Advanced Manufacturing of 2,5-Dichlorophenol for Scalable Dicamba Production
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN106458809B introduces a transformative methodology for the preparation of 2,5-dihalophenols, specifically targeting the production of 2,5-dichlorophenol, a pivotal precursor for the herbicide Dicamba. This technology addresses long-standing inefficiencies in traditional diazonium salt hydrolysis by leveraging high-temperature reaction conditions coupled with continuous steam distillation. By shifting from conventional batch processing to a more dynamic thermal regime, the process achieves exceptional purity and yield metrics that are essential for modern industrial scale-up. For procurement leaders and R&D directors seeking a reliable agrochemical intermediate supplier, understanding this technological leap is crucial for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-dihalophenols from corresponding anilines has been plagued by thermodynamic and kinetic limitations inherent to low-temperature batch processes. Conventional techniques typically involve heating diazonium salts in dilute aqueous acid solutions at temperatures not exceeding 140°C. Under these suboptimal conditions, the highly reactive diazonium intermediates are prone to forming insoluble resinous by-products, which drastically reduce the isolation yield of the desired phenol to approximately 70%. Furthermore, the batch nature of these legacy methods generates significant volumes of acidic wastewater, creating substantial environmental disposal burdens and increasing the overall cost of goods sold. The inability to continuously remove the product from the reactive medium allows for secondary decomposition reactions, further compromising the economic viability of large-scale manufacturing operations.
The Novel Approach
In stark contrast, the methodology disclosed in CN106458809B revolutionizes this transformation by operating at significantly elevated temperatures ranging from 140°C to 250°C within a highly concentrated mineral acid medium. The core innovation lies in maintaining an inorganic acid concentration of at least 60%, preferably around 72%, which suppresses unwanted side reactions while facilitating rapid hydrolysis. Crucially, the process integrates continuous steam distillation to immediately withdraw the 2,5-dihalophenol product from the reaction zone as it forms. This continuous removal prevents the product from undergoing further degradation or resinification, thereby pushing yields to unprecedented levels, experimentally demonstrated up to 98.8%. This shift not only enhances material efficiency but also streamlines the downstream purification workflow, offering a robust solution for cost reduction in herbicide manufacturing.
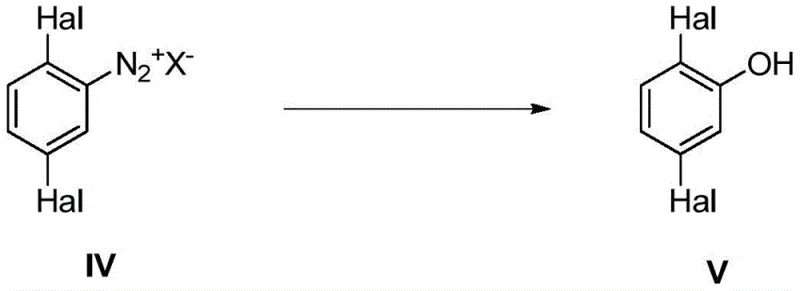
Mechanistic Insights into High-Temperature Diazonium Hydrolysis
The mechanistic advantage of this process stems from the precise control of the reaction medium's acidity and thermal energy. In standard dilute acid conditions, the nucleophilic attack by water on the diazonium cation competes with coupling reactions that lead to tars. However, by utilizing concentrated sulfuric acid (H2SO4), the activity of water is modulated, and the bisulfate anion (HSO4-) acts as a non-nucleophilic counterion that stabilizes the diazonium species until the activation energy for hydrolysis is met. Operating at temperatures between 170°C and 180°C provides the necessary kinetic energy to overcome the activation barrier rapidly, ensuring that the hydrolysis pathway dominates over decomposition pathways. This high-energy environment, when paired with the immediate physical removal of the organic product via steam, creates a unidirectional flow towards the desired phenol, effectively locking in high conversion rates.
Furthermore, the impurity profile is significantly managed through the continuous extraction mechanism. In traditional batch reactors, the accumulation of phenolic products can lead to electrophilic aromatic substitution reactions, generating poly-halogenated impurities or azo-coupling derivatives. The steam distillation component of this novel process ensures that the residence time of the 2,5-dichlorophenol in the hot acidic zone is minimized. As the volatile phenol co-distills with steam, it enters a cooler separation phase where it condenses and separates from the aqueous acid layer. This physical separation not only protects the product integrity but also allows for the direct recycling of the aqueous acid phase back into the reactor, creating a closed-loop system that minimizes waste generation and maximizes atom economy.
