Advanced Green Synthesis of Drospirenone Key Intermediates for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of safer, more sustainable manufacturing processes has brought significant attention to the synthesis of hormonal active pharmaceutical ingredients (APIs). Specifically, the production of Drospirenone, a potent progestogen widely used in oral contraceptives like Yasmin, relies heavily on the efficient preparation of its key precursors. A groundbreaking technical disclosure found in patent CN115947773A introduces a novel, environmentally benign methodology for synthesizing 3-pivaloyloxy-5β,6β-epoxy-7-bromo-15β,16β-dimethylene-pregnane-17-one. This critical intermediate serves as the cornerstone for the final assembly of the Drospirenone molecule. Unlike traditional routes that burden the environment with toxic waste, this new approach leverages organocatalysis to achieve high efficiency under mild conditions. By replacing hazardous stoichiometric reagents with catalytic amounts of thiourea derivatives, the process not only aligns with modern green chemistry principles but also offers a robust pathway for reliable pharmaceutical intermediate supplier networks seeking to optimize their production lines.
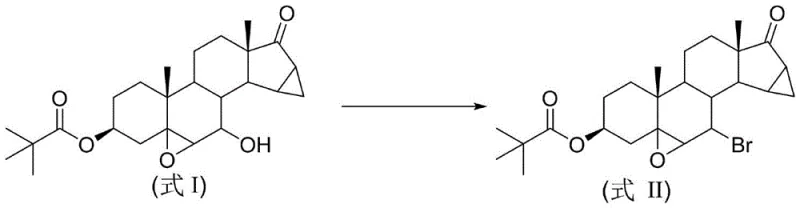
The transition from legacy chemical processes to this innovative thiourea-catalyzed protocol represents a paradigm shift in steroid functionalization. Historically, the installation of the C7-bromo functionality required harsh conditions or problematic reagents that complicated downstream processing. This new methodology addresses those historical bottlenecks by utilizing a substitution reaction that proceeds smoothly at room temperature. The strategic implementation of this technology allows manufacturers to bypass the extensive purification protocols typically associated with phosphorus-containing byproducts. For R&D teams evaluating process scalability, the ability to conduct this transformation in common solvents like dichloromethane or acetonitrile, without the need for cryogenic cooling or inert atmospheres, drastically simplifies the operational complexity. Consequently, this patent provides a blueprint for cost reduction in pharmaceutical intermediates manufacturing by streamlining the synthetic sequence and minimizing the environmental footprint associated with waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing this specific steroidal intermediate have long been plagued by significant chemical and operational inefficiencies. One prevalent historical route involves a sulfonylation followed by bromination, a process that necessitates the use of large excesses of pyridine and sulfonylating agents. Pyridine, while effective as a base, is notorious for its toxicity, unpleasant odor, and difficulty in complete removal from the final product, posing risks to patient safety and requiring rigorous purification steps. Furthermore, this pathway generates substantial quantities of sulfonate waste, complicating effluent treatment and increasing the overall cost of goods sold. Another common approach utilizes triphenylphosphine-mediated bromination, which suffers from the generation of stoichiometric amounts of triphenylphosphine oxide. This phosphorus-containing byproduct is notoriously difficult to separate from the lipophilic steroid product, often leading to dark-colored reaction mixtures and requiring extensive chromatographic purification or multiple recrystallizations. These limitations result in lower overall yields, higher solvent consumption, and a production timeline that is ill-suited for the high-volume demands of the global contraceptive market.
