Scalable Production of Chiral Decalin Intermediates Using Abundant Triterpenoid Feedstocks
Scalable Production of Chiral Decalin Intermediates Using Abundant Triterpenoid Feedstocks
The pharmaceutical and fragrance industries have long sought efficient, scalable routes to chiral multi-substituted decahydro and octahydronaphthalene structural fragments, which serve as critical scaffolds for bioactive molecules and high-value aromatics. Patent CN101153030B introduces a groundbreaking methodology that transforms abundant oleanane and ursane type pentacyclic triterpenoids into these valuable chiral synthons. Unlike traditional semi-synthetic approaches that rely on scarce natural precursors or harsh oxidative conditions, this invention leverages a sophisticated photochemical ring-opening strategy. By utilizing widely available biomass-derived starting materials such as oleanolic acid and ursolic acid, the process addresses fundamental supply chain vulnerabilities while delivering high-purity intermediates suitable for complex drug synthesis and fine fragrance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of decalin and octahydronaphthalene derivatives has been plagued by significant economic and environmental hurdles. Conventional semi-synthetic routes often depend on precursors like sclareol or abietic acid, which are not only expensive due to limited natural availability but also require aggressive oxidative degradation steps. Traditional protocols frequently employ toxic transition metal chlorides or high-valent metal oxides such as Chromium(VI) and Manganese(VII) to cleave carbon-carbon bonds. These reagents generate substantial quantities of hazardous heavy metal waste, imposing severe burdens on waste treatment infrastructure and regulatory compliance. Furthermore, thermal cracking methods often suffer from poor selectivity, leading to complex product mixtures that are difficult and costly to purify, ultimately reducing the overall viability of these processes for large-scale commercial application.
The Novel Approach
The methodology disclosed in CN101153030B represents a paradigm shift by utilizing oleanane and ursane type triterpenoids, which are extremely abundant and cost-effective natural resources. The core innovation lies in the strategic functionalization of the C-ring followed by a controlled photochemical cleavage. Instead of relying on brute-force thermal degradation or toxic metal oxidants, this route converts the C-ring into an acyloxy-substituted cyclohexadiene system. Upon exposure to ultraviolet irradiation, this system undergoes a specific bond cleavage to open the ring, generating a triene intermediate that retains the crucial stereochemical information of the AB and DE rings. This approach not only simplifies the reaction sequence but also drastically improves the environmental profile of the synthesis by eliminating the need for stoichiometric amounts of toxic heavy metals.
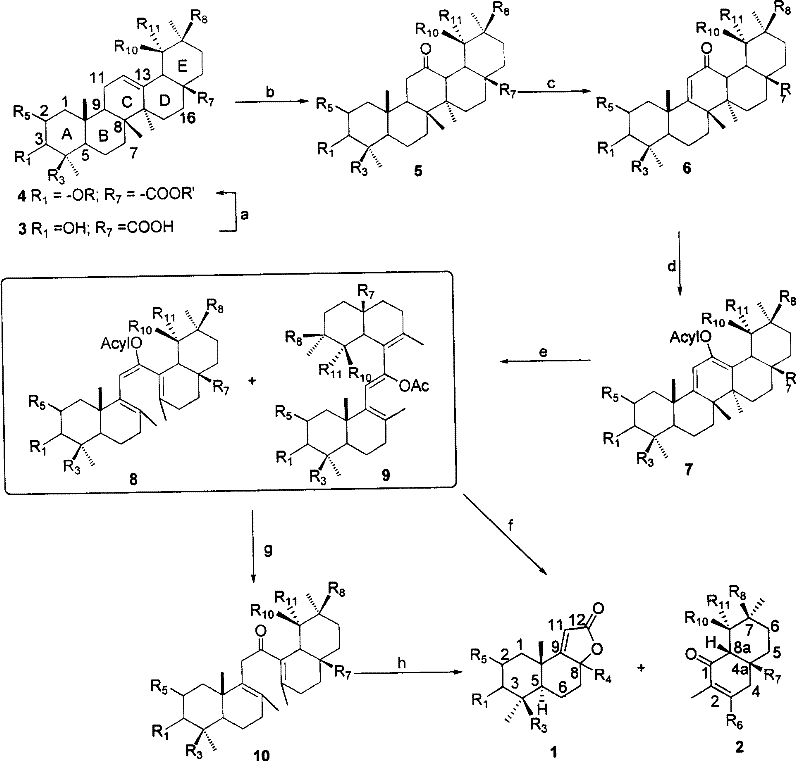
Mechanistic Insights into Photochemical C-Ring Cleavage
The mechanistic elegance of this synthesis centers on the photo-induced electrocyclic ring opening of the functionalized C-ring. After initial protection and oxidation steps convert the triterpenoid into a 12-position carbonyl compound, enol esterification creates a conjugated diene system sensitive to UV light. When irradiated, typically using a high-pressure mercury lamp, the bond between positions 8 and 14 undergoes homolytic cleavage. This photochemical event is highly specific, effectively dismantling the pentacyclic framework to reveal the bicyclic decalin or octalin core. The choice of reaction vessel material, such as quartz versus Pyrex glass, allows for fine-tuning of the wavelength transmission, thereby influencing the ratio of triene isomers produced. This level of control is critical for downstream applications where specific stereoisomers, such as 9-epi-Ambrox, are required for optimal biological or olfactory activity.
