Advanced Split-Feed Sulfonation Technology for High-Purity ATBS Manufacturing
Advanced Split-Feed Sulfonation Technology for High-Purity ATBS Manufacturing
The global demand for multifunctional water-soluble anionic monomers continues to surge, driven by critical applications in oilfield chemistry, water conditioning, and advanced polymer synthesis. At the forefront of this chemical evolution is N-tert-butyl acrylamide sulfonic acid, commonly known as ATBS, a molecule renowned for its exceptional stability and versatility. A pivotal advancement in the manufacturing landscape is documented in patent CN102952043B, which discloses a sophisticated synthesis method that fundamentally re-engineers the traditional sulfonation process. Unlike conventional techniques that often rely on harsh conditions or excessive additives to manage reaction kinetics, this novel approach utilizes a precise split-feed strategy involving acrylonitrile and sulfonating agents. By meticulously controlling the contact between reactants at specific low-temperature ranges and staging the introduction of isobutene, the process achieves a remarkable balance between high yield and superior purity. This technical breakthrough not only addresses long-standing challenges in by-product suppression but also offers a streamlined pathway for industrial scale-up, positioning it as a cornerstone technology for modern specialty chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of ATBS has been plagued by significant technical hurdles that impact both economic efficiency and product quality. Traditional methods, such as those reported in early patents like U.S. Pat 3544597, typically involve reacting acrylonitrile, oleum, and isobutene under relatively mild conditions but often suffer from harsh reaction environments that promote unwanted side reactions. To mitigate these issues and improve yields, subsequent innovations introduced various additives into the reaction system. For instance, some prior art methods necessitate the addition of organic acid anhydrides, such as acetic anhydride, ranging from 0.5% to 50% by weight, to enhance crystallization and yield. While effective to a degree, these approaches introduce substantial complexity and cost, as the inclusion of foreign materials requires additional downstream processing steps for removal. Furthermore, other techniques involving the pre-mixing of organic sulfonated bodies with sulfuric acid add layers of operational difficulty and raw material expense. These conventional pathways often result in products with higher impurity profiles, specifically elevated levels of sulfonation by-products, which can compromise the performance of the final polymer in sensitive applications like electronic industry membranes or high-grade ion exchange resins.
The Novel Approach
In stark contrast to the additive-heavy strategies of the past, the method disclosed in patent CN102952043B achieves superior results through elegant process engineering rather than chemical augmentation. The core innovation lies in the segmentation of the sulfonating agent into a first solution and a second solution, each prepared by contacting acrylonitrile with sulfonating agents at controlled low temperatures between -30°C and 15°C. This bifurcation allows for a more nuanced control over the reaction exotherm and the local concentration of reactive species. Subsequently, the process involves a staged introduction of isobutene, where the first solution contacts with a portion of the isobutene feed (10-100%) alongside the second solution to form a third intermediate solution. This is followed by a second reaction stage where the remaining isobutene is introduced. This sequential addition mechanism effectively minimizes the formation of sulfonation by-products without the need for any extra reagents. The result is a cleaner reaction profile that inherently produces a paste product with significantly reduced impurity content, thereby simplifying the purification workflow and eliminating the costs associated with purchasing and removing auxiliary chemicals.
Mechanistic Insights into Split-Feed Sulfonation and Ritter-Type Reaction
The chemical elegance of this synthesis route is rooted in the precise manipulation of reaction kinetics and thermodynamics during the formation of the sulfonic acid moiety. The process begins with the preparation of two distinct solutions where acrylonitrile acts as both a reactant and a solvent medium, interacting with sulfonating agents that are preferably oleum or concentrated sulfuric acid with concentrations between 98% and 103% by weight. The molar ratio of acrylonitrile to the total sulfonating agent is maintained within a broad window of 5-25:1, ensuring that the nitrile group is sufficiently available for the subsequent Ritter-type reaction with isobutene. By maintaining the initial contact temperatures between -30°C and 15°C, preferably -15°C to 0°C, the system prevents premature polymerization of the acrylonitrile and controls the vigorous nature of the sulfonation. The strategic splitting of the sulfonating agent allows the reaction to proceed with a gradient of acidity and water content, which is critical for stabilizing the intermediate carbocation species formed during the isobutene addition. This controlled environment ensures that the electrophilic attack on the nitrile nitrogen occurs selectively, favoring the formation of the desired amide linkage over competing sulfonation of the isobutene itself.
