Revolutionizing Chiral Spiro Oxindole Manufacturing with Efficient Copper Catalysis
Revolutionizing Chiral Spiro Oxindole Manufacturing with Efficient Copper Catalysis
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly those found in bioactive natural products. Patent CN112430236A introduces a groundbreaking preparation method for chiral spiro[pyrrolidone-3,3'-oxindole] derivatives, addressing long-standing inefficiencies in asymmetric synthesis. This technology leverages an asymmetric intramolecular carbon-carbon coupling reaction catalyzed by cuprous species, offering a pathway to high-value intermediates with exceptional enantioselectivity and yield. For R&D directors and procurement specialists, this represents a significant shift away from precious metal dependency towards more sustainable and cost-effective base metal catalysis. The method's ability to construct the core structural units of molecules like Horsfiline under mild conditions underscores its potential for widespread adoption in the manufacturing of analgesic and antibacterial agents.
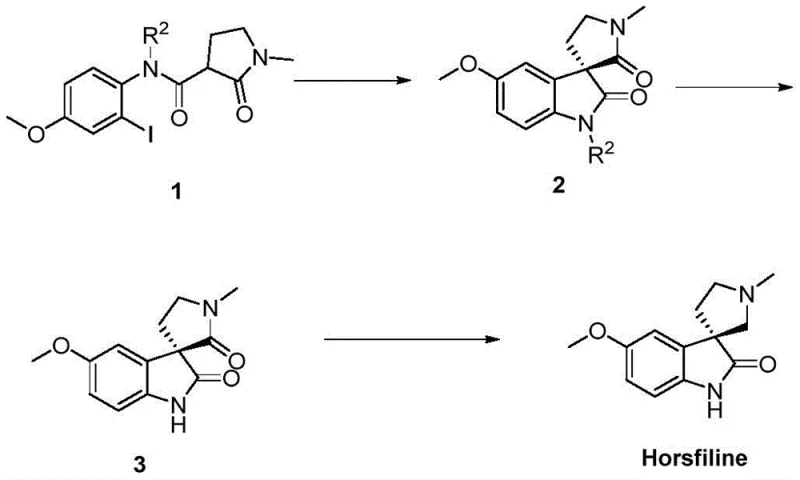
The strategic importance of this patent lies in its direct application to the synthesis of 'dominant structures' in drug design. Chiral spiro oxindoles are renowned for their strong intermolecular forces with biological macromolecules, making them privileged scaffolds in medicinal chemistry. By providing a streamlined route to these cores, the technology enables faster access to diverse chemical libraries for drug discovery. Furthermore, the specific embodiment detailing the synthesis of Horsfiline demonstrates the practical viability of the method for producing complex natural products, bridging the gap between academic methodology and industrial applicability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral spiro[pyrrolidone-3,3'-oxindole] skeletons has been fraught with significant technical and economic hurdles. Prior art, such as the methods reported in J. Am. Chem. Soc. 2005, relied heavily on expensive palladium catalysts to drive asymmetric aromatic nucleophilic substitution and cyclization. These processes often necessitated harsh reaction conditions, including low temperatures and the use of toxic solvent systems like toluene and chloroform mixtures. Additionally, other existing routes, such as those described in Org. Biomol. Chem. 2011, required multi-step sequences involving substrate pretreatment and the use of highly toxic tetrahydrofuran. These factors collectively result in elevated production costs, complex waste management issues, and limited scalability, posing severe challenges for supply chain managers aiming for consistent, high-volume output.
The Novel Approach
In stark contrast, the methodology disclosed in CN112430236A utilizes a cuprous-catalyzed system that dramatically simplifies the synthetic landscape. By employing readily available cuprous iodide in conjunction with a chiral diamine ligand, the reaction proceeds efficiently in acetonitrile at mild temperatures ranging from 15°C to 30°C. This eliminates the need for energy-intensive cooling systems and expensive noble metals. The process is characterized by its operational simplicity, requiring fewer steps and tolerating a broader range of functional groups on the substrate. For procurement teams, this translates to a substantial reduction in raw material costs and a decrease in the complexity of sourcing specialized reagents, thereby enhancing the overall resilience of the supply chain.
Mechanistic Insights into Cu-Catalyzed Asymmetric Cyclization
The core of this technological advancement is the asymmetric intramolecular carbon-carbon coupling reaction mediated by a chiral copper complex. The mechanism involves the activation of the amide substrate by the cuprous catalyst, facilitated by the chiral ligand (1S,2S)-N,N'-dimethyl-1,2-diphenyl-1,2-ethylenediamine. This coordination creates a rigid chiral environment that directs the stereochemical outcome of the cyclization, ensuring high enantiomeric excess (ee) values, often exceeding 90%. The use of sodium hydride as a base further enhances the catalytic activity by generating the necessary nucleophilic species in situ. This precise control over stereochemistry is critical for R&D directors, as it ensures the production of the biologically active enantiomer without the need for difficult downstream resolution processes.