The Novel Approach
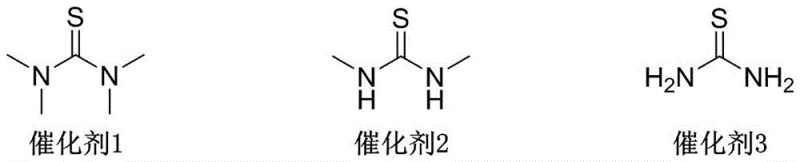
In stark contrast, the method disclosed in CN115947773A revolutionizes this transformation by employing a thiourea-catalyzed substitution mechanism that operates under remarkably mild conditions. By utilizing specific thiourea derivatives, such as tetramethylthiourea or N,N'-dimethylthiourea, the reaction activates the brominating agent effectively without the need for stoichiometric phosphorus reagents or toxic organic bases like pyridine. This "phosphorus-free and pyridine-free" strategy fundamentally alters the impurity profile of the reaction, resulting in a cleaner crude product that can be purified simply through recrystallization. The reaction proceeds efficiently at room temperature, typically reaching completion within 2 to 6 hours, which significantly reduces energy consumption compared to heated or cooled processes. Moreover, the use of commercially available brominating agents like N-bromosuccinimide (NBS) or dibromohydantoin ensures that raw material sourcing remains stable and cost-effective. This novel approach not only enhances the safety profile of the manufacturing facility by eliminating hazardous reagents but also delivers superior economic performance through simplified workup procedures and higher isolated yields.
Mechanistic Insights into Thiourea-Catalyzed Bromination
The core innovation of this synthesis lies in the unique activation mode provided by the thiourea catalyst. Thiourea derivatives function as potent nucleophilic catalysts that interact with the electrophilic bromine source, likely forming a reactive bromo-sulfonium intermediate in situ. This activated species is then capable of converting the 7β-hydroxyl group of the steroid substrate into a superior leaving group, facilitating a subsequent nucleophilic attack by the bromide ion. This mechanism avoids the formation of stable phosphorus-oxygen bonds that characterize traditional Appel-type reactions, thereby preventing the accumulation of hard-to-remove oxide byproducts. The steric and electronic properties of the thiourea catalyst, particularly the methyl substituents seen in Catalyst 1 (tetramethylthiourea), play a crucial role in modulating the reaction rate and selectivity. Experimental data indicates that Catalyst 1 provides optimal performance, achieving yields up to 87%, suggesting that the increased electron density on the sulfur atom enhances the catalyst's ability to activate the brominating agent. This fine-tuned catalytic cycle ensures that the reaction proceeds with high fidelity, minimizing side reactions such as elimination or epoxide ring opening which could compromise the structural integrity of the complex steroid skeleton.
Impurity control is another critical aspect where this mechanistic approach excels. In conventional phosphorus-mediated reactions, the presence of phosphine oxides often leads to emulsion formation during aqueous workup, trapping product and reducing recovery rates. The thiourea-catalyzed system, however, generates urea-like byproducts that are generally more polar and water-soluble, allowing for clean separation during the standard bicarbonate wash and extraction steps described in the patent. The mild reaction conditions further contribute to impurity suppression by avoiding thermal degradation of the sensitive 5β,6β-epoxide moiety. High-performance liquid chromatography (HPLC) analysis of the crude reaction mixture typically shows a dominant product peak with minimal adjacent impurities, validating the selectivity of the catalytic system. This high level of chemical purity at the crude stage is paramount for pharmaceutical manufacturing, as it reduces the burden on downstream purification units and ensures that the final API meets stringent regulatory specifications for genotoxic impurities and residual solvents.
How to Synthesize 3-pivaloyloxy-5β,6β-epoxy-7-bromo-15β,16β-dimethylene-pregnane-17-one Efficiently
The practical execution of this synthesis is designed for seamless integration into existing pilot and commercial plants. The protocol begins by dissolving the starting material, 3-pivaloyloxy-5β,6β-epoxy-7β-hydroxy-15β,16β-dimethylene-pregnane-17-one, in a suitable organic solvent such as dichloromethane, maintaining a concentration that balances reaction kinetics with mixing efficiency. A catalytic amount of the selected thiourea derivative is added to the solution, followed by the controlled addition of the brominating agent. The detailed standardized synthesis steps for optimizing yield and purity are outlined in the guide below.
- Dissolve the 7-beta-hydroxy steroid substrate and a catalytic amount of thiourea derivative in an organic solvent such as dichloromethane.
- Slowly add a solution of the brominating agent (e.g., N-bromosuccinimide) at room temperature and stir for 2 to 6 hours.
