Advanced Solvent-Free Synthesis of Prosulfocarb: Ensuring High Purity and Commercial Scalability
The global demand for high-efficacy herbicides continues to drive innovation in the synthesis of key agrochemical intermediates, with prosulfocarb standing out as a critical selective systemic herbicide for soil treatment. A recent breakthrough detailed in patent CN114685334A introduces a transformative preparation method that addresses long-standing challenges regarding impurity profiles and process safety. This novel technique achieves a product purity exceeding 98% and a yield greater than 92%, while critically suppressing the content of dibenzyl disulfide, a stubborn impurity, to levels lower than 0.04%. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this development represents a significant leap forward, ensuring compliance with stringent government registration limits which typically cap this impurity at 0.20%. By fundamentally altering the reaction environment to exclude water and organic solvents during the initial carbonylation phase, the process not only enhances chemical selectivity but also streamlines the downstream purification workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of prosulfocarb has been plagued by complex, hazardous, and inefficient synthetic routes that struggle to meet modern purity standards. Early methodologies, such as those described in US4740623, relied on a dangerous four-step sequence involving the generation of hydrogen sulfide from concentrated hydrochloric acid and sodium hydrosulfide, followed by reaction with phosgene. This approach presents severe safety risks due to the toxicity of phosgene and hydrogen sulfide, alongside significant environmental liabilities associated with handling highly corrosive acids. Furthermore, alternative aqueous-based routes disclosed in patents like CN108997181A and EP0697402A1 have failed to adequately control impurity formation. These methods typically involve introducing carbonyl sulfide into mixtures containing substantial amounts of water, ranging from 10% to over 40% by mass. The presence of water promotes the oxidative dimerization of thiol intermediates, leading to dibenzyl disulfide contents between 0.25% and 0.80%, which far exceeds the regulatory threshold of 0.20% required for commercial registration and sale.
The Novel Approach
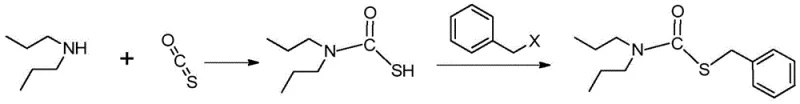
In stark contrast to these legacy processes, the method outlined in CN114685334A employs a sophisticated solvent-free strategy that fundamentally changes the reaction kinetics and thermodynamics. The core innovation lies in reacting di-n-propylamine directly with carbonyl sulfide in the absence of any water or organic diluents during the first step. This anhydrous environment effectively suppresses the side reactions responsible for generating dibenzyl disulfide. Following the formation of the dipropyl amino thioformic acid S-acid intermediate, benzyl halide is introduced under mild thermal conditions to complete the alkylation. This streamlined two-step sequence eliminates the need for toxic phosgene reagents and drastically reduces reaction times compared to the multi-hour drip additions required in aqueous systems. The result is a robust manufacturing protocol that delivers consistent quality with impurity levels consistently maintained below 0.04%, providing a clear competitive advantage for cost reduction in herbicide manufacturing.
Mechanistic Insights into Solvent-Free Thiocarbamate Formation
The success of this novel synthesis route can be attributed to the precise control of nucleophilic attack and the minimization of oxidative degradation pathways. In the initial step, di-n-propylamine acts as a potent nucleophile, attacking the electrophilic carbon of carbonyl sulfide to form the dipropyl amino thioformic acid S-acid intermediate. By strictly excluding water, the system prevents the hydrolysis of this sensitive intermediate and, more importantly, inhibits the formation of free thiol species that are prone to oxidation. In traditional aqueous media, the equilibrium shifts towards the formation of mercaptans, which readily oxidize in the presence of air or dissolved oxygen to form disulfides. The absence of a polar protic solvent like water in the new method ensures that the thioformic acid species remains stable and available for the subsequent alkylation step. This mechanistic clarity allows for the use of stoichiometric ratios of di-n-propylamine to carbonyl sulfide between 2.0:1 and 2.5:1, optimizing the conversion efficiency while maintaining a clean reaction profile.
Furthermore, the alkylation step utilizes benzyl chloride at controlled temperatures between 25°C and 30°C, which is critical for managing the exothermic nature of the substitution reaction. The moderate temperature range prevents thermal degradation of the product while ensuring sufficient energy for the sulfur atom to displace the chloride leaving group efficiently. Post-reaction processing involves a carefully designed workup where water is added only after the main synthesis is complete, followed by acidification to pH 1-2. This specific pH control facilitates the separation of the organic phase containing the crude prosulfocarb from the aqueous layer containing amine salts. The subsequent washing of the organic phase at elevated temperatures (65-75°C) further purifies the product by removing residual acidic impurities and unreacted starting materials. This rigorous control over every stage of the mechanism ensures that the final distilled product meets the high-purity prosulfocarb specifications required by top-tier agrochemical companies.
