Advanced Synthesis of Butenafine Hydrochloride: A Breakthrough in Reductive Amination for Commercial Scale-up
Advanced Synthesis of Butenafine Hydrochloride: A Breakthrough in Reductive Amination for Commercial Scale-up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for the production of high-value active pharmaceutical ingredients (APIs) and their intermediates. A pivotal development in this domain is documented in patent CN112209837A, which discloses a highly efficient synthesis method for Butenafine Hydrochloride, a potent allylamine antifungal agent widely used in dermatological treatments. This novel approach leverages a catalytic reductive amination strategy, reacting N-methyl-4-tert-butylbenzylamine with 1-naphthaldehyde under mild hydrogenation conditions. Unlike legacy processes that rely on hazardous halogenated intermediates or stoichiometric reducing agents, this method offers a streamlined, one-pot solution that significantly enhances product purity and yield while minimizing environmental impact. For R&D directors and procurement specialists, understanding this technological shift is critical for optimizing supply chains and ensuring the consistent quality of high-purity pharmaceutical intermediates.
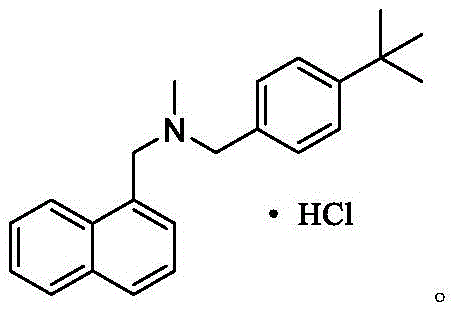
Butenafine Hydrochloride, chemically defined as N-(4-tert-butylphenyl)-N-methyl-1-naphthylmethylamine hydrochloride, functions by inhibiting squalene epoxidase, thereby disrupting fungal cell membrane biosynthesis. Its clinical efficacy relies heavily on the absence of genotoxic impurities and structural analogs that can arise from suboptimal synthetic routes. The patent highlights a direct condensation followed by catalytic reduction, bypassing the complex purification steps associated with older methodologies. This represents a significant leap forward in cost reduction in API manufacturing, as it consolidates multiple unit operations into a simpler workflow that is inherently safer and more economical.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Butenafine has been plagued by significant technical and economic hurdles. The mainstream technology, as referenced in patents such as EP221781 and CN1597660A, typically employs a chloromethylation strategy starting from naphthalene. This involves synthesizing 1-chloromethylnaphthalene, reacting it with methylamine to form N-methylnaphthalene methylamine, and finally substituting with p-tert-butyl benzyl halide. While the raw materials are accessible, this route suffers from severe drawbacks. The use of dry DMF as a solvent creates recovery challenges due to its high boiling point, and the reliance on p-tert-butylbenzyl bromide introduces substantial cost and pollution burdens. Furthermore, the nucleophilic substitution step is prone to over-alkylation, leading to the formation of quaternary ammonium salt impurities that are notoriously difficult to remove, ultimately compromising the final product's purity profile.
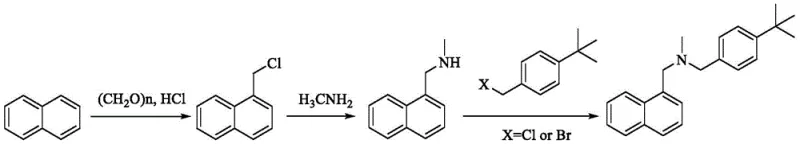
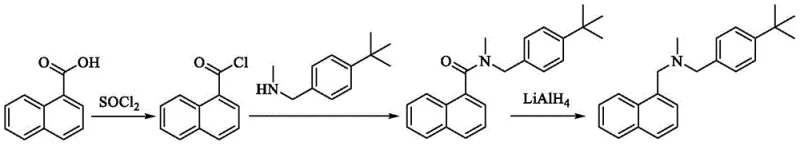
Alternative routes attempting to circumvent these alkylation issues have introduced their own set of complexities. Methods utilizing 1-naphthoic acid derivatives often require conversion to acyl chlorides followed by amidation and subsequent reduction. As illustrated in various prior art documents, these reductions frequently depend on Lithium Aluminum Hydride (LiAlH4), a reagent known for its extreme reactivity and safety hazards, necessitating specialized anhydrous conditions and increasing operational risks. Other approaches employing organosilane compounds with non-metallic boron catalysts or expensive Ruthenium complexes under high-pressure CO2/H2 atmospheres further escalate production costs. These methods not only demand sophisticated equipment capable of withstanding high pressures but also involve toxic heavy metals like cadmium in photocatalytic variants, creating stringent regulatory hurdles for residual metal limits in the final drug substance.
