Optimizing JAK Inhibitor Intermediates: A Novel Low-Cost Synthesis Strategy for Global Supply Chains
The pharmaceutical industry's relentless pursuit of potent Janus Kinase (JAK) inhibitors has placed immense strategic value on key heterocyclic building blocks, specifically 4-chloro-7H-pyrrolo[2,3-d]pyrimidine. As detailed in the groundbreaking patent CN110386936B, a novel preparation method has emerged that fundamentally reshapes the economic and technical landscape for producing this critical intermediate. This compound serves as the foundational scaffold for blockbuster medications such as Tofacitinib, Ruxolitinib, and SHR0302, making its efficient synthesis a priority for any forward-thinking reliable pharmaceutical intermediates supplier. The patented technology introduces a streamlined two-step approach that bypasses the cumbersome multi-step sequences of legacy methods, offering a pathway to high-purity pharmaceutical intermediates with exceptional selectivity. By leveraging accessible raw materials and optimizing reaction conditions, this innovation addresses the dual challenges of cost containment and environmental compliance that currently burden global supply chains.
![Chemical structures of JAK inhibitors Tofacitinib, Ruxolitinib, SHR0302 and the core intermediate 4-chloro-7H-pyrrolo[2,3-d]pyrimidine](/insights/img/4-chloro-7h-pyrrolo-pyrimidine-synthesis-supplier-20260308043727-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of 4-chloro-7H-pyrrolo[2,3-d]pyrimidine has been plagued by inefficient synthetic routes that impose severe constraints on cost reduction in pharmaceutical intermediates manufacturing. Early methodologies, such as those reported by Haobaoyu et al., relied on a five-step sequence involving expensive and toxic reagents like potassium osmate hydrate and sodium periodate for oxidation steps. These processes not only suffered from mediocre total yields hovering around 45.8% but also generated substantial quantities of hazardous waste acid and wastewater, creating significant disposal liabilities. Alternative routes utilizing thiourea and Raney Ni for desulfurization introduced potent odors and safety hazards, while methods employing sodium hydride posed operational risks due to their sensitivity and handling requirements. Furthermore, the reliance on ozone oxidation in some variations led to unpredictable side reactions and the formation of difficult-to-remove byproducts, complicating the purification process and undermining the overall economic viability of commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN110386936B represents a paradigm shift by condensing the synthesis into a highly efficient two-step sequence that prioritizes atom economy and operational safety. This innovative route utilizes 2-methyl-3,3-dichloroacrylonitrile and trimethyl orthoformate as starting materials, which are commercially abundant and cost-effective compared to the specialized precursors required by prior art. The process eliminates the need for heavy metal catalysts and hazardous desulfurization agents, thereby drastically simplifying the post-treatment workflow and reducing the environmental footprint. By employing a Lewis acid-catalyzed condensation followed by a one-pot cyclization and elimination strategy, the new method achieves total yields as high as 81.7%, nearly doubling the efficiency of older techniques. This substantial improvement in yield, coupled with the reduction in processing steps, directly translates to lower production costs and enhanced throughput for manufacturers seeking a reliable pharmaceutical intermediates supplier capable of meeting rigorous demand.
Mechanistic Insights into Lewis Acid-Catalyzed Condensation and Cyclization
The core technical brilliance of this patent lies in its sophisticated control over reaction kinetics and thermodynamics, particularly during the formation of the pivotal intermediate 1,1-dichloro-2-cyano-4-methoxy-1,3-butadiene. The use of Lewis acid catalysts, such as zinc chloride or cuprous chloride, facilitates a highly selective condensation reaction between the acrylonitrile derivative and the orthoformate, which critically minimizes the formation of polymeric byproducts that often plague traditional pyrrole syntheses. The reaction conditions are meticulously optimized, with temperatures maintained between 50-80°C to ensure complete conversion while preventing thermal degradation of the sensitive nitrile functionality. This precise control over the catalytic environment ensures that the resulting intermediate possesses the necessary electronic configuration to undergo subsequent nucleophilic attack without requiring harsh activation conditions. Such mechanistic precision is essential for maintaining the integrity of the heterocyclic system and ensuring that the final product meets the stringent purity specifications required for downstream API synthesis.