How to Synthesize Prosulfocarb Efficiently
The implementation of this advanced synthesis route requires strict adherence to the operational parameters defined in the patent to ensure reproducibility and safety at scale. The process begins with the charging of high-purity di-n-propylamine into a reactor equipped with efficient cooling and gas introduction systems. It is imperative to maintain the temperature between 10°C and 20°C during the introduction of carbonyl sulfide gas to manage the exotherm and prevent the loss of volatile reagents. Once the carbonylation is complete, the system is gently warmed to facilitate the addition of benzyl chloride. Detailed standardized operating procedures regarding addition rates, stirring speeds, and quenching protocols are essential for transferring this laboratory-scale success to commercial production. For a comprehensive breakdown of the exact experimental conditions and equipment setups, please refer to the technical guide below.
- Charge di-n-propylamine into a reactor and cool to 10-20°C, then introduce carbonyl sulfide gas over 45-60 minutes without adding water or organic solvents.
- Warm the reaction mixture to 25-30°C and add benzyl chloride dropwise over 1 hour, maintaining the temperature for an additional 3 hours to complete the alkylation.
- Quench the reaction with water, acidify to pH 1-2 at 45-50°C, separate the organic phase, wash twice at 65-75°C, and distill under reduced pressure to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology offers profound strategic benefits beyond mere chemical yield. The elimination of water and organic solvents in the primary reaction step translates directly into a drastic simplification of the manufacturing infrastructure. Without the need to handle, store, and dispose of large volumes of organic solvents or treat massive quantities of wastewater containing organic contaminants, the operational expenditure (OPEX) associated with production is significantly reduced. This lean manufacturing approach minimizes the burden on waste treatment facilities, allowing for faster batch turnover and improved facility utilization rates. Moreover, the ability to recover and recycle unreacted di-n-propylamine with high efficiency creates a closed-loop system that maximizes raw material utilization. This circular economy aspect not only lowers the effective cost of goods sold but also insulates the supply chain from volatility in raw material pricing, ensuring a more stable and predictable supply of high-purity agrochemical intermediates.
- Cost Reduction in Manufacturing: The removal of solvent usage and the reduction in wastewater generation lead to substantial cost savings in utility consumption and waste disposal fees. By avoiding the energy-intensive processes required to remove water or recover solvents from dilute streams, the overall energy footprint of the manufacturing process is drastically lowered. Additionally, the high recovery rate of the expensive amine starting material means that less fresh feedstock is required per kilogram of finished product, directly improving the gross margin profile for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The simplified process flow, characterized by fewer unit operations and shorter reaction times, inherently reduces the risk of production bottlenecks and equipment failures. The avoidance of highly toxic reagents like phosgene removes the need for specialized containment systems and complex safety interlocks, making the production line more flexible and easier to maintain. This operational robustness ensures consistent delivery schedules and reduces the likelihood of supply disruptions caused by safety incidents or regulatory inspections related to hazardous chemical handling.
- Scalability and Environmental Compliance: The solvent-free nature of the reaction makes the process exceptionally easy to scale from pilot plant to full commercial production without the engineering challenges associated with solvent management at large volumes. The significant reduction in organic waste liquid aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against tighter emission standards. This environmental stewardship enhances the brand reputation of the supplier and facilitates smoother regulatory approvals in key international markets.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is crucial for stakeholders evaluating its potential impact on their supply chains. The following questions address common inquiries regarding impurity control, raw material efficiency, and safety protocols based on the data provided in the patent documentation. These insights are derived directly from the experimental results and comparative examples found in CN114685334A, offering a transparent view of the technology's capabilities and limitations in an industrial setting.
Q: How does the solvent-free method reduce dibenzyl disulfide impurities?
A: By eliminating water and organic solvents in the initial carbonylation step, the process prevents the oxidative coupling of thiol intermediates, keeping dibenzyl disulfide levels below 0.04% compared to 0.25-0.80% in traditional aqueous methods.
Q: Can unreacted di-n-propylamine be recovered in this process?
A: Yes, the aqueous phase from the workup can be basified to recover unreacted di-n-propylamine with a recovery rate of 95-96%, significantly reducing raw material costs and waste treatment burdens.
Q: What are the safety advantages over the phosgene-based route?
A: This method avoids the use of highly toxic phosgene and hydrogen sulfide gas required in older four-step syntheses, utilizing safer carbonyl sulfide and benzyl chloride under mild temperatures (10-30°C).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prosulfocarb Supplier
As the agrochemical industry evolves towards greener and more efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands ready to leverage these cutting-edge synthetic innovations for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We are committed to delivering high-purity prosulfocarb that meets stringent purity specifications, utilizing our rigorous QC labs to verify that critical impurities like dibenzyl disulfide remain well below regulatory limits. Our state-of-the-art facilities are designed to handle complex thiocarbamate chemistries safely, providing a secure and reliable source for your herbicide formulation needs.
We invite you to engage with our technical procurement team to discuss how this advanced solvent-free route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical expertise. Let us collaborate to bring superior quality agrochemical intermediates to the market faster and more sustainably.
