Advanced Synthetic Route for Cefpirome Sulfate: Enhancing Yield and Supply Chain Stability
Advanced Synthetic Route for Cefpirome Sulfate: Enhancing Yield and Supply Chain Stability
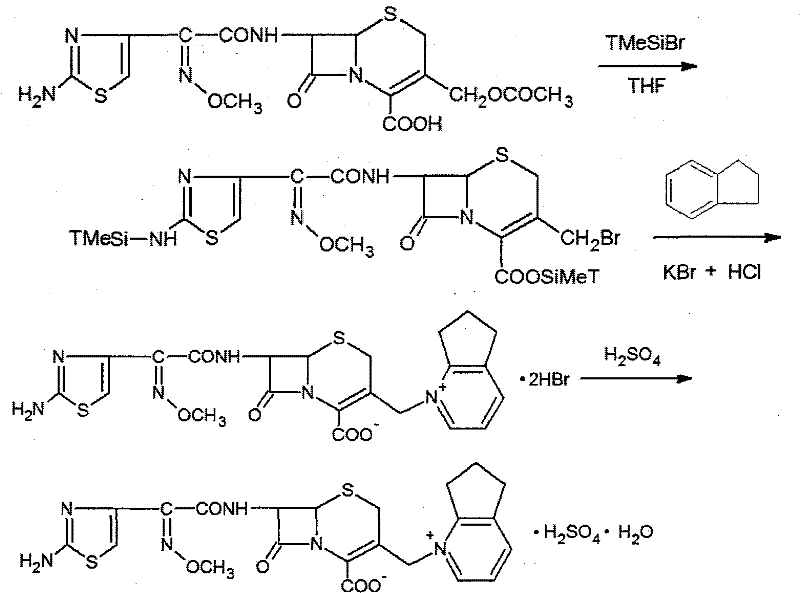
The pharmaceutical landscape for fourth-generation cephalosporins is continuously evolving, driven by the urgent need for broad-spectrum antibiotics capable of overcoming bacterial resistance mechanisms. A pivotal development in this sector is detailed in patent CN101284840B, which discloses a highly efficient synthetic method for producing Cefpirome Sulfate, a zwitterionic antibiotic known commercially as Cefrom. This technical breakthrough addresses critical bottlenecks in traditional manufacturing by utilizing Cefotaxime as a primary starting material and Bromotrimethylsilane (TMSBr) as a specialized protecting agent. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize the production of high-value antibiotic intermediates. The methodology not only simplifies the reaction sequence but also leverages the mature supply chain of Cefotaxime, ensuring that the transition to this new process can be achieved with minimal disruption to existing logistical frameworks while delivering superior product purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefpirome has been plagued by complex multi-step sequences that originate from 7-amino-cephalosporanic acid (7-ACA). Traditional routes often involve an initial acylation followed by a substitution reaction, or vice versa, creating a delicate balance of reactivity that is difficult to maintain on a large scale. One prevalent method utilizes Iodotrimethylsilane as a silylating reagent; while chemically effective, this reagent is prohibitively expensive due to the high cost of iodine precursors, severely impacting the gross margin of the final API. Furthermore, alternative routes that attempt to introduce the quaternary ammonium side chain early in the synthesis often suffer from harsh reaction conditions and low yields, generating significant amounts of hazardous waste. These legacy processes require rigorous purification steps to remove heavy metal residues and organic impurities, adding substantial operational expenditure (OpEx) and extending the overall lead time for batch release, which is a critical pain point for supply chain managers facing tight delivery schedules.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally shifts the paradigm by employing a "substitution-first" approach using readily available Cefotaxime. By reacting Cefotaxime directly with 2,3-cyclopentapyridine in the presence of Bromotrimethylsilane, the process bypasses the need for expensive iodine-based reagents while maintaining high reaction kinetics. Bromotrimethylsilane acts as a superior protecting group compared to chlorosilanes due to its higher activity, yet it remains significantly more cost-effective than its iodo-counterpart. This route allows for the direct formation of Cefpirome dihydrobromide, which is subsequently converted to the sulfate salt through a streamlined ion-exchange process. The simplicity of this approach reduces the number of isolation steps, minimizes solvent consumption, and eliminates the need for complex chromatographic separations typically required in older methodologies, thereby offering a robust pathway for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into TMSBr-Mediated Quaternization
The core chemical transformation in this synthesis relies on the precise activation of the C-3' position of the cephalosporin nucleus. In the presence of anhydrous tetrahydrofuran (THF), Bromotrimethylsilane reacts with the carboxylate groups to form silyl esters, effectively protecting the molecule and increasing its solubility in the organic phase. This silylation step is crucial as it prevents unwanted side reactions at the carboxylic acid moiety during the subsequent nucleophilic attack. The 2,3-cyclopentapyridine then acts as a potent nucleophile, displacing the leaving group at the C-3' position to form the quaternary ammonium structure characteristic of fourth-generation cephalosporins. The use of THF as the solvent is particularly strategic; its ability to dissolve both the organic substrates and the silylating agent ensures a homogeneous reaction environment, which is essential for maximizing yield and minimizing the formation of regio-isomers that could compromise the biological efficacy of the final drug product.
