Advanced Synthesis of Polycyclic Lactone Intermediates for Benastatin Production
Introduction to Next-Generation Polycyclic Lactone Synthesis
The pharmaceutical industry continuously seeks robust and efficient pathways for constructing complex natural product scaffolds, particularly those exhibiting potent biological activities such as anti-tumor properties. Patent CN108101879B discloses a groundbreaking methodology for the preparation of novel polycyclic lactone compounds, which serve as critical precursors for the synthesis of Benastatin derivatives. These compounds are characterized by a unique fused ring system containing an optional double bond, offering exceptional versatility for further functionalization. The disclosed technology addresses significant bottlenecks in traditional synthetic routes by introducing a streamlined sequence that combines modern cross-coupling techniques with specialized cyclization strategies. For R&D directors and procurement specialists, this represents a pivotal advancement in accessing high-purity pharmaceutical intermediates with improved economic viability. The ability to synthesize these structures with operational simplicity and environmental safety underscores the patent's value in modern medicinal chemistry supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polycyclic lactone frameworks similar to the Benastatin core has been plagued by synthetic inefficiencies and harsh reaction conditions. Traditional approaches often rely on multiple protection-deprotection sequences that drastically reduce overall yield and increase waste generation. Furthermore, conventional cyclization methods frequently require strong Lewis acids or high temperatures that can compromise sensitive functional groups, leading to complex impurity profiles that are difficult to purify. The lack of regioselectivity in earlier methods often results in isomeric mixtures, necessitating costly chromatographic separations that hinder commercial scalability. Additionally, many prior art routes utilize expensive transition metal catalysts that leave residual heavy metals in the final product, posing significant regulatory challenges for API manufacturing. These cumulative factors result in prolonged lead times and inflated production costs, making the reliable supply of such complex intermediates a persistent challenge for the global pharmaceutical market.
The Novel Approach
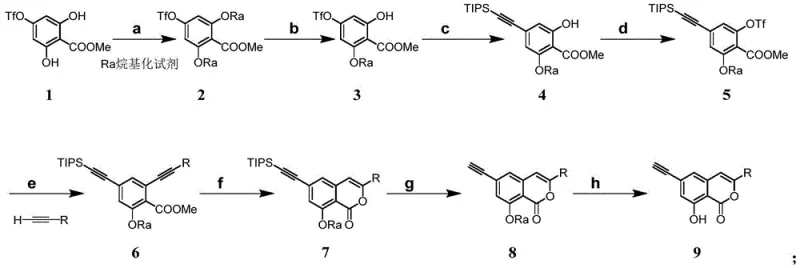
The methodology outlined in the patent introduces a sophisticated yet practical synthetic strategy that overcomes these historical hurdles through a carefully orchestrated eight-step sequence. Central to this innovation is the strategic use of Sonogashira coupling reactions to install alkyne handles, followed by a highly selective indium-catalyzed cyclization to close the lactone ring. This approach allows for the precise assembly of the carbon skeleton under mild conditions, preserving the integrity of sensitive substituents. As illustrated in the comprehensive reaction scheme below, the process begins with simple phenolic starting materials and progresses through controlled alkylation and triflation steps to set the stage for carbon-carbon bond formation. The use of triisopropylsilyl (TIPS) protecting groups ensures stability during the coupling phases, while the final deprotection steps reveal the active pharmacophore with high fidelity. This modular design not only enhances yield but also provides a flexible platform for generating diverse analogues by varying the alkyne components.
Mechanistic Insights into Indium-Catalyzed Cyclization and Coupling
A deep dive into the reaction mechanism reveals why this specific catalytic system offers superior performance for constructing the polycyclic lactone core. The indium tribromide-catalyzed cyclization (Step f) operates through a distinct activation mode where the indium species coordinates with the carbonyl oxygen and the alkyne pi-system, facilitating an intramolecular nucleophilic attack by the phenolic hydroxyl group. This Lewis acid activation lowers the energy barrier for ring closure, allowing the reaction to proceed at room temperature rather than requiring thermal activation. The mechanistic pathway favors the formation of the six-membered lactone ring with excellent regioselectivity, effectively suppressing the formation of unwanted five-membered isomers or polymerization byproducts. Furthermore, the choice of solvent system, specifically a mixture of 1,2-dichloroethane and trifluoroacetic acid, plays a crucial role in stabilizing the cationic intermediates generated during the cyclization event. This precise control over the reaction trajectory ensures a clean impurity profile, which is paramount for meeting the stringent quality standards required for pharmaceutical intermediates.
