Scalable Production of High-Purity Cis-D-Hydroxyproline via Dynamic Racemization Technology
Scalable Production of High-Purity Cis-D-Hydroxyproline via Dynamic Racemization Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce chiral amino acid derivatives, which serve as critical building blocks for complex drug molecules. A recent technological breakthrough detailed in patent CN112851560A introduces a robust and economically viable method for the preparation of cis-D-hydroxyproline, a valuable intermediate in the synthesis of various bioactive compounds. This innovation addresses long-standing challenges in stereochemical control and process economics by leveraging a dynamic racemization strategy coupled with a sophisticated mother liquor recycling system. Unlike traditional methods that rely on expensive protecting groups or hazardous hydrogenation processes, this approach utilizes readily available trans-L-hydroxyproline as a starting material, converting it directly through an acid-catalyzed isomerization mechanism. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates while drastically reducing the cost of goods sold through improved yield and simplified operational protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cis-D-hydroxyproline has been plagued by inefficient methodologies that hinder large-scale industrial adoption. One prevalent route involves the use of Boc-protected precursors, which necessitates the consumption of costly Boc-anhydride and requires multiple extraction and concentration steps to remove protecting groups, ultimately resulting in a complex workflow with moderate yields around 83%. Another common approach employs catalytic hydrogenation of cis-D-hydroxyproline ethyl ester under high pressure (50 psi) using palladium catalysts; while effective, this method introduces significant safety risks associated with high-pressure hydrogen gas and requires expensive noble metal catalysts that add to the production cost. Furthermore, older epimerization techniques using acetic anhydride often suffer from prolonged reaction times exceeding 24 hours and relatively low yields of approximately 74.7%, making them unsuitable for modern, high-throughput manufacturing environments where time and resource efficiency are paramount.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent offers a streamlined alternative that bypasses the need for protecting groups and high-pressure equipment entirely. By employing a dynamic racemization system using acetic acid and n-butyraldehyde, the process achieves a direct stereochemical inversion of trans-L-hydroxyproline to the desired cis-D-isomer under atmospheric pressure and moderate temperatures ranging from 85°C to 90°C. The true genius of this approach lies in its iterative nature; rather than discarding the mother liquor containing unreacted starting material, the process recycles this stream by supplementing it with fresh trans-L-hydroxyproline for subsequent reaction cycles. This continuous loop allows the system to drive the equilibrium towards the product over multiple batches, significantly enhancing the overall utilization of raw materials and enabling the accumulation of crude product with consistently high chiral purity before the final purification step.
Mechanistic Insights into Acid-Catalyzed Dynamic Racemization
The core chemical transformation relies on the interaction between the substrate, trans-L-hydroxyproline, and a specific combination of an organic acid and a racemizing agent. In this system, acetic acid serves a dual purpose as both the solvent and the proton source, facilitating the reversible opening and closing of the pyrrolidine ring or the formation of an imine intermediate with n-butyraldehyde. This transient intermediate lowers the energy barrier for epimerization at the alpha-carbon, allowing the thermodynamically less stable or kinetically trapped trans-isomer to equilibrate into the cis-configuration. The reaction is typically conducted at elevated temperatures between 85°C and 90°C for a duration of 8 to 15 hours, ensuring sufficient thermal energy to overcome activation barriers while maintaining the stability of the amino acid backbone. The precise stoichiometry is critical, with a mass ratio of trans-L-hydroxyproline to acetic acid optimized between 1:3 and 1:6, and the n-butyraldehyde catalyst employed in minute quantities ranging from 1:0.01 to 1:0.04 relative to the substrate.
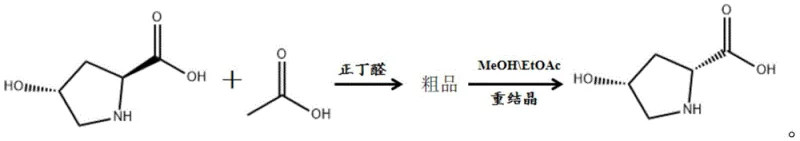
Following the reaction phase, the separation mechanism exploits the differential solubility of the cis and trans isomers in the reaction medium. Upon cooling the reaction mixture to 20-25°C, the target cis-D-hydroxyproline preferentially crystallizes out of the solution due to its lower solubility compared to the trans-isomer and other impurities. This solid-liquid separation yields a crude product with a chiral purity typically exceeding 96%, while the mother liquor retains a significant portion of unconverted trans-L-hydroxyproline. Instead of treating this mother liquor as waste, the process reintroduces it into the reactor along with a fresh charge of raw material, effectively creating a semi-continuous manufacturing loop. This dynamic kinetic resolution strategy ensures that the unreacted enantiomer is given multiple opportunities to convert, thereby maximizing the theoretical yield and minimizing raw material waste, which is a key consideration for sustainable chemical manufacturing.
