Advanced Manufacturing of Ergothioneine: A Scalable One-Pot Thionation Strategy for Global Supply Chains
Advanced Manufacturing of Ergothioneine: A Scalable One-Pot Thionation Strategy for Global Supply Chains
The global demand for high-purity antioxidants in the pharmaceutical and nutraceutical sectors has necessitated the development of more efficient synthetic routes for critical molecules like ergothioneine. Patent CN114409608A, published in April 2022, introduces a groundbreaking synthetic method that addresses the longstanding challenges of yield and scalability associated with traditional extraction and fermentation processes. This technology leverages a concise four-step sequence, prominently featuring a novel one-pot thionation strategy that dramatically streamlines the introduction of the sulfur atom into the histidine backbone. For R&D directors and procurement specialists seeking a reliable ergothioneine supplier, this patent represents a pivotal shift towards cost-effective and chemically robust manufacturing capabilities that can support the rigorous purity specifications required for human consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of ergothioneine has been hindered by significant bottlenecks inherent to both natural extraction and biological fermentation. Extraction from fungal sources such as Claviceps purpurea or edible mushrooms is plagued by extremely low yields and high operational costs, making it economically unviable for large-scale industrial applications. Furthermore, the isolation of the target molecule from complex biological matrices often requires extensive purification steps to remove impurities, leading to substantial material loss. Similarly, while deep fermentation of mushroom mycelium offers a biological route, it suffers from low product titers and requires complex metabolic regulation and process control to maintain consistency. These conventional methods struggle to meet the growing market demand for cost reduction in antioxidant manufacturing, often resulting in supply chain volatility and inconsistent quality profiles that fail to satisfy stringent regulatory standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN114409608A offers a streamlined chemical synthesis pathway that bypasses the inefficiencies of biological sourcing. The core innovation lies in the ability to realize efficient thionation via a one-pot method, effectively merging what were previously discrete reaction steps into a unified process. This approach not only shortens the overall synthetic route but also significantly enhances the overall yield of the final product. By utilizing readily available starting materials like histidine and employing robust reagents such as iodobenzene acetate and sodium sulfide, the new method ensures a stable and reproducible supply of high-purity ergothioneine. This strategic shift allows manufacturers to achieve substantial cost savings and improved supply chain reliability, positioning this synthetic route as the preferred choice for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into One-Pot Thionation and Methylation
The heart of this synthetic advancement is the sophisticated thionation mechanism that transforms Boc-protected histidine into the sulfur-containing intermediate. The process begins with the activation of the imidazole ring using iodobenzene acetate and sodium bromide in an acetonitrile-water solvent system. This generates a reactive intermediate that subsequently undergoes nucleophilic attack by sodium sulfide at elevated temperatures (70-100°C). This one-pot transformation is critical because it avoids the isolation of unstable intermediates, thereby minimizing degradation and side reactions. The precise control of pH during the workup, adjusting to 3-5 with sulfuric acid, ensures the selective precipitation of the sulfo-Boc histidine product, demonstrating a high level of impurity control that is essential for downstream processing.
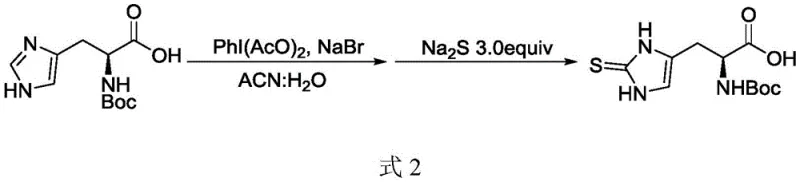
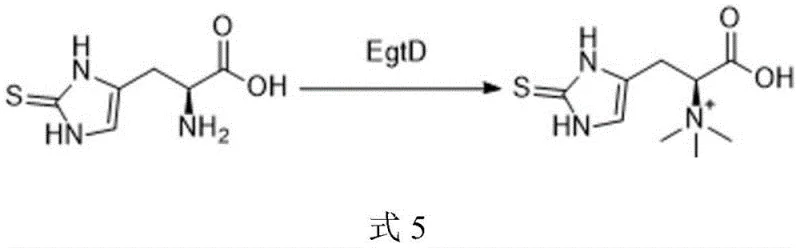
Following the successful introduction of the sulfur moiety and subsequent deprotection of the Boc group using trifluoroacetic acid, the final structural refinement involves methylation of the sulfur atom. The patent discloses two distinct pathways for this crucial step, offering flexibility based on specific production requirements. The chemical route utilizes dimethyl sulfate under basic conditions (NaOH) at controlled low temperatures to effect trimethylation. Alternatively, for applications demanding a greener profile, an enzymatic route employing ergothioneine methyltransferase (EgtD) and S-adenosylmethionine (SAM) is available. This biocatalytic option operates under mild conditions (35-40°C) and avoids the use of hazardous alkylating agents, showcasing the versatility of the platform in meeting diverse regulatory and environmental standards.