How to Synthesize 2,5-Dichlorophenol Efficiently
The implementation of this high-yield synthesis route requires careful attention to the preparation of the diazonium salt precursor and the management of the hydrolysis reactor parameters. The process begins with the diazotization of 2,5-dichloroaniline in sulfuric acid using nitrosyl sulfuric acid, followed by the critical high-temperature hydrolysis step. To ensure reproducibility and safety at an industrial scale, operators must strictly adhere to the specified acid concentrations and temperature ramps while maintaining the steam flow rate sufficient to carry over the product. The detailed standardized synthesis steps, including specific molar ratios and equipment configurations required to replicate the patent's success, are outlined in the guide below.
- Diazotize 2,5-dihaloaniline in concentrated sulfuric acid (approx. 60-65%) using nitrosyl sulfuric acid at 50-60°C to form the diazonium salt.
- Heat the reaction mixture to between 140°C and 250°C in the presence of mineral acid with a concentration of at least 60%.
- Continuously remove the resulting 2,5-dihalophenol product from the reaction system using steam distillation to prevent by-product formation and achieve yields exceeding 95%.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain executives and procurement managers, the adoption of this advanced synthesis protocol offers profound strategic benefits beyond mere chemical yield. The ability to recycle the mineral acid medium directly translates to a substantial reduction in raw material consumption and waste treatment costs. Unlike traditional methods that generate vast quantities of dilute acidic effluent requiring neutralization, this process allows for the recovery and reuse of concentrated sulfuric acid, significantly lowering the environmental footprint and associated compliance costs. Moreover, the continuous nature of the steam distillation setup facilitates a smoother production flow, reducing batch-to-batch variability and enhancing the predictability of delivery schedules for high-purity agrochemical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps and the drastic reduction in waste disposal fees contribute to a leaner cost structure. By avoiding the formation of insoluble resins, the process minimizes the loss of valuable starting materials, ensuring that a higher percentage of the input 2,5-dichloroaniline is converted into saleable product. This efficiency gain allows manufacturers to offer more competitive pricing without compromising on quality margins, providing a distinct economic advantage in the volatile agrochemical market.
- Enhanced Supply Chain Reliability: The robustness of the high-temperature hydrolysis method ensures consistent output quality, which is critical for downstream customers producing formulated herbicides. The process relies on readily available starting materials like 1,4-dichlorobenzene and common mineral acids, mitigating the risk of supply disruptions associated with exotic reagents. This stability enables suppliers to maintain steady inventory levels and meet urgent procurement demands, fostering stronger long-term partnerships with global agrochemical companies.
- Scalability and Environmental Compliance: Designed with industrial scalability in mind, the continuous steam distillation setup can be easily expanded from pilot plant to multi-ton production capacities. The closed-loop water recycling system significantly reduces fresh water consumption, aligning with increasingly stringent global environmental regulations. This proactive approach to sustainability not only future-proofs the manufacturing facility against regulatory changes but also appeals to environmentally conscious stakeholders and end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and preferred embodiments described in the patent documentation, providing clarity on process parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into existing production lines.
Q: How does the new process improve yield compared to conventional methods?
A: Conventional methods typically operate below 140°C with dilute acid, yielding around 70% due to resin formation. The patented process operates at 140-250°C with >60% acid concentration and continuous steam distillation, achieving yields up to 98.8% by minimizing side reactions.
Q: Can the mineral acid be recycled in this manufacturing process?
A: Yes, the process is designed to allow for the recycling of sulfuric acid. The aqueous phase separated after steam distillation can be reused, and the acid from the hydrolysis step can be recycled back to the diazotization step, significantly reducing waste and raw material costs.
Q: What represents the optimal temperature range for the hydrolysis step?
A: While the process functions between 140°C and 250°C, the preferred range is approximately 150°C to 190°C, with a most preferred range of 170°C to 180°C. Higher temperatures within this window correlate with improved reaction kinetics and higher product yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Dichlorophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy of final agrochemical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated high-temperature hydrolysis process described in CN106458809B can be seamlessly translated into reliable manufacturing output. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2,5-dichlorophenol meets the exacting standards required for Dicamba synthesis, providing our partners with peace of mind regarding product consistency and regulatory compliance.
We invite you to collaborate with us to optimize your supply chain for herbicide production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this advanced process can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey towards more efficient and sustainable agrochemical manufacturing.