How to Synthesize Cis-D-Hydroxyproline Efficiently
To implement this synthesis effectively, operators must adhere to strict parameters regarding temperature control and solvent ratios to ensure reproducibility and high purity. The process begins with the racemization step where precise amounts of acetic acid and n-butyraldehyde are combined with the substrate, followed by a controlled cooling crystallization to isolate the crude solid. The critical innovation lies in the recycling protocol, where the mother liquor is not discarded but rather reused for at least six consecutive cycles, with fresh substrate added to maintain the reaction volume and concentration. Detailed standard operating procedures regarding the exact timing of additions, filtration rates, and drying conditions are essential for maintaining the integrity of the crystal lattice and preventing the inclusion of impurities. For a comprehensive guide on the specific operational steps and safety precautions required for this synthesis, please refer to the standardized protocol outlined below.
- Racemize trans-L-hydroxyproline with acetic acid and n-butyraldehyde at 85-90°C for 8-15 hours.
- Cool the mixture to 20-25°C for recrystallization and separate the crude cis-D-hydroxyproline solid from the mother liquor.
- Recycle the mother liquor by adding fresh trans-L-hydroxyproline and repeating the racemization cycle at least 6 times to maximize yield.
- Dissolve all collected crude products in a methanol and ethyl acetate mixture, then recrystallize to obtain high-purity cis-D-hydroxyproline.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. By eliminating the need for expensive protecting group reagents like Boc-anhydride and avoiding the use of precious metal catalysts such as palladium, the raw material costs are significantly reduced, leading to a more competitive pricing structure for the final API intermediate. Furthermore, the simplification of the workflow removes several unit operations such as high-pressure hydrogenation and complex extraction sequences, which not only lowers capital expenditure requirements for equipment but also reduces the operational overhead associated with maintenance and safety compliance. This streamlined approach facilitates a more agile supply chain capable of responding quickly to market demands without the bottlenecks typically associated with multi-step synthetic routes.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of high-cost reagents with commodity chemicals. Acetic acid and n-butyraldehyde are bulk industrial chemicals with stable supply chains and low price volatility, unlike specialized chiral auxiliaries or protected amino acids. Additionally, the high efficiency of the mother liquor recycling means that the effective consumption of the primary starting material, trans-L-hydroxyproline, is minimized per kilogram of product produced. This drastic improvement in atom economy translates directly into lower variable costs per batch, allowing manufacturers to offer more aggressive pricing while maintaining healthy margins in a competitive global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by relying on widely available feedstocks rather than niche intermediates that may be subject to geopolitical or logistical disruptions. Trans-L-hydroxyproline is a commercially mature commodity with multiple global suppliers, ensuring that production schedules are not held hostage by single-source dependencies. Moreover, the robustness of the reaction conditions, which do not require specialized high-pressure reactors or cryogenic cooling, means that the process can be easily transferred between different manufacturing sites or scaled up in existing facilities without significant retrofitting, thereby guaranteeing consistent supply continuity for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than conventional methods, aligning with increasingly stringent global regulations on chemical manufacturing. By avoiding heavy metal catalysts and reducing the volume of organic solvents required for extractions, the generation of hazardous waste is substantially curtailed. The simplicity of the operation, involving primarily heating, cooling, and filtration, makes it highly amenable to scale-up from pilot plant to multi-ton commercial production. This scalability ensures that as demand for cis-D-hydroxyproline grows in the pharmaceutical sector, the manufacturing capacity can be expanded rapidly without compromising on quality or environmental standards, providing a sustainable long-term solution for industrial partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dynamic racemization technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs and for procurement specialists assessing the long-term viability of the supply source.
Q: What is the primary advantage of this racemization method over traditional Boc-protection routes?
A: The primary advantage is significant cost reduction and process simplification. Traditional methods often require expensive Boc-anhydride protection groups and complex deprotection steps involving hazardous reagents. This patented dynamic racemization utilizes inexpensive acetic acid and n-butyraldehyde, eliminating the need for protecting groups and allowing for direct conversion with high atom economy.
Q: How does the mother liquor recycling process impact the overall yield?
A: The mother liquor recycling is critical for maximizing yield. Since the racemization equilibrium does not convert 100% of the trans-L-isomer in a single pass, unreacted starting material remains in the mother liquor. By replenishing this liquor with fresh substrate and repeating the cycle at least 6 to 13 times, the process recovers valuable intermediates that would otherwise be wasted, pushing the total crude yield above 88% and final purity over 99.5%.
Q: What are the optimal solvent conditions for the final purification step?
A: The patent specifies that a mixed solution of methanol and ethyl acetate is superior to ethanol-based systems. The optimal mass ratio of the mixed solvent to the crude product is between 0.45:1 and 0.55:1, with a methanol to ethyl acetate ratio of approximately 1:1. This specific solvent system ensures complete dissolution at elevated temperatures (80-85°C) while facilitating efficient crystallization upon cooling, achieving purity levels exceeding 99.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-D-Hydroxyproline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of chiral intermediates for your drug development pipelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical phase or full-scale commercialization. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis via HPLC, to guarantee that every batch of cis-D-hydroxyproline meets the exacting standards required for pharmaceutical applications. We are committed to delivering not just a chemical product, but a reliable partnership that supports your regulatory filings and market launch timelines.
We invite you to engage with our technical procurement team to discuss how this advanced racemization technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your volume needs. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in the efficient and compliant manufacturing of high-value pharmaceutical intermediates.