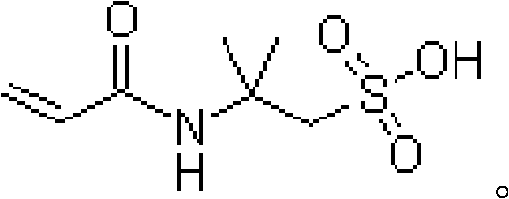
Furthermore, the structural integrity of the final molecule, as depicted in the chemical diagram, relies heavily on the protection offered by the amide group and the salt-insensitive sulfonic group. The patent highlights that by avoiding the introduction of extraneous materials, the reaction system remains chemically homogeneous, which facilitates the growth of larger, more uniform crystals during the precipitation phase. The purification mechanism typically involves a recrystallization step using aqueous acetic acid, where the crude product is dissolved at elevated temperatures (85-105°C) and slowly cooled. This thermal cycling exploits the solubility differences between the target ATBS and residual impurities like single sulfonation isobutylene. The result is a final product with purity specifications consistently exceeding 99% by weight, demonstrating that the mechanistic control exerted during the synthesis phase directly translates to exceptional downstream separability. This level of purity is paramount for applications requiring high-performance polymers, where even trace impurities can act as chain terminators or destabilizers.
How to Synthesize N-tert-butyl acrylamide sulfonic acid Efficiently
Implementing this advanced synthesis route requires strict adherence to the temperature profiles and addition rates defined in the patent to ensure reproducibility and safety. The process is designed to be scalable, moving seamlessly from laboratory verification to commercial production without the need for exotic equipment. Operators must focus on the precise preparation of the first and second sulfonating solutions, ensuring that the concentration gradients are maintained to optimize the reaction trajectory. The subsequent staged addition of isobutene is the critical control point; rushing this step can lead to localized hot spots and increased by-product formation. Detailed standard operating procedures regarding the specific timing of the isobutene valve closures and the agitation speeds during the paste formation are essential for maximizing the yield, which has been demonstrated to reach levels as high as 93.5% in optimized embodiments. For a comprehensive guide on the exact operational parameters and safety protocols required for this synthesis, please refer to the standardized technical documentation below.
- Prepare two separate solutions by contacting acrylonitrile with first and second sulfonating agents (oleum or sulfuric acid) at temperatures between -30°C and 15°C.
- React the first solution with 10-100% of the isobutene feed at -20°C to 50°C while introducing the second solution to form a third intermediate solution.
