Advanced Heck Coupling Strategy for High-Purity Acrivastine Precursors and Commercial Scalability
Introduction to Next-Generation Antihistamine Intermediate Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for high-value active pharmaceutical ingredients (APIs) and their precursors, particularly for widely used antihistamines like Acrivastine. Patent CN102311381A introduces a transformative methodology for synthesizing the critical alpha ethyl ester derivative, known structurally as Formula I, which serves as the direct precursor to Acrivastine. This invention addresses long-standing challenges in stereochemical control and process efficiency by leveraging a sophisticated palladium-catalyzed coupling strategy. By shifting away from legacy methodologies that struggle with isomeric purity, this technology offers a pathway to produce the biologically active E,E-isomer with exceptional fidelity. The strategic implementation of a combined catalyst system allows manufacturers to bypass complex purification bottlenecks, thereby aligning synthetic chemistry with the rigorous demands of modern Good Manufacturing Practice (GMP) standards.
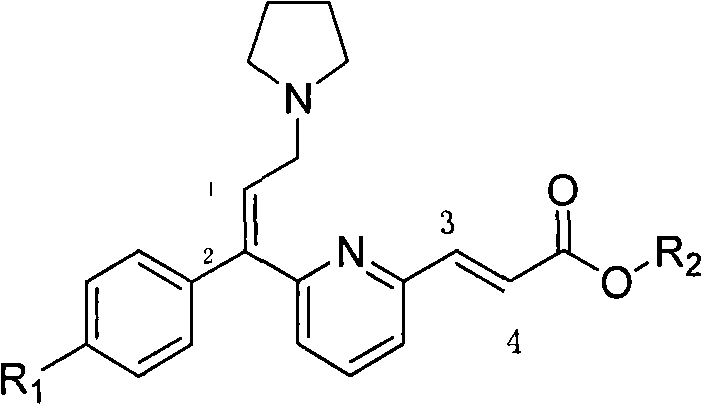
Furthermore, the economic implications of adopting this novel synthetic route are profound for stakeholders across the supply chain. Traditional methods often incur significant costs due to low yields and the necessity for multiple recrystallization steps to remove unwanted Z-isomers. In contrast, the process detailed in this patent demonstrates that high directional selectivity can be achieved without compromising operational simplicity. For R&D directors and procurement managers, this represents a dual opportunity: enhancing the quality profile of the final drug substance while simultaneously reducing the cost of goods sold (COGS). As we delve deeper into the technical specifics, it becomes evident that this patent provides a blueprint for the reliable pharmaceutical intermediates supplier seeking to optimize their portfolio with high-margin, technically superior compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Acrivastine precursors, such as those disclosed in U.S. Pat 4584382, have relied heavily on Wittig reactions involving 3-[6-[(4-fluoroform phenyl) carbonyl]-2-pyridyl] propenoate. While chemically feasible, these conventional pathways suffer from inherent stereochemical deficiencies that plague large-scale manufacturing. The primary drawback is the poor selectivity of the olefination step, which invariably generates a complex mixture of E and Z isomers, often in ratios as unfavorable as 5:6. This lack of control necessitates a downstream purification nightmare involving sequential operations such as ester extraction, alkalization, acid neutralization, and multiple rounds of recrystallization. Not only does this consume vast amounts of energy and solvents, but it also drastically reduces the overall throughput of the manufacturing line. Additionally, some reported methods utilize hazardous extraction agents like methyl tert-butyl ether (MTBE), introducing significant safety risks and environmental compliance burdens that modern facilities strive to eliminate.
The Novel Approach
The methodology presented in CN102311381A fundamentally reengineers the synthetic logic by employing a Heck coupling reaction between a halogenated pyridine derivative (Formula II) and an acrylate ester (Formula III). This transition from Wittig chemistry to palladium-catalyzed cross-coupling is a game-changer for cost reduction in pharmaceutical intermediates manufacturing. The new approach utilizes a unique combined catalyst system that ensures the reaction proceeds with high directional selectivity, favoring the formation of the desired E,E-configuration exclusively. By operating under milder thermal conditions and utilizing a streamlined workup procedure that involves simple cooling, water addition, and filtration, the process eliminates the need for chromatographic separation or extensive recrystallization. This radical simplification of the unit operations not only shortens the production cycle time but also significantly enhances the safety profile of the plant by removing hazardous solvent handling steps.
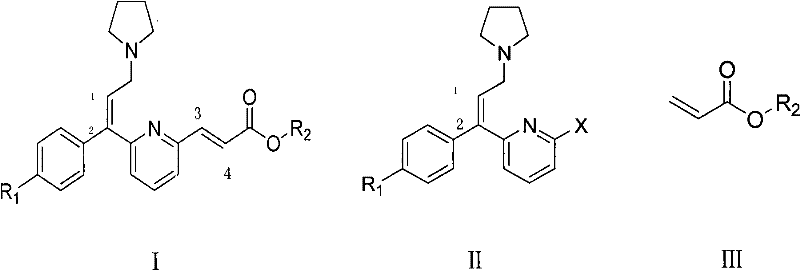
Mechanistic Insights into Combined Catalyst Palladium Coupling
The success of this synthetic route hinges on the intricate interplay within the proprietary combined catalyst system, which consists of triphenylphosphine, a divalent palladium compound, an alkali carbonate base, and a critical haloalkylamine additive. Mechanistically, the divalent palladium species initiates the catalytic cycle through oxidative addition into the carbon-halogen bond of the pyridine substrate. The presence of triphenylphosphine acts as a stabilizing ligand, maintaining the palladium in its active state and preventing the formation of inactive palladium black aggregates. However, the true innovation lies in the inclusion of the haloalkylamine, such as tetrabutyl ammonium chloride or bromobutylamine, which likely functions as a phase-transfer catalyst or a specialized ligand modifier. This component enhances the solubility of the inorganic base in the organic reaction medium and facilitates the deprotonation step necessary for the migratory insertion and subsequent beta-hydride elimination that yields the trans-alkene product.
