paradigms that do not compromise on purity or structural integrity. The ability to synthesize such complex pyrazine derivatives under ambient to moderate thermal conditions suggests a significant reduction in operational expenditure and safety risks.
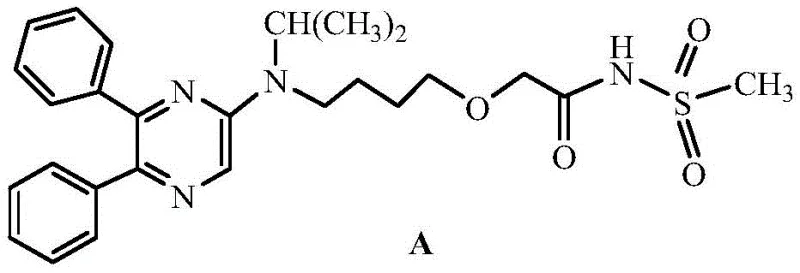
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 5,6-diphenylpyrazine core has been plagued by aggressive reaction conditions and expensive starting materials. Traditional routes, as illustrated in prior art, often commence with 2,3-diphenylpyrazine, necessitating a chlorination step using phosphorus oxychloride (POCl3). This reagent is not only highly corrosive to standard stainless steel reactor vessels, leading to frequent equipment downtime and replacement, but it also generates substantial amounts of acidic waste that require costly neutralization and disposal procedures. Furthermore, subsequent nucleophilic substitution reactions typically demand extreme thermal energy, often exceeding 190°C, to overcome the low reactivity of the chloro-pyrazine intermediate. These high-energy processes result in poor atom economy, significant formation of thermal degradation byproducts, and a reliance on scarce, high-cost raw materials that strain supply chain continuity.
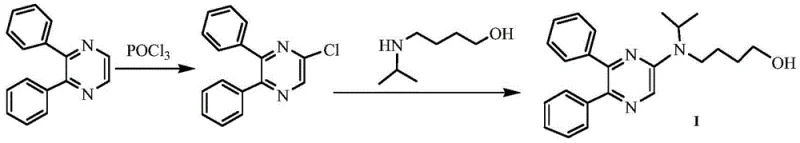
The Novel Approach
In stark contrast, the methodology disclosed in CN113214169B circumvents these bottlenecks by constructing the pyrazine ring de novo from 1,2-diphenyloxirane and 2-aminoacetamidine hydrobromide. This innovative pathway avoids the need for pre-functionalized pyrazine starting materials entirely. The new route operates under significantly milder thermal profiles, with key steps proceeding effectively between 5°C and 80°C, thereby eliminating the energy intensity associated with legacy methods. By shifting the synthetic logic from functional group manipulation of an existing ring to a constructive cyclization event, the process achieves superior yield consistency and simplifies the purification workflow. This strategic pivot not only enhances the safety profile of the manufacturing plant but also opens up access to cheaper, commodity-grade starting materials that are more readily available in the global chemical market.
Mechanistic Insights into Lithium Bromide-Catalyzed Cyclization
The cornerstone of this improved synthesis lies in the first step: the condensation of 1,2-diphenyloxirane with 2-aminoacetamidine hydrobromide. The inclusion of lithium bromide as a catalyst is a subtle yet powerful mechanistic enhancement. Lithium cations act as Lewis acids, coordinating with the oxygen atom of the epoxide ring to increase its electrophilicity, thereby facilitating nucleophilic attack by the amidine nitrogen. This coordination lowers the activation energy required for ring opening, allowing the reaction to initiate at near-ambient temperatures (5-30°C). Following the initial ring opening, an intramolecular cyclization occurs to aromatize the pyrazine system. The use of molecular sieves in this step is critical for water management; by sequestering the water produced during condensation, the equilibrium is driven decisively toward product formation, minimizing hydrolysis side reactions and ensuring high conversion rates without the need for excessive reagent loading.
Subsequent functionalization involves two distinct alkylation events that are meticulously controlled to prevent over-alkylation or polymerization. The first alkylation utilizes 4-bromo-1-butanol to attach the hydroxybutyl chain to the pyrazine nitrogen. The choice of acetonitrile as a solvent here provides an optimal balance of polarity to dissolve the ionic intermediates while maintaining a boiling point that allows for reflux at 70-80°C, sufficient to drive the SN2 reaction without degrading the sensitive alcohol moiety. The final N-isopropylation step employs sodium acetate as a mild base in DMF. This specific base selection is crucial; stronger bases like sodium hydride could induce elimination reactions on the butyl chain or deprotonate the pyrazine ring undesirably. Sodium acetate provides just enough basicity to deprotonate the secondary amine for nucleophilic attack on bromoisopropane, ensuring high regioselectivity and preserving the integrity of the final intermediate structure.