- Quench the reaction with saturated sodium bicarbonate, extract with organic solvent, concentrate, and purify the crude product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this thiourea-catalyzed process translates directly into tangible operational improvements and risk mitigation. The elimination of stoichiometric phosphorus reagents removes a major cost driver associated with both raw material purchase and waste disposal. Traditional methods require purchasing expensive triphenylphosphine in molar equivalents to the substrate, whereas this new method utilizes cheap thiourea catalysts in sub-stoichiometric amounts. Furthermore, the removal of pyridine from the process bill of materials simplifies regulatory compliance and reduces the costs associated with handling hazardous volatile organic compounds. The simplified workup procedure, which relies on basic extraction and recrystallization rather than complex chromatography, significantly shortens the batch cycle time. This efficiency gain allows manufacturing facilities to increase throughput without capital investment in new equipment, effectively expanding capacity to meet market demand. Additionally, the use of common, non-proprietary solvents and reagents ensures a resilient supply chain that is less vulnerable to geopolitical disruptions or single-source supplier failures.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in reagent costs and waste treatment expenses. By replacing expensive, stoichiometric phosphorus reagents with catalytic amounts of inexpensive thiourea derivatives, the direct material cost per kilogram of product is significantly lowered. Moreover, the absence of phosphorus oxide byproducts eliminates the need for specialized scavenging resins or extensive chromatographic purification, which are often the most expensive steps in steroid synthesis. The ability to isolate the product via simple recrystallization further reduces solvent consumption and energy usage associated with solvent recovery. These cumulative savings contribute to a substantially improved margin profile for the final API, making the end-product more competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: From a logistics perspective, this methodology relies on commodity chemicals that are widely available from multiple global suppliers. Reagents such as N-bromosuccinimide, dichloromethane, and ethanol are standard inventory items for most fine chemical manufacturers, reducing the lead time for raw material procurement. The robustness of the reaction conditions, which tolerate ambient temperatures and do not require specialized anhydrous or inert environments, minimizes the risk of batch failures due to minor operational deviations. This reliability ensures consistent delivery schedules to downstream API manufacturers, preventing production stoppages that can ripple through the entire pharmaceutical supply chain. The stability of the supply chain is further reinforced by the simplicity of the process, which requires less specialized operator training and maintenance compared to complex catalytic hydrogenation or cryogenic reactions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards and the use of standard unit operations. The reaction does not generate significant heat, allowing for safe scale-up in large reactors without the need for aggressive cooling systems. Environmentally, the process aligns perfectly with increasingly strict global regulations regarding pharmaceutical waste. By eliminating phosphorus and pyridine, the wastewater stream is much easier to treat, reducing the load on effluent treatment plants and lowering the carbon footprint of the manufacturing site. This green chemistry profile not only aids in obtaining environmental permits but also enhances the corporate social responsibility (CSR) standing of the manufacturer, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry professionals.
Q: What are the primary advantages of this thiourea-catalyzed method over traditional phosphorus-based routes?
A: This method eliminates the need for stoichiometric amounts of toxic phosphorus reagents like triphenylphosphine and avoids the use of pyridine. This results in significantly reduced waste generation, simpler product isolation without complex heavy metal or phosphorus removal steps, and a much cleaner reaction profile suitable for GMP manufacturing.
Q: Which brominating agents are compatible with this catalytic system?
A: The patent specifies that both N-bromosuccinimide (NBS) and dibromohydantoin (DBH) are effective brominating agents when used in conjunction with the thiourea catalyst, offering flexibility in reagent sourcing and cost management for large-scale production.
Q: How is the final product purified to meet pharmaceutical standards?
A: The process utilizes a straightforward recrystallization step, typically using ethanol or ethanol-water mixtures, to achieve high purity. This avoids the need for column chromatography, making the process highly scalable and cost-effective for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-pivaloyloxy-5β,6β-epoxy-7-bromo-15β,16β-dimethylene-pregnane-17-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving hormonal therapies. Our technical team has thoroughly analyzed the potential of the thiourea-catalyzed route described in CN115947773A and is fully prepared to implement this advanced methodology. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-pivaloyloxy-5β,6β-epoxy-7-bromo-15β,16β-dimethylene-pregnane-17-one meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this green and efficient synthesis technology for your projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product quality and your bottom line.
