Scalable Production of High-Purity Betulonic Acid via Mild IBX Oxidation Technology
Introduction to Advanced Betulonic Acid Synthesis
The pharmaceutical industry continuously seeks robust synthetic pathways for bioactive triterpenoids, particularly those serving as critical precursors for anti-cancer and anti-viral therapeutics. Patent CN101759759B introduces a transformative methodology for the preparation of Betulonic Acid, a pivotal intermediate in the synthesis of Betulinic Acid, which exhibits potent anti-tumor, anti-HIV, and immunostimulatory properties. This intellectual property details a sophisticated two-step oxidation protocol that fundamentally shifts the production paradigm from hazardous heavy metal chemistry to a more benign, high-efficiency organic oxidation system. By leveraging 2-Iodoxybenzoic acid (IBX) and Tetrabutylammonium Permanganate (Bu4NMnO4), this technology addresses the longstanding bottlenecks of yield loss and environmental toxicity associated with legacy methods. For R&D directors and procurement strategists, understanding this patent is crucial as it represents a viable pathway to securing a reliable betulonic acid supplier capable of meeting stringent purity specifications without the ecological footprint of chromium-based processes.
The Limitations of Conventional Methods vs. The Novel Approach
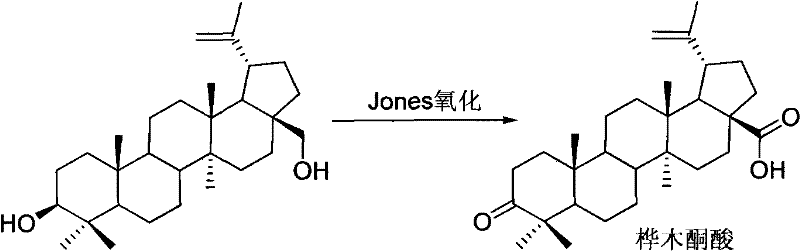
The Limitations of Conventional Methods
Historically, the industrial synthesis of Betulonic Acid has been plagued by reliance on archaic oxidation technologies that pose severe safety and environmental liabilities. The most prevalent traditional method involves Jones oxidation, utilizing chromium trioxide (CrO3) in concentrated sulfuric acid and acetone. While chemically effective, this approach suffers from catastrophic drawbacks including a mediocre yield range of merely 52% to 75%, necessitating extensive purification via column chromatography which further erodes overall throughput. More critically, the generation of hexavalent chromium waste creates a massive burden on wastewater treatment facilities and violates modern green chemistry principles, making it increasingly difficult to obtain regulatory approval for large-scale manufacturing. Alternative methods such as Swern oxidation require cryogenic conditions (often below -60°C) and strictly anhydrous environments, rendering them energetically expensive and operationally complex for multi-ton production campaigns. These conventional routes effectively cap the commercial viability of Betulonic Acid, creating supply chain fragility and inflated costs for downstream API manufacturers.
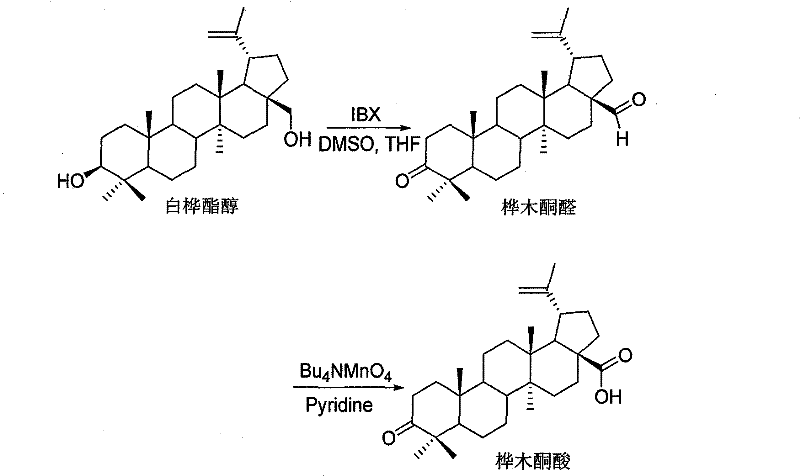
The Novel Approach
In stark contrast to these legacy constraints, the methodology disclosed in CN101759759B employs a tandem oxidation strategy that maximizes atom economy and operational simplicity. The process initiates with the selective oxidation of Betulin using IBX in solvents such as DMSO or THF, achieving a remarkable 90% yield of the intermediate Betulin Aldehyde under mild thermal conditions ranging from 0°C to 70°C. This intermediate is subsequently subjected to a second oxidation step using Tetrabutylammonium Permanganate (Bu4NMnO4), which efficiently converts the aldehyde moiety to the carboxylic acid without over-oxidizing other sensitive functional groups on the pentacyclic triterpene skeleton. This second step boasts an impressive 95% yield, culminating in an overall process yield of 85.5%, which significantly outperforms the background art. The elimination of cryogenic requirements and heavy metal catalysts not only simplifies the engineering controls needed for the reactor but also drastically reduces the cost of goods sold (COGS) by minimizing waste disposal fees and raw material consumption.