How to Synthesize 4-[(5,6-Diphenylpyrazin-2-yl)(isopropyl)amino]-1-butanol Efficiently
Implementing this synthesis requires precise control over stoichiometry and thermal gradients to maximize the benefits of the novel catalytic system. The process is divided into three telescoped or isolated stages, beginning with the catalytic cyclization in 1,4-dioxane, followed by workup and subsequent alkylation in acetonitrile and DMF. Operators must pay close attention to the addition rate of the epoxide substrate to manage the exotherm during the initial ring-opening phase. Detailed standard operating procedures regarding solvent drying, inert gas protection, and crystallization parameters are essential for reproducing the high yields reported in the patent data. The following guide outlines the critical operational phases derived from the exemplary embodiments.
- React 1,2-diphenyloxirane with 2-aminoacetamidine hydrobromide using a lithium bromide catalyst in 1,4-dioxane to form the pyrazine amine core.
- Perform nucleophilic substitution on the pyrazine amine with 4-bromo-1-butanol using a carbonate base in acetonitrile.
- Complete the synthesis via N-alkylation with bromoisopropane using sodium acetate in DMF under mild thermal conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route offers tangible strategic advantages beyond mere technical feasibility. The primary value proposition lies in the decoupling of production from volatile, high-cost specialty chemical markets. By utilizing 1,2-diphenyloxirane, a commodity chemical, instead of custom-synthesized chloropyrazines, manufacturers can secure raw material supply contracts with greater stability and lower price volatility. Furthermore, the elimination of phosphorus oxychloride removes a significant logistical burden; this reagent often requires specialized transport and storage due to its hazardous nature, and its removal simplifies regulatory compliance and reduces insurance premiums associated with chemical handling. The overall process intensification leads to shorter batch cycles, allowing for increased throughput within existing facility footprints.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the reaction sequence and the avoidance of expensive reagents. Traditional methods often require stoichiometric excesses of amines and harsh halogenating agents, which inflate the bill of materials. In this new route, the use of catalytic amounts of lithium salts and mild acetate bases significantly lowers the direct material costs. Additionally, the mild thermal conditions translate to reduced utility consumption for heating and cooling, as the process does not require maintaining reactors at extreme temperatures for extended periods. The higher crude purity achieved through controlled kinetics also reduces the load on downstream purification units, lowering solvent usage and waste treatment expenses.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. 1,2-Diphenyloxirane and 2-aminoacetamidine hydrobromide are produced by multiple global suppliers, mitigating the risk of single-source dependency that often plagues complex heterocyclic precursors. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures and ensuring consistent delivery schedules to downstream API manufacturers. This reliability is critical for maintaining continuous production lines for life-saving pulmonary hypertension medications, where interruptions can have severe clinical consequences.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns perfectly with modern green chemistry principles. The absence of heavy metal catalysts and corrosive mineral acids simplifies the effluent profile, making wastewater treatment more straightforward and less costly. The process is inherently safer, operating at near-atmospheric pressures and moderate temperatures, which reduces the engineering controls required for scale-up. This safety margin allows for larger batch sizes in standard glass-lined or stainless steel reactors without the need for specialized high-pressure autoclaves, facilitating a smoother transition from pilot plant to commercial metric-ton scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic pathway. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, focusing on the practical implications for industrial adoption. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: How does the new synthetic route improve safety compared to traditional methods?
A: The novel route eliminates the use of phosphorus oxychloride (POCl3), a highly corrosive reagent used in conventional chlorination steps, thereby significantly reducing equipment maintenance costs and safety hazards associated with hazardous waste disposal.
Q: What are the key cost drivers reduced in this manufacturing process?
A: By utilizing readily available 1,2-diphenyloxirane instead of expensive 2,3-diphenylpyrazine derivatives and avoiding high-temperature reactions (200°C), the process drastically lowers raw material procurement costs and energy consumption.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (5-80°C) and utilize common polar organic solvents like acetonitrile and DMF, making the protocol highly scalable and compatible with standard stainless steel reactor infrastructure without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selexipag Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent license; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand the stringent purity specifications required for pulmonary hypertension drug intermediates and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee batch-to-batch consistency. Our facility is designed to handle the specific solvent systems and thermal profiles required by this novel synthesis, ensuring that your supply chain remains uninterrupted and compliant with global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. We encourage potential partners to contact us directly to request specific COA data from our pilot batches and to discuss detailed route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-purity intermediates backed by a team dedicated to scientific excellence and operational efficiency.