- Contact the third solution with the remaining 0-90% of isobutene feed to obtain a paste product, followed by separation and recrystallization to achieve high-purity ATBS.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this split-feed sulfonation technology offers profound advantages that extend far beyond simple yield metrics. The most immediate impact is observed in the raw material cost structure, as the elimination of organic acid anhydrides and other auxiliary additives removes a significant line item from the bill of materials. In traditional manufacturing, these additives not only represent a direct purchase cost but also incur hidden expenses related to storage, handling, and the energy-intensive processes required to separate them from the final product. By streamlining the reaction to rely solely on acrylonitrile, oleum, and isobutene, manufacturers can achieve substantial cost reductions in oilfield chemical intermediate manufacturing. This simplification of the chemical recipe also reduces the complexity of the supply chain, minimizing the risk of disruptions caused by the scarcity of niche reagents. Consequently, procurement teams can secure a more stable and predictable supply of high-purity ATBS, insulating their operations from the volatility often associated with specialty chemical markets.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction system. By removing the requirement for expensive organic anhydrides or pre-formed organic sulfonated bodies, the process eliminates the capital and operational expenditures associated with dosing systems for these additives. Furthermore, the absence of these foreign materials means that the downstream purification train does not need to accommodate complex separation steps to remove them, leading to significant savings in solvent usage and energy consumption during the drying and recrystallization phases. This lean manufacturing approach ensures that the cost per kilogram of the final active ingredient is optimized, providing a competitive edge in price-sensitive markets such as water treatment and drilling fluids.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on commodity-grade raw materials that are widely available in the global chemical market. Acrylonitrile, sulfuric acid, and isobutene are produced at massive scales for various industries, ensuring that suppliers are not dependent on single-source vendors for specialized catalysts or modifiers. This diversification of raw material sources reduces the lead time for high-purity oilfield chemical intermediates and mitigates the risk of production stoppages due to raw material shortages. Additionally, the robustness of the process, which tolerates a range of molar ratios and temperatures without compromising product quality, allows for greater operational flexibility, enabling manufacturers to maintain continuous production schedules even when facing minor fluctuations in feedstock quality.
- Scalability and Environmental Compliance: The environmental footprint of ATBS production is markedly reduced through this methodology, aligning with increasingly stringent global regulations on industrial emissions and waste. The reduction in by-product formation, particularly single sulfonation isobutylene, means that the waste streams generated during filtration and purification are less contaminated, simplifying wastewater treatment requirements. Moreover, the process operates efficiently without the need for transition metal catalysts, which often pose disposal challenges and potential contamination risks in the final product. This green chemistry advantage facilitates easier regulatory approval for new facilities and supports the sustainability goals of downstream customers in the agrochemical and personal care sectors who are demanding greener supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific ATBS synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of the split-feed mechanism and its impact on impurity profiles is crucial for R&D teams evaluating this route for technology transfer. The answers below clarify how this method distinguishes itself from legacy processes in terms of operational simplicity and product performance.
Q: How does the split-feed sulfonation method improve ATBS purity compared to conventional single-step processes?
A: By dividing the sulfonating agent into two portions and controlling the addition of isobutene in stages, the reaction effectively suppresses the formation of sulfonation by-products, specifically reducing single sulfonation isobutylene content and achieving crude purities exceeding 96% without additional additives.
Q: Does this synthesis route require expensive organic anhydrides or transition metal catalysts?
A: No, the patented method eliminates the need for extra organic acid anhydrides or complex catalysts often used in prior art to improve yield, relying instead on precise stoichiometric control of acrylonitrile, oleum, and isobutene to drive the reaction efficiency.
Q: What are the typical purity specifications achievable for ATBS produced via this method?
A: Following the standard recrystallization purification step described in the patent, the final ATBS product consistently achieves purity levels greater than 99% by weight, making it highly suitable for demanding applications in oilfield chemistry and water treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-tert-butyl acrylamide sulfonic acid Supplier
As the chemical industry evolves towards more efficient and sustainable manufacturing paradigms, the ability to translate complex patent technologies into commercial reality becomes a defining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging deep technical expertise to master advanced synthesis routes like the split-feed sulfonation process for ATBS. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high purity and yield are fully realized in large-scale operations. We understand that consistency is key for our partners, which is why our rigorous QC labs enforce stringent purity specifications on every batch, guaranteeing that the ATBS supplied meets the exacting standards required for oilfield and water treatment applications. Our commitment to quality assurance ensures that the impurity profiles remain tightly controlled, delivering a product that performs reliably in your most critical formulations.
We invite forward-thinking enterprises to collaborate with us to unlock the full potential of this innovative manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this additive-free synthesis route for your operations. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Together, we can build a more resilient and cost-effective supply chain for high-performance specialty chemicals.