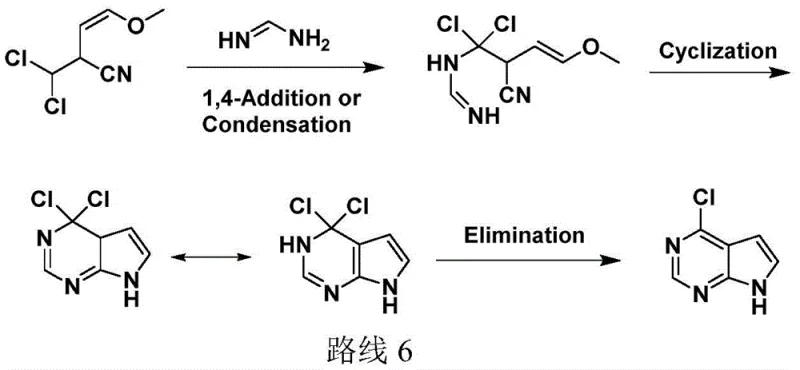
Furthermore, the subsequent cyclization and elimination steps are governed by a unique temperature-programming strategy that maximizes selectivity and minimizes impurity generation. The process involves a controlled dropwise addition of the intermediate into a solution of formamidine salt and alkali, maintaining a low concentration effect that suppresses intermolecular condensation reactions. Initially, the reaction is conducted at moderate temperatures (20-40°C) to favor the 1,4-addition of formamidine to the conjugated system, forming the requisite pyrimidine ring precursor. Subsequently, the temperature is raised to 60-80°C to drive the elimination of hydrogen chloride, finalizing the aromatization of the pyrrole ring. This sequential thermal manipulation prevents the competing substitution of the methoxy group, which would otherwise lead to structural impurities, thereby ensuring that the final 4-chloro-7H-pyrrolo[2,3-d]pyrimidine is obtained with liquid phase purity exceeding 99%. This level of control is indispensable for reducing lead time for high-purity pharmaceutical intermediates by minimizing the need for extensive recrystallization or chromatographic purification.
How to Synthesize 4-Chloro-7H-Pyrrolo[2,3-d]Pyrimidine Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters regarding solvent selection, catalyst loading, and thermal profiles to replicate the high yields reported in the patent data. The process begins with the condensation step in a non-polar or moderately polar solvent such as n-hexane or tetrahydrofuran, where the Lewis acid catalyst is introduced in minute quantities (0.5-10% mass ratio) to initiate the transformation without promoting side reactions. Following the isolation of the diene intermediate, the second stage involves a one-pot reaction in an alcoholic solvent like methanol, where the stoichiometry of the base and formamidine salt is critical for driving the cyclization to completion. Operators must carefully monitor the exothermic nature of the addition phase and strictly follow the temperature ramping protocol to ensure the elimination step proceeds efficiently. Detailed standardized operating procedures and critical process parameters are essential for technology transfer and successful implementation at an industrial scale.
- Condense 2-methyl-3,3-dichloroacrylonitrile with trimethyl orthoformate using a Lewis acid catalyst like zinc chloride at 50-80°C.
- React the resulting intermediate with formamidine salt and alkali in a solvent like methanol under controlled temperature programming.
