Advanced Manufacturing of Doravirine Intermediate: Overcoming Cost and Safety Barriers
The pharmaceutical landscape for HIV treatment has been significantly reshaped by the introduction of Doravirine, a next-generation non-nucleoside reverse transcriptase inhibitor developed to combat drug-resistant strains. As depicted in the structural analysis, the molecule relies on a sophisticated architecture where the 3-chloro-5-iodophenol moiety serves as a critical building block for the final assembly. 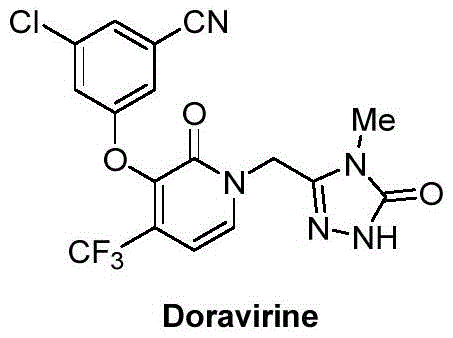 . The efficient production of this specific intermediate, chemically defined as 3-chloro-5-iodophenol (Compound I), represents a major bottleneck in the supply chain due to the complexity of introducing the iodine atom at the meta-position relative to the hydroxyl group while maintaining the chlorine substituent. Recent intellectual property developments, specifically patent CN114163309B, have unveiled a transformative preparation method that shifts the paradigm from precious metal catalysis to classical, robust organic transformations. This innovation addresses the urgent need for a reliable doravirine intermediate supplier by offering a pathway that is not only economically viable but also inherently safer for large-scale industrial operations. The strategic importance of this intermediate cannot be overstated, as it directly impacts the availability and cost of the final antiretroviral therapy, making the optimization of its synthesis a priority for global procurement teams.
. The efficient production of this specific intermediate, chemically defined as 3-chloro-5-iodophenol (Compound I), represents a major bottleneck in the supply chain due to the complexity of introducing the iodine atom at the meta-position relative to the hydroxyl group while maintaining the chlorine substituent. Recent intellectual property developments, specifically patent CN114163309B, have unveiled a transformative preparation method that shifts the paradigm from precious metal catalysis to classical, robust organic transformations. This innovation addresses the urgent need for a reliable doravirine intermediate supplier by offering a pathway that is not only economically viable but also inherently safer for large-scale industrial operations. The strategic importance of this intermediate cannot be overstated, as it directly impacts the availability and cost of the final antiretroviral therapy, making the optimization of its synthesis a priority for global procurement teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-chloro-5-iodophenol has been plagued by significant technical and economic hurdles that hinder commercial scalability. One prominent prior art route, illustrated in the reaction scheme below, relies on a meta C-H bond borylation-oxidation strategy mediated by an Iridium catalyst. 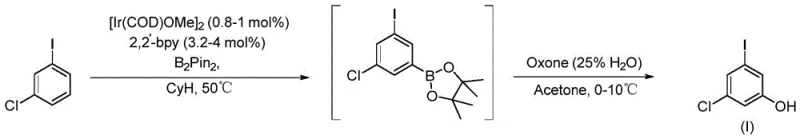 . While chemically elegant, this approach is commercially prohibitive due to the exorbitant cost of the methoxy(cyclooctadiene)iridium dimer and the requisite bipyridine ligands, which drive up the Bill of Materials (BOM) significantly. Furthermore, the use of Oxone as an oxidant introduces severe safety liabilities; it is highly hygroscopic and can decompose rapidly to release oxygen gas, creating potential over-pressurization hazards in closed reactor systems. Another existing methodology attempts to construct the ring system starting from 3,5-dinitroiodobenzene, as shown in the following diagram.
. While chemically elegant, this approach is commercially prohibitive due to the exorbitant cost of the methoxy(cyclooctadiene)iridium dimer and the requisite bipyridine ligands, which drive up the Bill of Materials (BOM) significantly. Furthermore, the use of Oxone as an oxidant introduces severe safety liabilities; it is highly hygroscopic and can decompose rapidly to release oxygen gas, creating potential over-pressurization hazards in closed reactor systems. Another existing methodology attempts to construct the ring system starting from 3,5-dinitroiodobenzene, as shown in the following diagram.  . This pathway is equally problematic, necessitating the use of Boron Tribromide (BBr3) for demethylation, a reagent known for its fuming nature, strong corrosiveness, and violent reaction with water. Additionally, the reduction of nitro groups using iron powder generates massive quantities of iron mud waste, creating a substantial environmental burden and complicating waste disposal logistics, thereby rendering these conventional methods unsuitable for modern green chemistry standards.