Following the substitution reaction, the process incorporates a sophisticated workup procedure involving the addition of Potassium Bromide and hydrochloric acid. This step serves a dual purpose: it quenches excess silylating reagent and facilitates the precipitation of the Cefpirome dihydrobromide intermediate as a solid. The subsequent conversion to the sulfate salt utilizes a strongly basic anion exchange resin, which selectively replaces bromide ions with sulfate ions without degrading the sensitive beta-lactam ring. This mild ion-exchange mechanism is far superior to direct acidification methods that might induce hydrolysis of the beta-lactam core. The final crystallization, controlled by pH adjustment to 1.3 using sulfuric acid and the addition of cold ethanol, ensures the formation of large, well-defined crystals with high purity, effectively excluding residual solvents and trace impurities that could affect the stability of the API during storage.
How to Synthesize Cefpirome Sulfate Efficiently
Implementing this synthetic route requires strict adherence to anhydrous conditions and temperature controls to ensure optimal conversion rates. The process begins with the preparation of a dry reaction vessel under nitrogen protection, followed by the sequential addition of solvents and reagents to manage exothermic risks. The reaction mixture is subjected to reflux to drive the substitution to completion, after which careful cooling and acidification precipitate the intermediate. For a comprehensive understanding of the specific operating parameters, including exact molar ratios, stirring speeds, and drying temperatures, please refer to the standardized protocol below which details the step-by-step execution derived from the patent embodiments.
- Prepare the reaction system by mixing Cefotaxime and 2,3-cyclopentapyridine in anhydrous THF with Bromotrimethylsilane as a protecting agent under inert atmosphere.
- Execute the substitution reaction via reflux, followed by cooling and treatment with Potassium Bromide and hydrochloric acid to precipitate Cefpirome dihydrobromide.
- Convert the dihydrobromide intermediate to Cefpirome Sulfate using strong base anion exchange resin and sulfuric acid crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound advantages for procurement managers and supply chain heads tasked with securing reliable sources of antibiotic intermediates. The primary driver of value is the substitution of expensive raw materials with cost-effective alternatives that do not compromise quality. By utilizing Cefotaxime, a commodity chemical with a mature and stable global supply chain, manufacturers can mitigate the risks associated with sourcing niche starting materials. Additionally, the replacement of Iodotrimethylsilane with Bromotrimethylsilane results in a direct reduction in raw material costs, as bromine-based reagents are generally more abundant and less volatile in price than iodine derivatives. This strategic sourcing shift allows for more predictable budgeting and protects margins against fluctuations in the halogen market, providing a competitive edge in the pricing of the final API.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers operational expenditures by reducing the number of unit operations required. Traditional methods often necessitate multiple purification stages to remove heavy metal catalysts or complex byproducts, whereas this route utilizes a simple filtration and ion-exchange workflow. The elimination of expensive chromatography steps for bulk purification, combined with the high recovery yield of the intermediate (reported around 84%), means that less raw material is wasted per kilogram of final product. Furthermore, the use of common solvents like THF and ethanol, which are easily recoverable and recyclable, contributes to a lower environmental compliance cost and reduced waste disposal fees, driving down the total cost of ownership for the manufacturing facility.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for life-saving antibiotics, and this route enhances reliability by relying on domestically available and technically mature raw materials. Cefotaxime is produced at a massive scale globally, ensuring that supply disruptions are unlikely to impact production schedules. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring specialized high-pressure or cryogenic setups, further ensures that production can be maintained consistently across different manufacturing sites. This flexibility allows supply chain managers to diversify their vendor base and reduce lead times for high-purity pharmaceutical intermediates, ensuring that downstream formulation teams receive their materials on schedule to meet market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without significant re-engineering. The reaction generates minimal hazardous waste compared to older routes that utilize toxic heavy metals or corrosive acids in excessive quantities. The incorporation of a decolorization step using activated carbon and alumina not only improves product quality but also simplifies the effluent treatment process, as the spent adsorbents are easier to handle and dispose of than liquid chemical waste streams. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, a key metric for modern ESG-compliant supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefpirome Sulfate synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process feasibility and quality control measures. Understanding these details is essential for technical teams evaluating the potential for technology transfer and for procurement officers assessing the long-term viability of this supply source.
Q: Why is Bromotrimethylsilane preferred over Iodotrimethylsilane in this synthesis?
A: Bromotrimethylsilane offers a superior balance of reactivity and cost. While Iodotrimethylsilane is highly active, it is significantly more expensive. Bromotrimethylsilane is more active than Trimethylchlorosilane, ensuring efficient silylation protection without the prohibitive cost of iodine-based reagents, thereby optimizing the overall production economics.
Q: How does this route improve impurity control compared to traditional methods?
A: The process utilizes a specific decolorization step involving activated carbon and alumina chromatography prior to final crystallization. This effectively removes pyrogens, intracellular toxins, and colored impurities. Furthermore, the use of anhydrous THF minimizes side reactions that often generate difficult-to-remove byproducts in aqueous or less pure solvent systems.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route is designed for scalability. It relies on Cefotaxime, a mature and widely available starting material, and employs standard unit operations such as reflux, filtration, and crystallization. The avoidance of exotic catalysts and the use of common solvents like THF facilitate easy technology transfer from pilot plant to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpirome Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global antibiotic market. Our technical team has extensively analyzed the methodology described in patent CN101284840B and possesses the expertise to scale this diverse pathway from 100 kgs to 100 MT/annual commercial production. We are committed to delivering Cefpirome Sulfate with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the highest international pharmacopoeia standards. Our state-of-the-art facilities are equipped to handle the specific solvent systems and ion-exchange processes required by this route, guaranteeing a consistent supply of high-quality intermediates for your API manufacturing needs.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and cost-effective supply chain for your cephalosporin products.