- Perform elimination reaction at elevated temperatures (60-80°C) to finalize the pyrrolo-pyrimidine ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthesis method offers profound advantages that extend far beyond simple unit cost savings, fundamentally altering the risk profile of the supply chain for JAK inhibitor precursors. By eliminating the dependency on scarce and expensive reagents like potassium osmate and Raney Ni, manufacturers can secure a more stable and predictable supply of raw materials, insulating the production process from volatile market fluctuations associated with specialty chemicals. The reduction in synthetic steps from four or five down to just two significantly decreases the cumulative processing time and equipment occupancy, allowing for faster batch turnover and improved responsiveness to sudden spikes in market demand. Moreover, the simplified workflow reduces the potential for human error and operational deviations, leading to more consistent batch-to-batch quality and reliability. These factors collectively contribute to a robust supply chain architecture that can support the long-term commercialization of downstream therapies without the bottlenecks typically associated with complex heterocyclic synthesis.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic simplification of the material bill and the elimination of costly purification stages associated with heavy metal removal. By avoiding the use of precious metal oxidants and hazardous desulfurization catalysts, the process removes significant line items from the production budget, including the expenses related to specialized waste treatment and regulatory compliance for toxic effluents. The high selectivity of the reaction means that raw materials are converted into the desired product with minimal loss, maximizing the return on investment for every kilogram of input material. Additionally, the ability to recover and recycle solvents like trimethyl orthoformate further enhances the overall cost efficiency, making this route exceptionally competitive for large-volume production. These cumulative savings allow for a more aggressive pricing strategy while maintaining healthy margins, providing a distinct competitive edge in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 2-methyl-3,3-dichloroacrylonitrile and trimethyl orthoformate ensures that the supply chain is not vulnerable to the shortages that often affect specialized fine chemical intermediates. These starting materials are produced by multiple vendors globally, reducing the risk of single-source dependency and enabling procurement teams to negotiate better terms through competitive bidding. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply output. This resilience is crucial for pharmaceutical companies that require guaranteed continuity of supply to maintain their own clinical and commercial timelines. By partnering with a manufacturer utilizing this technology, buyers can secure a long-term source of critical intermediates that is both dependable and scalable.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental protection intensify globally, the green chemistry attributes of this synthesis route provide a significant strategic advantage for sustainable manufacturing. The substantial reduction in wastewater and waste acid generation simplifies the environmental permitting process and lowers the operational costs associated with effluent treatment facilities. The absence of heavy metals and noxious sulfur compounds aligns the process with modern ESG (Environmental, Social, and Governance) goals, making it easier for downstream pharmaceutical clients to justify their sourcing decisions to stakeholders. Furthermore, the straightforward nature of the two-step process facilitates seamless scale-up from pilot plant to multi-ton commercial production without the need for complex engineering modifications. This scalability ensures that the supply can grow in tandem with the market demand for JAK inhibitors, supporting the expansion of therapeutic indications and patient access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology for 4-chloro-7H-pyrrolo[2,3-d]pyrimidine. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs. The answers highlight the practical implications of the chemical innovations on quality, cost, and operational safety.
Q: What are the primary advantages of this new synthesis route over prior art?
A: The novel method eliminates the need for expensive heavy metal oxidants like potassium osmate and hazardous reagents like Raney Ni, significantly reducing environmental waste and operational costs while achieving yields over 80%.
Q: How does the process ensure high purity for pharmaceutical applications?
A: By utilizing temperature programming and controlled dropwise addition, the method minimizes intermolecular condensation and polymer formation, resulting in liquid phase purity exceeding 99%.
Q: Is this process scalable for industrial production?
A: Yes, the process uses commercially available raw materials and avoids complex purification steps, making it highly suitable for large-scale commercial manufacturing of JAK inhibitor intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-7H-Pyrrolo[2,3-d]Pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent literature are faithfully reproduced in our manufacturing facilities. We understand that for critical oncology and immunology intermediates, there is no margin for error; therefore, we adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex chemistries, such as the Lewis acid-catalyzed condensations described in CN110386936B, positions us as a strategic ally for pharmaceutical companies seeking to optimize their supply chains. We are dedicated to delivering not just a chemical product, but a comprehensive solution that enhances your project's velocity and reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your specific supply network. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments that demonstrate our readiness to support your development and commercialization goals. Let us collaborate to bring life-saving JAK inhibitors to patients faster and more efficiently through superior chemical manufacturing.