In parallel, the Sonogashira coupling steps (Steps c and e) utilize a palladium-copper co-catalytic cycle that is optimized for sterically hindered substrates. The use of bis(triphenylphosphine)palladium(II) dichloride in conjunction with cuprous iodide facilitates the oxidative addition and transmetallation processes necessary for linking the aryl triflate with the terminal alkynes. The presence of organic bases like triethylamine serves to regenerate the active copper acetylide species while neutralizing the triflic acid byproduct. This dual-catalyst system is particularly effective in tolerating the electron-rich aromatic rings present in the substrate, preventing competitive side reactions such as homocoupling of the alkyne. By fine-tuning the molar ratios of the catalysts and ligands, the process achieves near-quantitative conversion, minimizing the loss of valuable starting materials. This mechanistic robustness translates directly into process reliability, ensuring consistent batch-to-batch quality essential for long-term supply agreements.
How to Synthesize Polycyclic Lactone Compound Efficiently
The synthesis of the target polycyclic lactone involves a logical progression of functional group transformations designed to maximize efficiency and minimize purification burdens. The process initiates with the selective methylation of a dihydroxy benzoate, followed by the installation of a triflate leaving group to enable subsequent cross-coupling. The introduction of the alkyne moieties is achieved through sequential Sonogashira reactions, utilizing TIPS-protected acetylene to mask reactivity until the appropriate stage. The pivotal cyclization step employs indium tribromide to forge the lactone ring, after which the silyl protecting groups are removed using tetrabutylammonium fluoride. The final step involves the removal of the methyl ether protecting groups using boron trichloride to reveal the free phenolic hydroxyls. Detailed standardized operating procedures for each transformation, including exact stoichiometry, temperature profiles, and workup protocols, are provided in the guide below to ensure reproducibility at scale.
- Perform selective alkylation and deprotection on the starting phenol to prepare the triflate precursor.
- Execute dual Sonogashira coupling reactions to introduce alkyne functionalities at specific positions.
- Conduct indium tribromide-catalyzed cyclization followed by deprotection to yield the final polycyclic lactone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial benefits that directly address the pain points of procurement managers and supply chain directors. The elimination of exotic reagents and the reliance on commodity chemicals significantly lowers the raw material cost base, creating a more resilient supply chain less susceptible to market volatility. The operational simplicity of the process, which avoids cryogenic temperatures or high-pressure reactors, reduces the capital expenditure required for manufacturing infrastructure. This accessibility allows for faster technology transfer from laboratory to pilot plant, accelerating the time-to-market for downstream drug candidates. Moreover, the high atom economy of the coupling and cyclization steps minimizes waste disposal costs, aligning with increasingly strict environmental regulations and sustainability goals. These factors combine to create a compelling value proposition for partners seeking a reliable pharmaceutical intermediate supplier capable of delivering cost-effective solutions.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive starting materials such as methyl gallate derivatives and common alkynes, avoiding the need for precious metal catalysts in the final cyclization step. The high yields reported in the patent examples indicate minimal material loss, which directly improves the cost of goods sold (COGS). Furthermore, the simplified purification requirements, primarily relying on standard column chromatography or crystallization rather than preparative HPLC, reduce processing time and solvent consumption. This economic efficiency makes the large-scale production of these complex intermediates financially viable, offering a competitive edge in the pricing of final API ingredients.
- Enhanced Supply Chain Reliability: By employing robust chemical transformations that are tolerant to minor variations in reaction conditions, the manufacturing process ensures consistent output quality and quantity. The use of stable intermediates that can be stored or transported without degradation adds a layer of security to the supply chain, mitigating risks associated with just-in-time delivery models. The scalability of the Sonogashira coupling and indium-catalyzed steps means that production volumes can be ramped up rapidly to meet surging demand without compromising purity specifications. This reliability is critical for pharmaceutical companies managing tight development timelines and regulatory filing schedules.
- Scalability and Environmental Compliance: The synthetic route is designed with green chemistry principles in mind, utilizing solvents that are easier to recover and recycle, such as dichloromethane and ethyl acetate. The absence of toxic heavy metal residues in the final product simplifies the regulatory approval process and reduces the burden on quality control laboratories. The mild reaction conditions also enhance operator safety, lowering the risk of industrial accidents and associated downtime. These attributes facilitate seamless commercial scale-up from 100 kgs to 100 MT annual production capacities, ensuring a continuous supply of high-purity intermediates for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing pipelines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of the indium-catalyzed cyclization step?
A: The use of indium tribromide allows for mild reaction conditions and high regioselectivity during the formation of the lactone ring, significantly reducing byproduct formation compared to traditional acid-catalyzed methods.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes standard solvents like dichloromethane and DMF, and avoids extreme pressures or temperatures, making it highly suitable for commercial scale-up from kilograms to metric tons.
Q: How does this intermediate contribute to Benastatin G synthesis?
A: The polycyclic lactone serves as a versatile building block that can be coupled with other aromatic systems via Sonogashira reaction to construct the complex tetracyclic core found in Benastatin G and J.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycyclic Lactone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced methodologies like the one described in CN108101879B to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality and consistency makes us the preferred partner for multinational corporations seeking to secure their supply chains for critical oncology ingredients.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of adopting this technology for your pipeline. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to accelerate the development of next-generation therapeutics through superior chemical manufacturing excellence.
