Advanced Manufacturing of 1,2-Benzisothiazolin-3-one via Novel Chloronitrile Route
Advanced Manufacturing of 1,2-Benzisothiazolin-3-one via Novel Chloronitrile Route
The global demand for effective antimicrobial agents continues to surge across industries ranging from construction materials to personal care, driving the need for robust supply chains of key preservative intermediates. Patent CN102491955A introduces a transformative process method for synthesizing 1,2-benzisothiazolin-3-one (BIT), a cornerstone molecule in the biocide sector. This technical disclosure outlines a strategic departure from legacy manufacturing protocols, offering a pathway that significantly mitigates environmental liabilities while enhancing process efficiency. For R&D directors and procurement strategists, understanding this shift is critical, as it represents a move towards greener chemistry without compromising the structural integrity or efficacy of the final active ingredient. The innovation lies in utilizing o-chlorobenzonitrile as a starting material, bypassing the hazardous reagents associated with traditional dithiosalicylic acid (DTSA) routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 1,2-benzisothiazolin-3-one has relied heavily on dithiosalicylic acid (DTSA) as the primary feedstock. While chemically viable, this conventional pathway presents severe operational and environmental bottlenecks that modern manufacturing facilities strive to eliminate. The core issue stems from the chlorination step, typically employing thionyl chloride (SOCl2), which inevitably generates substantial quantities of sulfur dioxide (SO2) gas as a byproduct. Managing this toxic exhaust requires sophisticated and costly scrubbing systems to meet increasingly stringent environmental regulations. Furthermore, the DTSA route is characterized by the generation of large volumes of saline wastewater, complicating effluent treatment and increasing the overall ecological footprint of the production site. These factors collectively inflate the cost of goods sold (COGS) and introduce significant regulatory risk for manufacturers relying on this outdated technology.
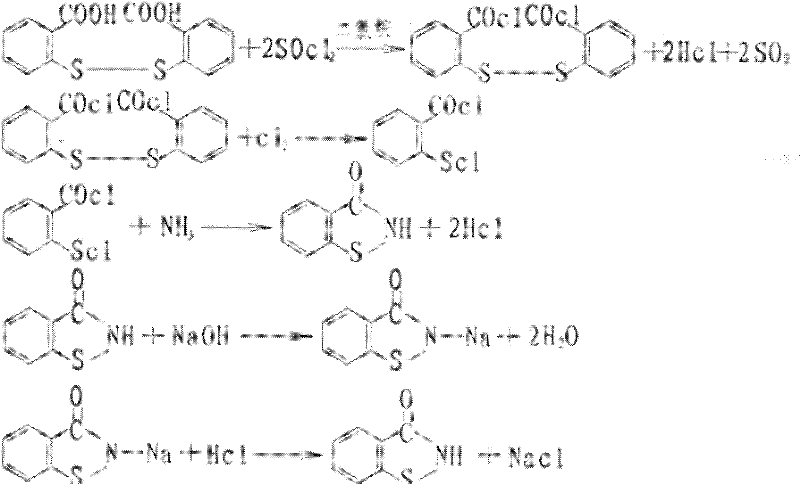
The Novel Approach
In stark contrast, the methodology described in CN102491955A leverages o-chlorobenzonitrile, a readily available commodity chemical, to construct the benzisothiazole scaffold through a more direct and atom-economical sequence. This novel approach fundamentally alters the reaction landscape by replacing the hazardous thionyl chloride step with a controlled oxidative cyclization using chlorine gas in an aqueous medium. By shifting the synthetic logic to a nucleophilic substitution followed by oxidation, the process effectively eliminates the generation of sulfur dioxide废气,thereby removing the need for complex gas abatement infrastructure. Additionally, the workflow is streamlined into fewer distinct operational units, which not only reduces capital expenditure on equipment but also minimizes the potential for yield loss during transfer between stages. This represents a paradigm shift towards sustainable manufacturing that aligns with modern green chemistry principles.
Mechanistic Insights into Nucleophilic Substitution and Oxidative Cyclization
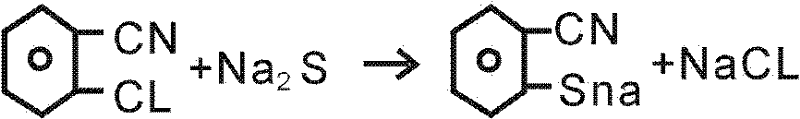
The chemical elegance of this new route lies in its two-stage mechanism, beginning with the formation of the sulfur-carbon bond. In the first stage, o-chlorobenzonitrile undergoes a nucleophilic aromatic substitution where the chlorine atom is displaced by a sulfide ion derived from anhydrous sodium sulfide. This reaction is typically conducted in a polar aprotic solvent like DMF at elevated temperatures, around 90°C, to ensure complete conversion. The resulting intermediate, the sodium salt of o-mercaptobenzonitrile, is then carefully acidified to liberate the free thiol group. This step is crucial as it prepares the molecule for the subsequent ring-closing event, ensuring that the sulfur nucleophile is positioned correctly for intramolecular attack.
