Advanced Synthetic Route for Daclatasvir Starting Material: Scalability and Cost Efficiency
Introduction to Next-Generation Daclatasvir Precursor Synthesis
The global demand for direct-acting antivirals (DAAs) targeting Hepatitis C Virus (HCV) continues to drive innovation in pharmaceutical intermediate manufacturing, specifically for potent NS5A inhibitors like Daclatasvir. Patent CN107814789B discloses a breakthrough synthetic methodology that addresses critical bottlenecks in the production of key Daclatasvir starting materials. This technology leverages a streamlined two-step sequence starting from readily available commodity chemicals, offering a robust alternative to legacy processes that often suffer from operational complexity. By focusing on atom economy and process safety, this approach provides a reliable pharmaceutical intermediate supplier pathway that aligns with modern green chemistry principles while maintaining high stereochemical integrity.
The structural complexity of Daclatasvir, characterized by its symmetric bis-imidazole core linked by a biphenyl spacer, necessitates highly precise construction of the chiral imidazole building blocks. Traditional routes often involve multiple protection-deprotection cycles or harsh conditions that compromise yield and optical purity. The methodology outlined in CN107814789B circumvents these issues by employing a mild nucleophilic substitution followed by an efficient cyclization. This strategic design not only simplifies the operational workflow but also enhances the overall economic viability of the manufacturing process, making it an attractive option for large-scale commercial production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Daclatasvir precursors has been plagued by several significant technical hurdles that impede efficient commercial scale-up of complex pharmaceutical intermediates. Existing literature, including various international patents, frequently describes routes that rely on expensive transition metal catalysts or require cryogenic conditions to control regioselectivity. These methods often result in intricate workup procedures involving multiple solvent exchanges, which drastically increase production time and waste generation. Furthermore, the use of hazardous reagents poses safety risks in large-scale reactors, while the formation of difficult-to-remove impurities necessitates costly chromatographic purification steps. Such inefficiencies translate directly into higher manufacturing costs and extended lead times, creating supply chain vulnerabilities for downstream API producers who require consistent, high-volume delivery.
The Novel Approach
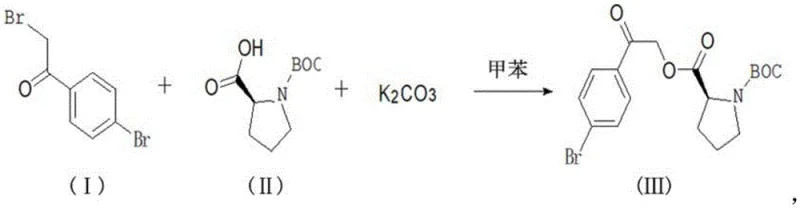
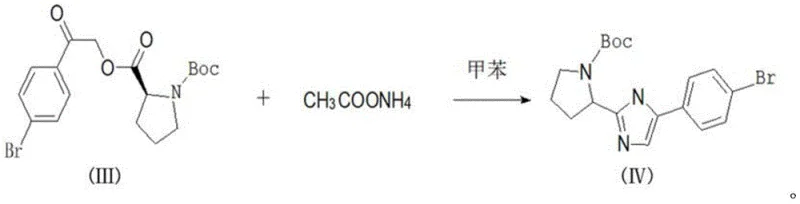
In stark contrast, the novel approach detailed in the patent utilizes a direct condensation strategy that dramatically simplifies the synthetic landscape. The process initiates with the reaction of 2,4'-dibromoacetophenone and L-BOC-proline in the presence of potassium carbonate, utilizing toluene as a singular, versatile solvent. This elimination of exotic reagents in favor of robust, commodity-grade chemicals represents a paradigm shift in cost reduction in API manufacturing. The subsequent cyclization step employs ammonium acetate under reflux conditions, facilitating ring closure without the need for additional activating agents. This streamlined workflow minimizes unit operations, reduces energy consumption, and simplifies equipment requirements, thereby enhancing the overall throughput and reliability of the production line.
Mechanistic Insights into Base-Promoted Substitution and Cyclization
The core of this synthetic innovation lies in the precise control of the nucleophilic substitution mechanism during the formation of Intermediate III. In this step, the carboxylate anion generated from L-BOC-proline by potassium carbonate acts as a potent nucleophile, attacking the alpha-carbon of the bromoacetyl group on the aromatic ring. The choice of toluene as the solvent is critical here, as it provides an optimal polarity balance that solubilizes the organic substrates while allowing the inorganic base to function effectively at the interface. The reaction temperature is meticulously maintained between 25°C and 30°C, a range that is sufficiently energetic to drive the substitution forward yet mild enough to prevent racemization of the chiral center on the proline ring. This preservation of stereochemistry is paramount for the biological activity of the final antiviral agent.
