Advanced Green Synthesis of 2,2-Dimethyl-5-(4-Chlorobenzyl)Cyclopentanone for Industrial Metconazole Production
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of key agrochemical intermediates, specifically targeting the production of metconazole precursors. Patent CN106045830B introduces a transformative preparation method for 2,2-dimethyl-5-(4-chlorobenzyl)cyclopentanone, a critical building block in modern crop protection chemistry. This technical disclosure addresses long-standing industry challenges regarding process safety and environmental compliance by fundamentally redesigning the synthetic pathway. Instead of relying on hazardous alkylation agents, the invention leverages a sophisticated sequence of aldol condensation and catalytic hydrogenation steps. This strategic shift not only enhances the safety profile of the manufacturing process but also ensures a robust supply chain for high-purity intermediates essential for broad-spectrum fungicide formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,2-dimethyl-5-(4-chlorobenzyl)cyclopentanone has been plagued by significant safety and environmental hurdles inherent to traditional alkylation strategies. As illustrated in prior art routes, such as those originating from dimethyl adipate, the process necessitates multiple methylation steps that rely heavily on methyl bromide, a compound known for its high toxicity and ozone-depleting potential. Furthermore, these conventional pathways often require the use of sodium hydride, a pyrophoric reagent that poses severe fire and explosion risks upon contact with moisture, thereby demanding expensive and complex safety infrastructure. The accumulation of halogenated waste streams and the difficulty in controlling exothermic methylation reactions further complicate the post-treatment phase, leading to increased operational costs and regulatory scrutiny for manufacturers attempting to scale these legacy processes.
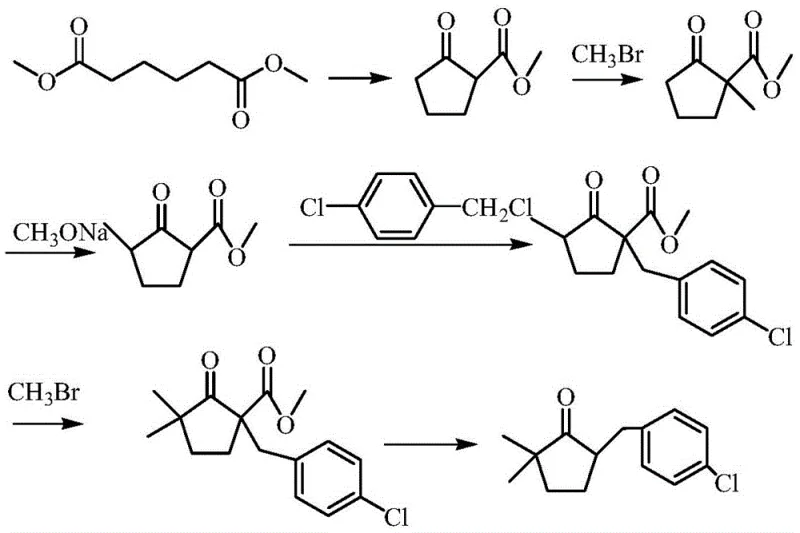
The Novel Approach
In stark contrast to these hazardous legacy methods, the patented approach initiates synthesis from 2,2-dimethyl-4-cyclopenten-1-one, a strategically selected starting material that inherently possesses the required dimethyl substitution pattern. By bypassing the need for exogenous methylation entirely, this novel route eliminates the consumption of methyl bromide and the associated risks of handling sodium hydride. The process flow is streamlined into a logical sequence of condensation, reduction, and dehydration reactions that proceed under significantly milder conditions. This fundamental redesign reduces the complexity of the reaction engineering, minimizes the generation of toxic byproducts, and simplifies the purification workflow, ultimately delivering a greener and more economically viable manufacturing solution for the agrochemical sector.
Mechanistic Insights into DBU-Catalyzed Aldol Condensation and Hydrogenation
The core chemical innovation lies in the initial aldol condensation between 2,2-dimethyl-4-cyclopenten-1-one and p-chlorobenzaldehyde, catalyzed by 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU). Unlike strong inorganic bases that can promote unwanted side reactions or polymerization, DBU acts as a non-nucleophilic, sterically hindered organic base that facilitates the formation of the carbon-carbon bond with high selectivity. This step generates the unsaturated intermediate (Formula II) with excellent conversion rates at ambient temperatures, preserving the integrity of the sensitive functional groups. The subsequent transfer hydrogenation using ammonium formate and palladium carbon allows for the selective reduction of the double bond without requiring high-pressure hydrogen gas equipment, offering a safer alternative for reducing the enone system while maintaining the ketone functionality intact for downstream processing.
