Advanced Manufacturing of Ethynyl Aniline: A Technical Breakthrough for Oncology Drug Intermediates
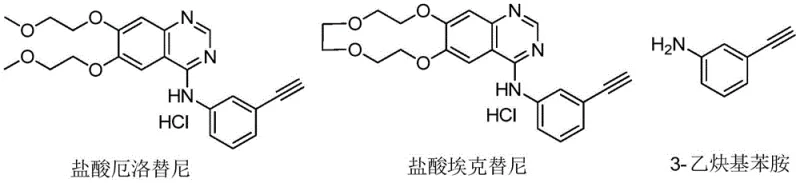
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical oncology intermediates, particularly those required for small molecule targeted therapies. Patent CN112645826B introduces a transformative preparation method for ethynyl aniline, a pivotal building block in the synthesis of life-saving drugs such as erlotinib hydrochloride and icotinib hydrochloride used for lung cancer treatment. This technology addresses the longstanding challenges of high production costs and complex purification associated with traditional alkyne-functionalized aniline derivatives. By leveraging a novel three-step sequence involving radical bromination, elimination, and chemoselective reduction, the process ensures high purity and yield while utilizing readily available starting materials. The strategic design of this pathway not only enhances reaction safety but also aligns with modern green chemistry principles by minimizing hazardous waste generation.
Historically, the synthesis of ethynyl aniline derivatives has relied heavily on transition metal-catalyzed cross-coupling reactions or the use of specialized silyl-protected acetylene reagents. Conventional methods often necessitate the use of expensive palladium or copper catalysts, which introduce significant cost burdens and require rigorous downstream processing to remove trace metal residues to meet pharmaceutical standards. Furthermore, alternative routes involving the degradation of bulky alcohol derivatives or the use of unstable diazo compounds present substantial safety risks and operational complexities on a large scale. These traditional approaches frequently suffer from moderate selectivity, leading to difficult separations and reduced overall throughput, which ultimately impacts the supply chain stability for downstream API manufacturers seeking reliable sources.
In stark contrast, the novel approach detailed in the patent utilizes a pragmatic and economically viable strategy starting from nitroethylbenzene. This method bypasses the need for precious metal catalysts entirely, replacing them with an in-situ generated brominating system composed of alkali metal bromides and bromates in an acidic medium. The subsequent elimination step proceeds under mild alkaline conditions to form the alkyne functionality with high fidelity, avoiding the harsh conditions typical of dehydration reactions. Finally, the use of iron powder in hydrochloric acid for the reduction step provides exceptional chemoselectivity, ensuring the nitro group is converted to an amine without compromising the integrity of the terminal alkyne. This sequence represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
Mechanistic Insights into Radical Bromination and Chemoselective Reduction
The core of this innovative synthesis lies in the efficient generation of the 1,1-dibromo-1-(nitrophenyl)ethane intermediate through a controlled radical mechanism. The reaction employs a mixture of MBr and MBrO3 (where M is Na or K) in the presence of sulfuric acid to generate molecular bromine in situ, which then reacts with the benzylic position of nitroethylbenzene initiated by a radical initiator such as AIBN. This specific bromination protocol offers superior control over reaction selectivity compared to direct bromination, minimizing poly-brominated byproducts and ensuring high conversion rates. The resulting gem-dibromo compound serves as a stable precursor that can be easily isolated and purified, setting the stage for the subsequent elimination reaction which proceeds via an E2 mechanism to install the triple bond.
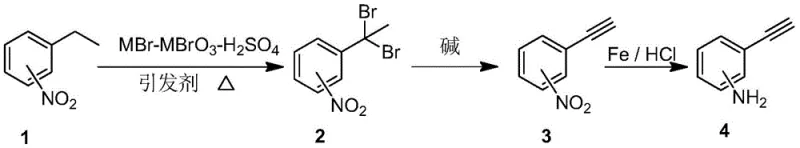
Following the formation of the nitrophenylacetylene intermediate, the final transformation involves the reduction of the nitro group to an amine using an Fe/HCl system. This classical reduction method is particularly advantageous in this context because it exhibits remarkable chemoselectivity, reducing the nitro functionality while leaving the electron-rich carbon-carbon triple bond intact, a common failure point in catalytic hydrogenation methods. The process conditions are optimized to maintain mild temperatures and utilize aqueous workups that facilitate the separation of iron sludge from the organic product. Additionally, the bromide salts generated as byproducts during the elimination step are insoluble in the alcoholic solvent used, allowing for simple filtration and recovery, which can then be recycled back into the first step, thereby closing the material loop and enhancing the overall sustainability of the process.