Mechanistic Insights into IBX and Phase-Transfer Permanganate Oxidation
The success of this synthetic route lies in the specific mechanistic advantages offered by the chosen oxidants. IBX acts as a hypervalent iodine reagent that facilitates the dehydrogenation of alcohols through a ligand exchange mechanism followed by reductive elimination. In the context of the Betulin substrate, IBX demonstrates exceptional chemoselectivity, capable of oxidizing the secondary hydroxyl group at the C-3 position to a ketone and the primary hydroxyl at C-28 to an aldehyde simultaneously or sequentially depending on stoichiometry, without inducing skeletal rearrangement or double bond migration which are common side reactions in acid-catalyzed oxidations. The use of polar aprotic solvents like DMSO enhances the solubility of the hypervalent iodine species, ensuring homogeneous reaction kinetics that are critical for consistent batch-to-batch reproducibility. Furthermore, the byproduct of IBX reduction is 2-iodobenzoic acid, which is non-toxic and easily separable from the organic product during the aqueous workup phase, thereby simplifying the impurity profile.
The second stage utilizes Tetrabutylammonium Permanganate (Bu4NMnO4), often referred to as purple benzene, which functions as a lipophilic oxidant. Unlike inorganic permanganates that require biphasic systems and phase transfer catalysts, Bu4NMnO4 is soluble in organic media such as pyridine or dichloromethane, allowing for a homogeneous oxidation of the aldehyde intermediate to the carboxylic acid. This homogeneity ensures that mass transfer limitations do not hinder the reaction rate, allowing the process to proceed rapidly at temperatures as low as 0°C to 50°C. The quaternary ammonium cation stabilizes the permanganate anion in the organic phase, facilitating a direct oxygen transfer to the carbonyl carbon. This mechanism avoids the formation of stubborn manganese dioxide emulsions that typically complicate the filtration and isolation steps in traditional permanganate oxidations, thus enhancing the overall process robustness and facilitating easier scale-up for commercial manufacturing.
How to Synthesize Betulonic Acid Efficiently
The practical implementation of this synthesis requires precise control over stoichiometry and temperature to maintain the high yields reported in the patent data. The process begins by dissolving Betulin in a suitable solvent system, followed by the controlled addition of IBX to manage the exotherm, ensuring the reaction temperature remains within the optimal 0°C to 70°C window. Following the isolation of the aldehyde intermediate, the second oxidation is performed by introducing Bu4NMnO4, where the disappearance of the characteristic purple color serves as a visual indicator of reaction progress. Detailed standardized operating procedures regarding specific solvent volumes, quenching protocols with sodium bisulfite, and chromatographic purification parameters are essential for replicating the 85.5% total yield in a GMP environment.
- Oxidize Betulin using 2-Iodoxybenzoic acid (IBX) in DMSO/THF/Toluene at 0-70°C to obtain Betulin Aldehyde with 90% yield.
- Further oxidize the intermediate Betulin Aldehyde using Tetrabutylammonium Permanganate (Bu4NMnO4) in Pyridine/DCM at 0-50°C.