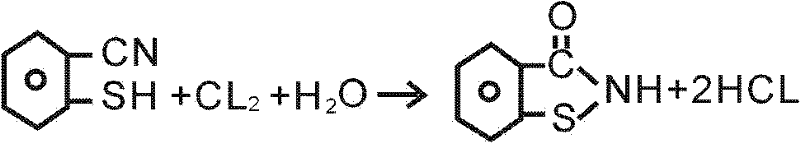
The second and most critical mechanistic phase involves the oxidative cyclization of the o-mercaptobenzonitrile intermediate. Upon exposure to chlorine gas in the presence of water, the thiol group is oxidized, facilitating an intramolecular attack on the nitrile carbon. This cyclization forms the five-membered isothiazolinone ring, yielding the target 1,2-benzisothiazolin-3-one structure. The reaction conditions are tightly controlled, typically maintained between 10°C and 20°C, to prevent over-oxidation or side reactions that could compromise purity. Following the reaction, the crude product undergoes a sophisticated purification regimen involving dissolution in alkali, decolorization with activated carbon, and reprecipitation via acidification. This multi-step purification ensures that the final API intermediate meets rigorous quality standards, free from colored impurities and residual starting materials.
How to Synthesize 1,2-Benzisothiazolin-3-one Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the charging of o-chlorobenzonitrile and anhydrous sodium sulfide into a reactor under nitrogen protection, followed by heating to promote the substitution reaction. Once the intermediate is formed and isolated via acidification, it is immediately subjected to chlorination in a cooled aqueous environment to drive the cyclization. The detailed standardized operating procedures, including specific molar ratios, temperature ramps, and workup protocols, are essential for reproducibility and safety. For a comprehensive guide on the exact execution of these steps, please refer to the technical instructions below.
- React o-chlorobenzonitrile with anhydrous sodium sulfide in DMF at 90°C for 3 hours, followed by acidification to obtain o-mercaptobenzonitrile.
- Oxidize o-mercaptobenzonitrile with chlorine gas in water at 10-20°C to induce cyclization.
- Purify the crude product via alkali dissolution, decolorization, and re-acidification to isolate the final ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials; o-chlorobenzonitrile is a bulk chemical with stable pricing and widespread availability, unlike some specialized precursors used in older methods. This shift reduces dependency on niche suppliers and mitigates the risk of raw material shortages that can disrupt production schedules. Furthermore, the elimination of hazardous gas generation translates directly into lower operational expenditures related to environmental compliance and waste disposal. By removing the need for extensive SO2 scrubbing and reducing saline wastewater volume, manufacturers can achieve significant cost reductions in preservative manufacturing while enhancing their corporate sustainability profiles.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the removal of expensive and hazardous reagents like thionyl chloride. By substituting these with chlorine gas and sodium sulfide, the direct material costs are optimized, and the indirect costs associated with handling dangerous chemicals are minimized. Additionally, the simplified workflow reduces energy consumption and labor hours per batch, contributing to a leaner production model. The qualitative improvement in process efficiency allows for better resource allocation, ensuring that capital is invested in value-added activities rather than waste management infrastructure.
- Enhanced Supply Chain Reliability: From a logistics perspective, sourcing o-chlorobenzonitrile is far more straightforward than securing high-purity DTSA, which may have limited production capacity globally. This abundance of feedstock ensures a more resilient supply chain capable of withstanding market fluctuations. The robustness of the reaction conditions also means that production can be scaled up or down with greater flexibility, allowing suppliers to respond rapidly to changes in downstream demand. This reliability is crucial for maintaining continuous operations in the fast-paced biocide and agrochemical sectors.
- Scalability and Environmental Compliance: The environmental profile of this method makes it inherently more scalable. With fewer toxic byproducts and a cleaner reaction matrix, obtaining regulatory approvals for capacity expansion becomes less burdensome. The process aligns with global trends towards greener manufacturing, potentially qualifying facilities for environmental incentives or preferred vendor status with eco-conscious multinational corporations. This forward-thinking approach future-proofs the supply chain against tightening environmental regulations, ensuring long-term viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 1,2-benzisothiazolin-3-one using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these nuances is vital for making informed decisions about sourcing strategies and technology licensing.
Q: How does this new process reduce environmental impact compared to the DTSA route?
A: The traditional DTSA route generates significant sulfur dioxide (SO2)废气 and brine waste. This novel chloronitrile route eliminates the use of thionyl chloride, thereby removing SO2 emissions and simplifying waste treatment.
Q: What is the expected purity profile of BIT produced via this method?
A: The patent data indicates a yield of over 99% with rigorous purification steps involving pH-controlled precipitation and decolorization, ensuring a high-purity profile suitable for sensitive industrial applications.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process utilizes commodity raw materials like o-chlorobenzonitrile and standard unit operations (substitution, oxidation, crystallization), making it highly amenable to scale-up from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Benzisothiazolin-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to maintain a competitive edge in the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity 1,2-benzisothiazolin-3-one that adheres to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch meets the exacting standards required by the pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this superior manufacturing route for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this process can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to build a more efficient and sustainable supply chain for your critical intermediates.