The Novel Approach
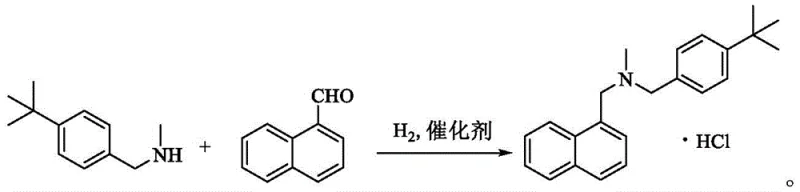
In stark contrast to these convoluted pathways, the methodology presented in patent CN112209837A introduces a paradigm shift through direct reductive amination. This innovative route utilizes 1-naphthaldehyde and N-methyl-4-tert-butylbenzylamine as the primary building blocks. By employing a heterogeneous catalyst such as Raney Nickel or Palladium on Carbon (Pd/C) under a controlled hydrogen atmosphere, the reaction proceeds through an imine intermediate that is selectively reduced to the target tertiary amine. This strategy elegantly sidesteps the generation of quaternary ammonium impurities inherent in alkyl halide substitutions. Moreover, the process operates at moderate temperatures (60-85°C) and low hydrogen pressures (0.05-0.5 MPa), utilizing benign solvents like methanol or ethanol. The culmination of the reaction allows for a direct salification step using HCl in an organic solvent, facilitating immediate crystallization of the hydrochloride salt. This integration of reaction and isolation steps drastically simplifies the downstream processing, offering a compelling solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Catalytic Reductive Amination
The core of this technological advancement lies in the precise control of the reductive amination mechanism. The reaction initiates with the condensation of the primary amine group of N-methyl-4-tert-butylbenzylamine (acting as a secondary amine precursor in the broader context, though here it reacts as a secondary amine with the aldehyde to form an iminium ion or reacts directly depending on protonation state - actually, looking at the reactants: N-methyl-4-tert-butylbenzylamine is a secondary amine. Wait, let's re-read the patent carefully. The patent says "N-methyl-4-tert-butylbenzylamine and 1-naphthaldehyde". N-methyl-4-tert-butylbenzylamine is a SECONDARY amine. Reacting a secondary amine with an aldehyde forms an iminium ion directly, which is then reduced to a tertiary amine. This is chemically sound. The mechanism involves the nucleophilic attack of the secondary amine nitrogen on the carbonyl carbon of 1-naphthaldehyde, facilitated by the acidic environment provided by glacial acetic acid or HCl. This leads to the formation of a reactive iminium ion intermediate. The presence of the acid catalyst is crucial as it activates the carbonyl group and stabilizes the leaving water molecule, driving the equilibrium towards iminium formation.
Once the iminium species is generated, the heterogeneous catalyst (Raney Ni or Pd/C) adsorbs molecular hydrogen onto its surface. The hydrogen atoms are then transferred to the electron-deficient carbon of the iminium bond in a stereoselective manner. This hydrogenation step is highly chemoselective; under the optimized conditions described (0.3 MPa H2, 60-65°C), the catalyst reduces the C=N bond without affecting the aromatic rings of the naphthalene or the phenyl group. This selectivity is paramount for maintaining the structural integrity of the molecule and preventing the formation of saturated ring byproducts. Furthermore, the use of a heterogeneous catalyst allows for easy filtration and recovery, contrasting sharply with homogeneous systems that require complex separation techniques. The final step involves neutralizing the reaction mixture and treating it with HCl, which protonates the tertiary amine to form the stable hydrochloride salt, precipitating out of the solvent matrix with exceptional purity levels exceeding 99.7% as verified by HPLC analysis.