Following the formation of the keto-ester intermediate, the system undergoes a transformative cyclization upon the addition of ammonium acetate. This step involves the condensation of the ketone carbonyl with ammonia (generated in situ) and the adjacent amide nitrogen to form the imidazole ring. The mechanism proceeds through a hemiaminal intermediate which subsequently dehydrates to establish the aromatic heterocycle. The use of ammonium acetate serves a dual purpose: it acts as both the nitrogen source and a buffer to maintain the pH within a range that favors cyclization over hydrolysis. The reaction is conducted under reflux (65°C to 110°C) to provide the necessary activation energy for water elimination, driving the equilibrium towards the desired product. This elegant cascade ensures high conversion rates and minimizes the formation of open-chain byproducts.
How to Synthesize Daclatasvir Starting Material Efficiently
Implementing this synthetic route requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process is designed to be telescoped where possible, reducing the need for isolating unstable intermediates. Operators should ensure rigorous exclusion of moisture during the initial substitution phase to prevent hydrolysis of the activated ketone, although the subsequent aqueous workup is forgiving due to the stability of Intermediate III. The crystallization step is particularly sensitive to cooling rates; slow cooling from the reflux temperature promotes the growth of large, pure crystals that are easy to filter and dry. For a comprehensive breakdown of the specific operational parameters, temperatures, and molar ratios required for GMP-compliant manufacturing, please refer to the standardized synthesis guide below.
- React 2,4'-dibromoacetophenone with L-BOC-proline and potassium carbonate in toluene at 25-30°C to form Intermediate III.
- Add ammonium acetate to the reaction mixture and heat under reflux (65-110°C) overnight to induce cyclization.
- Cool the mixture, wash with brine, dry the toluene phase, and concentrate to crystallize the final pale white solid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic methodology offers profound advantages that extend beyond simple yield improvements. The reliance on universally available starting materials such as 2,4'-dibromoacetophenone and L-BOC-proline mitigates the risk of supply disruptions often associated with specialized custom synthons. This accessibility ensures that purchasing managers can secure raw materials from multiple qualified vendors, fostering a competitive pricing environment and enhancing supply chain resilience. Furthermore, the simplified process flow reduces the burden on quality control laboratories, as fewer intermediates require testing and release, accelerating the overall batch cycle time and enabling faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the elimination of expensive catalysts and the reduction of solvent usage. By utilizing toluene for both the reaction medium and the final recrystallization, the process avoids the capital and operational expenditures associated with solvent swapping and recovery of multiple distinct organic phases. Additionally, the high atom economy of the cyclization step means that a greater proportion of the input mass is converted into valuable product, significantly lowering the cost of goods sold (COGS) per kilogram of active intermediate produced.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply continuity. The tolerance of the process to minor variations in temperature and mixing rates reduces the likelihood of batch failures, which are a major cause of delivery delays in fine chemical manufacturing. Moreover, the use of stable, non-hazardous reagents simplifies logistics and storage requirements, allowing for larger inventory buffers to be held safely. This stability ensures that production schedules can be maintained consistently, providing downstream partners with the predictability they need for their own formulation planning.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to multi-ton commercial production is straightforward due to the absence of exothermic hazards or gas evolution that typically complicate reactor design. The simplified workup procedure generates less aqueous waste compared to traditional acid-base extraction methods, aligning with increasingly stringent environmental regulations. The ability to recycle the toluene solvent further reduces the facility's environmental footprint and waste disposal costs, making this a sustainable choice for long-term manufacturing partnerships focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided are derived directly from the experimental data and process descriptions found in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary cost drivers eliminated in this new synthetic route?
A: This route eliminates expensive transition metal catalysts and complex solvent exchange steps by utilizing toluene for both reaction and crystallization, significantly reducing raw material and waste disposal costs.
Q: How does this method improve supply chain reliability for API manufacturers?
A: By using commercially available, low-cost starting materials like 2,4'-dibromoacetophenone and L-BOC-proline, the method reduces dependency on specialized reagents, ensuring consistent availability and shorter lead times.
Q: What purity levels can be achieved with this crystallization process?
A: The optimized crystallization protocol yields a pale white solid with HPLC purity exceeding 95%, meeting stringent requirements for downstream API synthesis without extensive purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Daclatasvir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving antivirals depends on the reliability and quality of the supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical clients. We are committed to delivering stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in handling chiral intermediates allows us to maintain the optical integrity essential for the efficacy of HCV treatments.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can be tailored to your specific production needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this more efficient methodology. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our Daclatasvir starting materials in your downstream processes, ensuring a seamless transition to a more cost-effective and reliable supply source.