. This pathway is equally problematic, necessitating the use of Boron Tribromide (BBr3) for demethylation, a reagent known for its fuming nature, strong corrosiveness, and violent reaction with water. Additionally, the reduction of nitro groups using iron powder generates massive quantities of iron mud waste, creating a substantial environmental burden and complicating waste disposal logistics, thereby rendering these conventional methods unsuitable for modern green chemistry standards.
The Novel Approach
In stark contrast to the aforementioned complexities, the novel approach disclosed in the patent utilizes a readily available starting material, 4-amino-3-chlorophenol, to construct the target scaffold through a logical sequence of electrophilic substitution and functional group interconversion.  . This strategy bypasses the need for rare earth catalysts and cryogenic conditions entirely. The process begins with a highly regioselective iodination of the amino-phenol precursor, leveraging the directing effects of the substituents to place the iodine atom precisely at the desired position. Following this, the amino group is converted into a diazonium salt and subsequently reduced in situ to a hydrogen atom, effectively unmasking the final phenolic structure. This two-step sequence operates under mild thermal conditions, typically between 0°C and 50°C, eliminating the energy-intensive requirements of ultra-low temperature reactors. By utilizing commodity reagents such as N-iodosuccinimide (NIS) or elemental iodine coupled with common oxidants, the process achieves a dramatic simplification of the operational workflow. The result is a streamlined manufacturing protocol that delivers high-purity product with minimal equipment constraints, positioning it as the superior choice for cost reduction in pharmaceutical intermediates manufacturing.
. This strategy bypasses the need for rare earth catalysts and cryogenic conditions entirely. The process begins with a highly regioselective iodination of the amino-phenol precursor, leveraging the directing effects of the substituents to place the iodine atom precisely at the desired position. Following this, the amino group is converted into a diazonium salt and subsequently reduced in situ to a hydrogen atom, effectively unmasking the final phenolic structure. This two-step sequence operates under mild thermal conditions, typically between 0°C and 50°C, eliminating the energy-intensive requirements of ultra-low temperature reactors. By utilizing commodity reagents such as N-iodosuccinimide (NIS) or elemental iodine coupled with common oxidants, the process achieves a dramatic simplification of the operational workflow. The result is a streamlined manufacturing protocol that delivers high-purity product with minimal equipment constraints, positioning it as the superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Regioselective Iodination and Diazotization
The success of this synthetic route hinges on the precise control of electronic effects during the initial iodination step. When 4-amino-3-chlorophenol is subjected to electrophilic aromatic substitution, the hydroxyl and amino groups act as powerful ortho/para directors. However, the steric and electronic environment dictates that the position para to the hydroxyl group (which is meta to the chlorine) is the most nucleophilic site available for attack by the iodinating species. Whether using N-iodosuccinimide in a polar aprotic solvent like DMF or a system of elemental iodine with iodic acid in dichloromethane, the reaction proceeds with high fidelity to yield the 4-amino-3-chloro-5-iodophenol intermediate. The choice of solvent plays a critical role here; polar solvents stabilize the transition state of the electrophilic attack, ensuring complete conversion of the starting material while minimizing the formation of di-iodinated byproducts. This selectivity is paramount for maintaining the purity profile required for downstream pharmaceutical applications, as isomeric impurities can be notoriously difficult to separate later in the synthesis of the final API.
The subsequent transformation involves a classic Sandmeyer-type reduction mechanism, adapted here for high efficiency and safety. The amino group of the iodinated intermediate is first diazotized using sodium nitrite in an acidic medium, generating a highly reactive diazonium salt. In traditional protocols, isolating diazonium salts can be hazardous due to their potential explosivity; however, this patent employs an in-situ reduction strategy that mitigates this risk. Upon the addition of hypophosphorous acid or its salts, the diazonium group undergoes a radical-mediated reduction where the nitrogen gas is expelled, and a hydrogen atom replaces the diazo functionality. This step is conducted at controlled temperatures, typically below 30°C, to prevent the decomposition of the diazonium species before reduction occurs. The use of hypophosphite as the reducing agent is particularly advantageous as it generates phosphorous acid as a byproduct, which is water-soluble and easily removed during the aqueous workup, unlike the heavy metal residues associated with catalytic hydrogenation or the iron sludge from nitro reductions. This mechanistic elegance ensures that the final product, 3-chloro-5-iodophenol, is obtained with exceptional chemical purity, often exceeding 98% after simple crystallization or distillation.
How to Synthesize 3-Chloro-5-Iodophenol Efficiently
The implementation of this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and minimize impurity formation. The process is designed to be robust, allowing for flexibility in reagent selection—such as choosing between NIS for cleaner reactions or elemental iodine for lower raw material costs—depending on the specific economic constraints of the production facility. Operators must ensure that the diazotization step is monitored closely, typically via TLC or HPLC, to confirm the complete consumption of the amine before proceeding to the reduction phase. The workup procedure is notably straightforward, often involving simple filtration or extraction with common organic solvents like ethyl acetate or toluene, followed by a purification step that can be tailored to the desired purity specifications. For a detailed breakdown of the specific operational parameters, reagent ratios, and safety precautions necessary for executing this synthesis, please refer to the standardized guide below.