How to Synthesize Butenafine Hydrochloride Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and reaction parameters to maximize yield and minimize impurity profiles. The patent outlines a robust protocol where the molar ratio of the amine to the aldehyde is tightly controlled between 1:1.0 and 1:1.1, ensuring complete consumption of the more valuable or difficult-to-remove component. The choice of solvent plays a critical role, with lower alcohols like methanol and ethanol proving optimal for solubility and reaction kinetics. The addition of a weak acid like glacial acetic acid promotes iminium formation without causing excessive degradation. Following the hydrogenation, the workup procedure is remarkably straightforward: neutralization, extraction, concentration, and direct salification. This simplicity is a key driver for its industrial viability.
- Condensation: React N-methyl-4-tert-butylbenzylamine and 1-naphthaldehyde in a solvent like methanol with an acid catalyst to form the imine intermediate.
- Hydrogenation: Introduce hydrogen gas (0.05-0.5 MPa) in the presence of Raney Ni or Pd/C catalyst at 60-85°C to reduce the imine to the amine.
- Salification: Neutralize the mixture, extract the product, and treat with HCl in an organic solvent to crystallize pure Butenafine Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this reductive amination pathway offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of hazardous reagents and the simplification of the process flow translate directly into operational resilience and cost efficiency. By adopting this method, manufacturers can mitigate the risks associated with volatile raw material markets and stringent environmental regulations.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the replacement of expensive and hazardous reagents. Traditional routes relying on Lithium Aluminum Hydride or noble metal catalysts like Ruthenium incur high raw material costs and require specialized containment infrastructure. In contrast, the use of Raney Nickel or Pd/C represents a fraction of the cost, and these catalysts can often be recycled. Furthermore, the avoidance of high-boiling solvents like DMF reduces energy consumption during solvent recovery and distillation. The consolidation of the synthesis and salification steps also reduces labor hours and equipment occupancy time, leading to substantial overall cost savings in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically 1-naphthaldehyde and N-methyl-4-tert-butylbenzylamine, are commercially available and stable, unlike sensitive acyl chlorides or alkyl halides which may have shorter shelf lives or stricter transport regulations. The robustness of the reaction conditions—operating at near-atmospheric pressure and moderate temperatures—means that the process can be executed in standard stainless steel reactors available in most multipurpose chemical plants. This flexibility reduces dependency on specialized facilities, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring a more consistent supply to downstream API manufacturers.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is vastly superior. It eliminates the generation of halogenated waste streams associated with chloromethylation and avoids the toxic heavy metal residues (like Cadmium) found in photocatalytic alternatives. The aqueous workup and use of alcohol solvents simplify wastewater treatment protocols. The high selectivity of the reaction minimizes the formation of byproducts, reducing the burden on purification columns and lowering the volume of organic waste generated per kilogram of product. This alignment with green chemistry principles facilitates easier regulatory approval and supports sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners.
Q: How does the new reductive amination route improve purity compared to chloromethylation?
A: The traditional chloromethylation route often generates quaternary ammonium salt impurities due to over-alkylation. The new reductive amination method selectively forms the secondary amine via an imine intermediate, effectively eliminating these difficult-to-remove quaternary impurities and achieving HPLC purity above 99.7%.
Q: What are the safety advantages of using Raney Ni over Lithium Aluminum Hydride?
A: Traditional amide reduction routes require Lithium Aluminum Hydride (LiAlH4), which is pyrophoric and requires strict anhydrous conditions and specialized equipment. The patented method uses heterogeneous catalysts like Raney Ni under moderate hydrogen pressure, significantly reducing operational risk and eliminating the need for hazardous hydride handling.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like methanol and ethanol, operates at moderate temperatures (60-85°C) and pressures (0.05-0.5 MPa), and avoids toxic heavy metals like cadmium or expensive noble metals like ruthenium, making it highly scalable and economically viable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Butenafine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN112209837A is essential for maintaining competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of Butenafine Hydrochloride adheres to the highest international standards. Our facility is equipped to handle the specific requirements of catalytic hydrogenation, including high-pressure reactors and advanced filtration systems necessary for heterogeneous catalyst management.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your sourcing strategy with a reliable partner dedicated to quality, innovation, and long-term supply security.