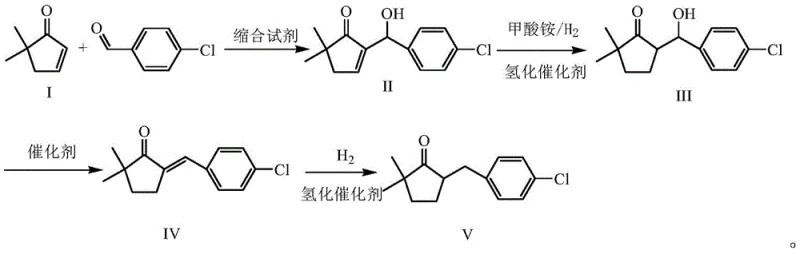
Following the initial reduction, the dehydration step utilizes acetyl chloride as a mild dehydrating agent to convert the hydroxyl intermediate into the conjugated enone (Formula IV). This choice of reagent is critical for impurity control, as it avoids the harsh acidic conditions associated with hydrobromic acid used in older methods, which often lead to ring degradation or chlorination side products. The final catalytic hydrogenation step saturates the remaining double bond to yield the target 2,2-dimethyl-5-(4-chlorobenzyl)cyclopentanone. The cumulative effect of these mechanistic choices is a process that consistently achieves a total yield exceeding 65% and a final product purity greater than 95%, demonstrating superior control over the reaction trajectory and impurity profile compared to prior art.
How to Synthesize 2,2-Dimethyl-5-(4-Chlorobenzyl)Cyclopentanone Efficiently
The execution of this synthesis requires precise control over stoichiometry and reaction parameters to maximize the efficiency of each transformation stage. The process begins with the careful addition of DBU to the aldehyde and ketone mixture in an alcohol solvent, followed by a controlled workup to isolate the condensation product. Subsequent steps involve the sequential addition of hydrogenation catalysts and dehydrating agents under monitored thermal conditions to ensure complete conversion while minimizing thermal stress on the intermediates. For detailed operational parameters, including specific molar ratios, temperature ramps, and isolation techniques, please refer to the standardized synthesis guide provided below.
- Perform aldol condensation between 2,2-dimethyl-4-cyclopenten-1-one and p-chlorobenzaldehyde using DBU catalyst.
- Execute transfer hydrogenation of the condensation product using ammonium formate and palladium carbon.
- Conduct dehydroxylation using acetyl chloride followed by final catalytic hydrogenation to obtain the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers substantial strategic advantages by decoupling production from volatile and regulated hazardous material markets. The elimination of methyl bromide and sodium hydride removes the need for specialized storage facilities and expensive hazard mitigation protocols, directly translating to lower overhead costs and reduced insurance premiums for manufacturing sites. Furthermore, the simplified waste profile, characterized by the absence of bromine-containing wastewater, significantly lowers the cost and complexity of effluent treatment, ensuring smoother compliance with increasingly stringent environmental regulations across global jurisdictions.
- Cost Reduction in Manufacturing: The removal of high-cost, high-risk reagents such as methyl bromide and sodium hydride drastically simplifies the raw material sourcing strategy and reduces the overall bill of materials. By avoiding the need for specialized containment systems required for pyrophoric and toxic substances, capital expenditure for plant setup and maintenance is significantly lowered, while the milder reaction conditions reduce energy consumption associated with heating and cooling cycles.
- Enhanced Supply Chain Reliability: Sourcing 2,2-dimethyl-4-cyclopenten-1-one and p-chlorobenzaldehyde is generally more stable and less subject to regulatory restrictions compared to controlled substances like methyl bromide. This shift ensures a more resilient supply chain that is less vulnerable to sudden regulatory bans or supply disruptions, guaranteeing consistent availability of the intermediate for downstream fungicide production schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations without the need for exotic high-pressure or cryogenic equipment. The green chemistry principles embedded in the route, such as the use of ammonium formate for transfer hydrogenation and the avoidance of heavy metal contamination, facilitate easier regulatory approval and faster time-to-market for commercial scale-up initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route in an industrial setting. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process capabilities and limitations.
Q: How does this new method improve safety compared to traditional routes?
A: The novel process eliminates the use of highly toxic methyl bromide and high-risk sodium hydride, replacing them with safer reagents like DBU and ammonium formate, significantly reducing operational hazards and environmental pollution.
Q: What is the expected purity of the final intermediate?
A: According to patent data, the optimized synthesis route consistently achieves a target product purity exceeding 95%, meeting stringent requirements for downstream fungicide synthesis without extensive purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions (20-80°C) and avoids high-pressure operations where possible, making it highly scalable and compliant with green chemistry standards for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Dimethyl-5-(4-Chlorobenzyl)Cyclopentanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for high-value agrochemical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and robust. We are committed to delivering 2,2-dimethyl-5-(4-chlorobenzyl)cyclopentanone with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the highest industry standards.
We invite global partners to collaborate with us to leverage this advanced technology for their supply chains. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this green synthesis method can enhance your operational efficiency and sustainability goals.