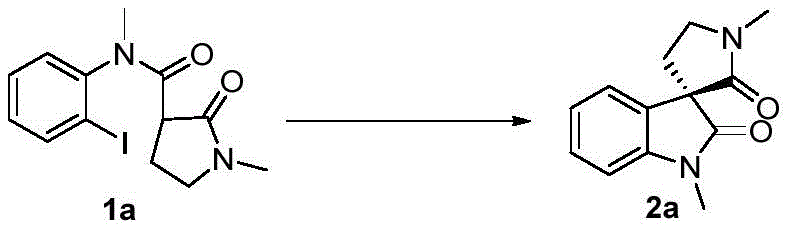
Furthermore, the reaction is conducted under an inert gas atmosphere, which effectively suppresses side reactions such as oxidation of the copper catalyst or the substrate. This protective measure not only improves the chemical yield but also ensures the purity of the final product by minimizing the formation of oxidative impurities. The robustness of this catalytic cycle allows for the accommodation of various substituents on the aromatic ring, including halogens, alkoxy groups, and esters, without significant loss in efficiency. This functional group tolerance is a key attribute for medicinal chemists looking to diversify their lead compounds, as it allows for late-stage functionalization strategies that were previously impractical with more sensitive catalytic systems.
How to Synthesize Chiral Spiro Pyrrolidone-3,3'-oxindole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the dissolution of the amide precursor and base in a dry organic solvent, followed by the sequential addition of the copper catalyst and ligand. The reaction is then allowed to proceed under nitrogen protection until completion. This standardized approach minimizes variability and ensures reproducible results across different batches. For detailed operational parameters and specific stoichiometric ratios, please refer to the structured guide below which encapsulates the critical steps for successful implementation.
- Dissolve the amide substrate and sodium hydride in dry acetonitrile under an inert atmosphere.
- Add cuprous iodide catalyst and the chiral diamine ligand (1S,2S)-N,N'-dimethyl-1,2-diphenyl-1,2-ethylenediamine.
- Stir the reaction mixture at room temperature (15-30°C) to effect asymmetric cyclization, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this copper-catalyzed methodology offers profound commercial benefits that extend beyond mere technical feasibility. For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, the switch from palladium to copper represents a direct and significant saving on catalyst costs. Moreover, the elimination of toxic solvents like tetrahydrofuran and chloroform simplifies environmental compliance and reduces the burden on waste treatment facilities. The mild reaction conditions also lower energy consumption, contributing to a greener and more sustainable production profile that aligns with modern corporate responsibility goals.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive cuprous iodide drastically lowers the bill of materials. Additionally, the simplified one-step cyclization reduces labor hours and equipment usage time compared to multi-step legacy processes. The high yields reported in the patent examples mean less raw material is wasted, further driving down the cost per kilogram of the final API intermediate. This economic efficiency makes the process highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: Copper salts and the specified diamine ligands are commodity chemicals with stable global supply chains, unlike some specialized palladium complexes which can be subject to geopolitical volatility. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or humidity, ensuring consistent batch-to-bquality. This reliability is essential for supply chain heads who need to guarantee uninterrupted delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The use of acetonitrile as a primary solvent and the absence of highly toxic reagents facilitate easier scale-up from gram to ton quantities. The process generates less hazardous waste, simplifying disposal and reducing regulatory hurdles. The ability to operate at near-ambient temperatures reduces the need for specialized cryogenic equipment, allowing the reaction to be run in standard stainless steel reactors, thus lowering capital expenditure for facility upgrades.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their manufacturing portfolios.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium routes?
A: Unlike traditional methods requiring expensive palladium catalysts, toxic solvents like tetrahydrofuran, and strict low-temperature conditions, this novel approach utilizes cheap and readily available cuprous iodide. It operates under mild room temperature conditions in acetonitrile, significantly simplifying the operational complexity and reducing raw material costs while maintaining high enantioselectivity.
Q: Can this synthetic route be applied to the production of natural products like Horsfiline?
A: Yes, the patent explicitly demonstrates the effective application of this chiral spiro pyrrolidone-3,3'-oxindole construction method in the total synthesis of the natural product Horsfiline. The intermediate compounds generated through this asymmetric cyclization serve as critical precursors that can be further converted into bioactive molecules with analgesic properties.
Q: What represents the key challenge in scaling up spiro oxindole synthesis, and how does this patent address it?
A: Scaling up is often hindered by the need for strict anhydrous and oxygen-free environments and complex multi-step sequences found in prior art. This invention addresses these bottlenecks by employing a robust catalytic system that tolerates mild conditions and offers a wide substrate scope, making the transition from laboratory scale to commercial production more feasible and reliable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Pyrrolidone-3,3'-oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes described in CN112430236A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of copper-catalyzed reactions, ensuring stringent purity specifications and rigorous QC labs verify every batch against the highest industry standards. We are committed to delivering high-purity chiral spiro oxindoles that meet the exacting demands of global drug development programs.
We invite you to collaborate with us to leverage this efficient technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the next generation of bioactive therapeutics.