How to Synthesize Ergothioneine Efficiently
The synthesis of ergothioneine via this patented route is designed for operational simplicity and high efficiency, making it ideal for translation from laboratory benchtop to industrial reactor. The process initiates with the protection of histidine, followed by the critical one-pot thionation, deprotection, and final methylation. Each step has been optimized to maximize yield and minimize waste, ensuring that the final product meets the rigorous purity specifications demanded by the pharmaceutical industry. The detailed standardized synthesis steps below outline the precise conditions, reagent ratios, and workup procedures necessary to replicate this high-yielding process successfully.
- Protect histidine using di-tert-butyl dicarbonate in a THF/water system to form Boc-histidine.
- Perform one-pot thionation using iodobenzene acetate and sodium sulfide to introduce the sulfur moiety efficiently.
- Remove the Boc protecting group using trifluoroacetic acid and finalize the structure via chemical or enzymatic methylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology translates directly into tangible operational benefits and risk mitigation. The shift from extraction or fermentation to a defined chemical synthesis eliminates the variability associated with biological sources, such as seasonal fluctuations in raw material availability or batch-to-batch inconsistencies in fermentation titers. By relying on stable, commodity-grade starting materials like histidine and standard chemical reagents, manufacturers can secure a more predictable and continuous supply of ergothioneine. This stability is crucial for maintaining production schedules and meeting the just-in-time delivery requirements of global clients, effectively reducing lead time for high-purity antioxidants.
- Cost Reduction in Manufacturing: The implementation of the one-pot thionation strategy fundamentally alters the cost structure of ergothioneine production. By consolidating multiple reaction steps into a single vessel, the process drastically reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours associated with intermediate isolation and purification. Furthermore, the elimination of expensive transition metal catalysts or complex fermentation media components leads to significant raw material cost optimization. These efficiencies compound to deliver a much lower cost of goods sold (COGS), allowing for competitive pricing strategies without compromising on the quality or purity of the final active ingredient.
- Enhanced Supply Chain Reliability: Dependence on biological extraction often ties supply chains to agricultural cycles and geographic limitations, creating vulnerabilities in the face of climate change or logistical disruptions. This chemical synthesis route decouples production from these biological constraints, enabling manufacturing to occur in standard chemical facilities anywhere in the world. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, further ensures that production can continue uninterrupted even under varying operational conditions. This resilience guarantees a steady flow of material to downstream formulators, safeguarding against stockouts and ensuring business continuity for partners relying on a reliable ergothioneine supplier.
- Scalability and Environmental Compliance: Scaling chemical processes often presents challenges regarding heat transfer and mixing, but the stepwise nature of this synthesis, particularly the controlled addition of reagents in the thionation step, facilitates smooth scale-up from pilot plants to multi-ton reactors. Additionally, the availability of an enzymatic methylation pathway provides a clear route to greener manufacturing, aligning with increasingly strict environmental regulations and corporate sustainability goals. The ability to choose between chemical and enzymatic final steps allows manufacturers to tailor their environmental footprint, minimizing hazardous waste generation and simplifying effluent treatment processes, which is a key factor in long-term operational licensing and compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of ergothioneine based on the proprietary data found in CN114409608A. These insights are derived directly from the patent's experimental examples and technical disclosures, providing a transparent view into the feasibility and advantages of this manufacturing route. Understanding these details is essential for stakeholders evaluating the potential for integrating this high-quality intermediate into their own product portfolios or supply networks.
Q: What are the primary advantages of the one-pot thionation method described in CN114409608A?
A: The primary advantage is the significant simplification of the synthetic route. By combining the oxidation and sulfuration steps into a single pot, the process eliminates intermediate isolation, reduces solvent consumption, and minimizes waste generation compared to traditional multi-step thionation protocols.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent explicitly highlights the method's suitability for large-scale popularization. The use of readily available raw materials like histidine and standard reagents, combined with robust reaction conditions (e.g., temperature control below 40°C followed by heating), ensures the process is adaptable for commercial scale-up from kilograms to metric tons.
Q: Is there an environmentally friendly option for the final methylation step?
A: Absolutely. The patent offers a dual-pathway approach for the final methylation. While chemical methylation using dimethyl sulfate is effective, an enzymatic method using ergothioneine methyltransferase (EgtD) and S-adenosylmethionine (SAM) is also disclosed, providing a greener, biocatalytic alternative that avoids harsh alkylating agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ergothioneine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic technologies described in CN114409608A and are fully equipped to leverage them for our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle complex organic syntheses, including sensitive thionation reactions and biocatalytic transformations, while adhering to stringent purity specifications and rigorous QC labs to guarantee product integrity.
We invite you to collaborate with us to optimize your supply chain for ergothioneine and related antioxidant intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and to discuss how we can support your long-term manufacturing goals with reliable, high-quality chemical solutions.