How to Synthesize Ethynyl Aniline Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for producing high-purity ethynyl aniline suitable for pharmaceutical applications. The procedure begins with the radical bromination of nitroethylbenzene in a chlorinated solvent, followed by a base-mediated elimination in ethanol to generate the alkyne intermediate. The final step involves the reduction of the nitro group using iron powder and hydrochloric acid under reflux conditions. Detailed standardized synthesis steps are provided in the guide below to ensure consistent quality and yield during scale-up operations.
- Perform radical bromination of nitroethylbenzene using MBr-MBrO3-H2SO4 to generate 1,1-dibromo-1-(nitrophenyl)ethane.
- Conduct an elimination reaction under alkaline conditions to convert the dibromo intermediate into nitrophenylacetylene.
- Execute chemoselective reduction of the nitro group using Fe/HCl to yield the final ethynyl aniline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of expensive precious metal catalysts such as palladium removes a significant variable cost component and mitigates the risk associated with volatile metal prices. Furthermore, the reliance on commodity chemicals like nitroethylbenzene and sodium bromide ensures a stable and diversified supply base, reducing the risk of raw material shortages that often plague specialized reagent markets. The simplicity of the equipment requirements, utilizing standard glass-lined reactors and filtration units, allows for rapid deployment in existing manufacturing facilities without the need for capital-intensive infrastructure upgrades.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing high-value catalytic systems with inexpensive stoichiometric reagents that are globally sourced. By avoiding the use of protected acetylene reagents or specialized organometallic coupling partners, the raw material bill of materials is drastically simplified. The ability to recover and recycle bromide salts from the elimination step further contributes to cost efficiency by reducing waste disposal fees and lowering the consumption of fresh brominating agents. This economic model supports a more competitive pricing structure for the final intermediate, enabling downstream partners to optimize their own production costs.
- Enhanced Supply Chain Reliability: Utilizing widely available bulk chemicals ensures that production schedules are not held hostage by the lead times of niche specialty suppliers. The robust nature of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, leading to more consistent batch-to-batch performance. This reliability is critical for maintaining continuous supply lines for essential oncology medications, where interruptions can have severe consequences. The simplified purification steps also reduce the turnaround time between batches, allowing for greater flexibility in responding to market demand fluctuations.
- Scalability and Environmental Compliance: The methodology is inherently designed for industrial scale-up, having been demonstrated effectively from gram to multi-kilogram scales with consistent results. The reduction in hazardous waste generation, particularly through the recycling of bromide byproducts and the avoidance of heavy metal contamination, simplifies regulatory compliance and environmental reporting. This alignment with green chemistry principles not only reduces the environmental footprint but also minimizes the operational costs associated with waste treatment and disposal. The process safety profile is enhanced by avoiding high-pressure hydrogenation or pyrophoric reagents, making it a safer choice for large-scale manufacturing environments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ethynyl aniline synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the Fe/HCl reduction method preferred over catalytic hydrogenation for this synthesis?
A: The Fe/HCl reduction system offers superior chemoselectivity, effectively reducing the nitro group while preserving the sensitive carbon-carbon triple bond, which is often susceptible to over-reduction in catalytic hydrogenation processes.
Q: How does this process address the high cost associated with traditional palladium-catalyzed coupling?
A: By utilizing inexpensive commodity chemicals like nitroethylbenzene and sodium bromide instead of precious metal catalysts and specialized silyl reagents, this method drastically lowers raw material costs and eliminates the need for expensive metal scavenging steps.
Q: What are the environmental benefits of the bromide recycling step in this protocol?
A: The process allows for the recovery of bromide salts during the elimination step, which can be reused in the initial bromination reaction, significantly reducing hazardous waste discharge and improving the overall atom economy of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethynyl Aniline Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, leveraging advanced technologies like the one described in CN112645826B to deliver superior value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the rigorous demands of the international pharmaceutical market. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of ethynyl aniline meets the highest quality standards required for API synthesis. Our technical team is adept at optimizing reaction parameters to maximize yield and minimize impurities, ensuring a consistent supply of this critical oncology intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this metal-free protocol. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to enhance the efficiency and sustainability of your drug development pipeline with our reliable supply of high-quality pharmaceutical intermediates.