Following the photochemical step, the resulting triene intermediate undergoes oxidative cleavage to finalize the structure. The patent details the use of peracids, such as m-chloroperoxybenzoic acid (mCPBA), to cleave the remaining double bonds and install the necessary oxygen functionality. This step is pivotal in determining the final oxidation state of the product, yielding either decahydronaphthalene lactones or octahydronaphthalene ketones depending on the stoichiometry of the oxidant. The mechanism ensures that the multiple chiral centers inherent in the starting triterpenoid are preserved and correctly positioned in the final product. This preservation of chirality is a distinct advantage over total synthesis routes, which often struggle to establish multiple contiguous stereocenters with high fidelity without extensive resolution steps.
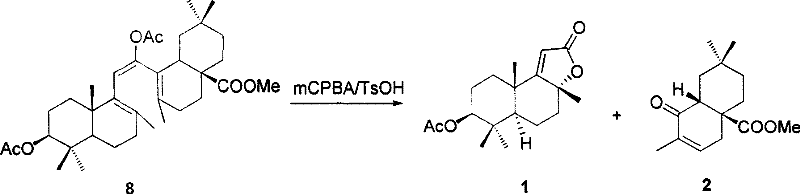
How to Synthesize Multi-substituted Hydrogenated Naphthalene Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these high-value intermediates. The process begins with the protection of hydroxyl groups and esterification of carboxyl groups on the starting triterpenoid to prevent side reactions during subsequent oxidation steps. Following this, the C-ring is activated through oxidation and dehydrogenation to form an alpha,beta-unsaturated ketone, which is then converted into an enol ester. The critical photochemical ring-opening step follows, after which oxidative cleavage yields the target compounds. For detailed operational parameters, reagent ratios, and specific workup procedures required to replicate this high-yielding transformation, please refer to the standardized synthesis guide below.
- Protect hydroxyl groups via acylation or etherification and esterify carboxyl groups on the starting triterpenoid.
- Oxidize the C-ring double bond to a carbonyl group and perform dehydrogenation to form an alpha,beta-unsaturated ketone.
- Convert the enol to an ester using acid anhydride, then subject the compound to UV irradiation to cleave the C-ring.
- Perform oxidative cleavage on the resulting triene using peracids to yield the final decahydro or octahydronaphthalene products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers tangible strategic benefits beyond mere technical feasibility. The primary advantage stems from the raw material base; oleanolic acid and ursolic acid are ubiquitous in the plant kingdom and can be sourced from agricultural by-products at a fraction of the cost of specialized terpenes like sclareol. This shift in feedstock fundamentally alters the cost structure of the final intermediate, providing a buffer against price volatility associated with niche natural extracts. Additionally, the elimination of toxic heavy metal catalysts simplifies the supply chain for reagents, removing the need for specialized handling and disposal protocols associated with chromium and manganese waste.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive, scarce starting materials with abundant, low-cost triterpenoids. By avoiding the use of precious or toxic metal oxidants, the process significantly reduces the cost of goods sold (COGS) related to reagent procurement. Furthermore, the simplified purification requirements, resulting from higher reaction selectivity and the absence of heavy metal residues, lead to substantial savings in downstream processing and waste management expenditures. These factors combine to create a more economically resilient manufacturing model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Relying on widely available biomass-derived starting materials mitigates the risk of supply disruptions that often plague the fragrance and pharmaceutical sectors when depending on specific plant harvests or complex fermentation processes. The robustness of the synthetic route, which utilizes standard organic transformations and photochemistry, ensures consistent production output. This reliability is crucial for maintaining continuous manufacturing schedules for high-demand products like Ambrox analogs or antiviral intermediates, ensuring that downstream customers receive their orders on time without unexpected delays caused by raw material shortages.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that can be adapted to large-scale photoreactors and standard oxidation vessels. From an environmental perspective, the avoidance of Cr(VI) and Mn(VII) oxidants aligns with increasingly stringent global regulations regarding heavy metal discharge. This compliance reduces the regulatory burden on manufacturing facilities and minimizes the environmental footprint of the production process. The ability to scale this green chemistry approach allows companies to meet growing market demand for sustainable ingredients while maintaining full adherence to environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of adopting this route for commercial production. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this chemistry into their existing pipelines.
Q: What are the primary advantages of using oleanolic acid over sclareol for decalin synthesis?
A: Oleanolic acid and related triterpenoids are extremely abundant natural resources with significantly lower costs compared to sclareol. Furthermore, this route avoids the use of toxic heavy metal oxidants like Chromium(VI) typically required in traditional sclareol degradation pathways.
Q: How does the photochemical step influence the stereochemistry of the final product?
A: The photochemical ring opening of the acyloxy-substituted cyclohexadiene intermediate allows for specific cleavage of the 8,14-bond. By controlling the reaction vessel material (quartz vs. Pyrex), manufacturers can influence the ratio of triene isomers, thereby managing the stereochemical outcome of the subsequent oxidative cleavage.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is simple, easy to implement, and capable of industrialization. The use of readily available starting materials and the elimination of complex purification steps associated with toxic metal waste contribute to its scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decahydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photochemical synthesis route described in CN101153030B for producing high-purity decahydronaphthalene and octahydronaphthalene intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch of chiral intermediates meets the exacting standards required for pharmaceutical and fine fragrance applications, delivering consistency that your downstream processes depend on.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this triterpenoid-based route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable, cost-effective supply of these critical chiral building blocks.