- Quench with sodium bisulfite, extract with ethyl acetate, and purify via silica gel chromatography to achieve 95% yield for the second step.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this IBX-mediated synthesis route offers profound strategic advantages beyond mere chemical elegance. The primary value driver is the substantial cost reduction in pharmaceutical intermediates manufacturing achieved by eliminating the need for expensive and regulated heavy metal waste disposal associated with chromium-based methods. By replacing toxic reagents with organic-soluble oxidants that generate benign byproducts, the facility can operate with lower environmental compliance overheads and reduced insurance liabilities. Furthermore, the high overall yield of 85.5% means that less raw Betulin is required to produce the same amount of final product, directly lowering the material input costs per kilogram of Betulonic Acid. This efficiency gain is compounded by the ability to use simpler isolation techniques, such as extraction and crystallization, rather than resource-intensive preparative chromatography, which further drives down operational expenditures.
- Cost Reduction in Manufacturing: The transition away from Jones oxidation eliminates the capital and operational expenses related to handling corrosive sulfuric acid and toxic chromium waste streams. The use of IBX and Bu4NMnO4 allows for milder reaction conditions that reduce energy consumption for heating or cooling, while the high selectivity minimizes the formation of byproducts that would otherwise require costly purification steps. This streamlined process flow results in a significantly lower cost of goods, enabling more competitive pricing for downstream API producers without sacrificing margin.
- Enhanced Supply Chain Reliability: Betulin, the starting material, is abundantly available as a natural extract from birch bark, where it constitutes up to 25% of the dry weight, ensuring a stable and scalable feedstock supply unlike rare synthetic precursors. The robustness of the oxidation steps, which tolerate a broader range of operating temperatures and do not require strictly anhydrous conditions like Swern oxidation, reduces the risk of batch failures due to minor process deviations. This reliability translates to consistent lead times and the ability to ramp up production volume rapidly in response to market demand fluctuations for anti-cancer therapeutics.
- Scalability and Environmental Compliance: The homogeneous nature of the Bu4NMnO4 oxidation in organic solvents facilitates straightforward scale-up from laboratory to pilot and commercial scales without the mixing and heat transfer issues common in heterogeneous slurry reactions. The absence of heavy metals simplifies the regulatory filing process for the drug master file (DMF), as residual metal limits are easier to meet, accelerating time-to-market for new drug applications. Additionally, the greener profile of the synthesis aligns with corporate sustainability goals, reducing the carbon footprint and hazardous waste generation associated with the manufacturing of complex triterpenoids.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Betulonic Acid using this patented methodology. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, focusing on yield optimization, safety profiles, and scalability factors. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the IBX/Bu4NMnO4 route superior to traditional Jones oxidation for Betulonic Acid?
A: Traditional Jones oxidation uses toxic chromium trioxide and concentrated sulfuric acid, resulting in heavy metal waste and lower yields (52-75%). The novel IBX route avoids heavy metals, operates under milder conditions, and achieves a total yield of 85.5%.
Q: What are the scalability advantages of using Tetrabutylammonium Permanganate?
A: Unlike aqueous potassium permanganate which requires phase transfer catalysts and generates manganese dioxide sludge, Bu4NMnO4 is soluble in organic solvents, allowing for homogeneous reaction conditions, easier workup, and simplified waste treatment suitable for industrial scale-up.
Q: Is Betulin a sustainable starting material for this synthesis?
A: Yes, Betulin is abundantly available as it constitutes up to 25% of dry birch bark weight. This high natural abundance ensures a stable, low-cost supply chain compared to extracting the final acid directly from plant sources where content is only 0.025%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Betulonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology and antiviral medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering Betulonic Acid with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical grade materials. Our facility is equipped to handle the specific solvent systems and oxidation chemistries described in CN101759759B, guaranteeing a supply chain that is both resilient and compliant with international environmental regulations.
We invite global partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain economics. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential financial benefits of switching to this greener, higher-yield process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your development programs proceed without interruption due to material shortages.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