- Dissolve 4-amino-3-chlorophenol in an organic solvent such as DMF or DCM and react with an iodinating agent like N-iodosuccinimide (NIS) or elemental iodine with an oxidant to form the amino-iodo intermediate.
- Dissolve the intermediate in acid and perform a diazotization reaction using sodium nitrite at low temperatures to generate the diazonium salt.
- Add a reducing agent such as hypophosphorous acid or sodium hypophosphite to reduce the diazonium group in situ, followed by filtration or extraction and purification to obtain high-purity 3-chloro-5-iodophenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers profound strategic benefits that extend far beyond simple unit cost savings. The shift away from proprietary or scarce catalysts to ubiquitous commodity chemicals fundamentally de-risks the supply chain, ensuring continuity of supply even during periods of global raw material volatility. By eliminating the dependency on Iridium, a precious metal subject to significant price fluctuations and geopolitical supply constraints, manufacturers can stabilize their long-term cost structures. Furthermore, the removal of hazardous reagents like Boron Tribromide and the avoidance of heavy metal waste streams significantly lowers the environmental compliance burden, reducing the costs associated with waste treatment and regulatory reporting. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also facilitates faster regulatory approvals for the manufacturing sites, accelerating time-to-market for the final drug product.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, driven primarily by the substitution of high-value inputs with low-cost alternatives. The elimination of the Iridium catalyst and specialized ligands removes a major cost driver from the variable cost equation, while the use of inexpensive reagents like sodium nitrite and hypophosphorous acid keeps the operating expenses minimal. Additionally, the simplified workup procedures reduce the consumption of solvents and energy, as there is no need for complex chromatography or extensive metal scavenging steps. The overall process efficiency translates into a significantly lower cost of goods sold (COGS), providing a competitive margin advantage for generic manufacturers or contract development and manufacturing organizations (CDMOs) producing this high-purity pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the reliance on globally available starting materials. 4-amino-3-chlorophenol is a bulk chemical produced by multiple vendors worldwide, preventing single-source bottlenecks that often plague specialized fine chemical syntheses. The robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation, offering flexibility in production planning. Moreover, the stability of the reagents and intermediates allows for safer storage and transportation, reducing the logistical complexities and insurance costs associated with handling hazardous or unstable compounds. This reliability ensures that production schedules can be met consistently, safeguarding against delays that could impact the broader HIV treatment supply network.
- Scalability and Environmental Compliance: From an operational perspective, the mild reaction conditions and absence of extreme temperatures or pressures make this process ideally suited for scale-up from pilot plant to multi-ton commercial production. The equipment requirements are standard, utilizing glass-lined or stainless steel reactors without the need for specialized cryogenic capabilities or high-pressure vessels. Environmentally, the process generates significantly less hazardous waste; the absence of iron mud and heavy metal residues simplifies effluent treatment and reduces the carbon footprint of the manufacturing operation. This ease of scale-up combined with superior environmental performance positions the technology as a sustainable solution for the long-term manufacturing of complex pharmaceutical intermediates, aligning with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-chloro-5-iodophenol using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of the process for potential partners and technical stakeholders. Understanding these details is crucial for evaluating the integration of this route into existing manufacturing portfolios.
Q: Why is the new synthesis route for Doravirine intermediate considered safer than prior art?
A: The new route eliminates the use of Boron Tribromide (BBr3), which is highly corrosive and poses explosion risks upon contact with water, and avoids the use of moisture-sensitive Oxone which can release oxygen rapidly. It utilizes mild reagents like hypophosphorous acid at moderate temperatures.
Q: How does this method reduce manufacturing costs compared to Iridium-catalyzed routes?
A: By replacing expensive noble metal catalysts like Iridium dimers and specialized ligands with commodity chemicals such as elemental iodine or N-iodosuccinimide, the raw material costs are drastically lowered. Additionally, the removal of heavy metal catalysts simplifies the purification process, reducing downstream processing costs.
Q: What is the expected purity of the final intermediate using this method?
A: The patent data indicates that through simple purification methods like reduced pressure distillation or recrystallization, the final compound (I) can achieve an HPLC purity of greater than 98.0%, with specific examples reaching up to 99.2%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-5-Iodophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent literature are faithfully reproduced at an industrial level. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify every batch of high-purity pharmaceutical intermediate against the most demanding international standards. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this synthesis, guaranteeing a consistent and reliable supply for your Doravirine production needs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the efficiency and profitability of your supply chain.