From an impurity control perspective, this mechanistic precision translates directly into superior product quality. The high specificity of the catalyst system suppresses the formation of the thermodynamic Z-isomer, ensuring that the crude reaction mixture contains less than 1% of unwanted stereoisomers. This level of purity, often reaching 99% as confirmed by HPLC analysis in the patent examples, means that the burden of impurity management is shifted upstream to the reaction design rather than downstream to purification. For quality assurance teams, this predictability is invaluable, as it minimizes the risk of genotoxic impurities or difficult-to-remove byproducts carrying over into the final API. The ability to tune the catalyst loading—ranging from 0.001 to 0.1 molar equivalents—further allows process chemists to balance reaction kinetics with cost efficiency, optimizing the turnover number (TON) of the precious metal catalyst.
How to Synthesize Alpha Ethyl Ester Derivative Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the combined catalyst and the selection of appropriate polar aprotic solvents such as N-methyl-2-pyrrolidone (NMP) or dimethyl sulfoxide (DMSO). The patent outlines a robust protocol where the reactants are heated to temperatures between 90°C and 180°C, allowing the reaction to reach completion within 3 to 10 hours. The detailed standardized synthesis steps below provide a clear roadmap for replicating this high-yield process in a pilot or production setting, ensuring consistent batch-to-batch quality.
- Prepare the reaction mixture by combining Formula II (halogenated pyridine derivative) and Formula III (acrylate ester) in a polar aprotic solvent such as NMP or DMF.
- Introduce the combined catalyst system consisting of triphenylphosphine, a divalent palladium source, alkali carbonate, and a haloalkylamine additive.
- Heat the mixture to 90-180°C for 3-10 hours, then cool, add water to precipitate the product, and isolate via filtration and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of complex purification sequences directly correlates to a reduction in utility consumption, solvent waste disposal costs, and labor hours per kilogram of product. By simplifying the workflow to a single reaction vessel followed by a straightforward filtration, manufacturers can significantly increase their asset utilization rates. This efficiency gain is crucial for maintaining competitiveness in the global market for high-purity pharmaceutical intermediates, where margin compression is a constant challenge. Furthermore, the reliance on readily available starting materials and common industrial solvents mitigates supply chain risks associated with exotic reagents.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic simplification of the downstream processing. Traditional routes require multiple unit operations including extractions and recrystallizations, each adding cost in terms of equipment time, solvent purchase, and waste treatment. By achieving >99% purity directly from the reaction quench, this method effectively removes these costly steps. Additionally, the high yield (>80%) ensures that raw material costs are amortized over a larger quantity of saleable product, driving down the unit cost significantly without the need for speculative percentage claims.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that are sensitive to minor variations or rely on hard-to-source reagents. This Heck coupling method utilizes commodity chemicals like acrylates and halopyridines, which are produced at scale globally, ensuring a stable supply base. The robustness of the reaction conditions, which tolerate a range of temperatures and catalyst loadings, means that production schedules are less likely to be disrupted by batch failures. This reliability allows supply chain planners to operate with leaner inventory buffers while maintaining high service levels to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental liabilities, particularly when using hazardous solvents like MTBE or generating large volumes of aqueous waste from acid/base washes. This new process avoids these pitfalls by using safer polar aprotic solvents and generating a solid product that can be isolated by filtration. The reduction in solvent usage and the elimination of acidic/alkaline waste streams simplify the environmental permitting process and reduce the facility's ecological footprint. This alignment with green chemistry principles future-proofs the manufacturing site against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: How does this Heck coupling method improve upon traditional Wittig reactions for Acrivastine synthesis?
A: Unlike traditional Wittig reactions which often produce difficult-to-separate E/Z isomer mixtures requiring complex extraction and recrystallization, this patented Heck coupling method achieves over 99% selectivity for the desired E,E-isomer, eliminating the need for tedious purification steps.
Q: What represents the key innovation in the catalyst system described in CN102311381A?
A: The core innovation lies in the 'combined catalyst' approach, which synergistically utilizes triphenylphosphine, a divalent palladium compound, and a specific haloalkylamine additive to enhance reaction directionality and yield under milder thermal conditions.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial suitability, featuring simple post-reaction treatment (filtration and drying), avoidance of hazardous extraction solvents like MTBE, and robust yields exceeding 80% with high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acrivastine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Heck coupling technology are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the stereochemical integrity of every batch. Our commitment to quality ensures that the commercial scale-up of complex pharmaceutical intermediates is executed with precision, delivering materials that meet the exacting standards of global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits relevant to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing endeavors.
