Advanced Magnesium-Lithium Bimetallic Catalysis for High-Purity Pharmaceutical Intermediates
Introduction to Next-Generation Cyanohydrin Synthesis
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more sustainable catalytic processes. A pivotal advancement in this domain is detailed in Chinese Patent CN108912161B, which discloses a novel preparation method for cyanohydrin silyl ether compounds. This technology represents a paradigm shift from reliance on scarce and toxic transition metals to the utilization of an earth-abundant magnesium-lithium bimetallic system stabilized by a tridentate pincer ligand. For R&D directors and procurement strategists in the pharmaceutical sector, this innovation offers a compelling solution to longstanding challenges regarding catalyst toxicity, cost volatility, and environmental compliance. The patent highlights a catalytic system that not only achieves near-quantitative conversion rates but also operates under exceptionally mild conditions, thereby redefining the efficiency standards for synthesizing key pharmaceutical intermediates.
Cyanohydrin derivatives serve as indispensable building blocks in the synthesis of alpha-hydroxy acids, alpha-amino acids, and beta-amino alcohols, which are critical motifs in numerous active pharmaceutical ingredients (APIs). Traditionally, the synthesis of these valuable intermediates has been plagued by the use of heavy metal catalysts such as Ruthenium, Manganese, Vanadium, Titanium, and Platinum. While effective, these metals introduce severe liabilities including high raw material costs, complex waste disposal requirements due to toxicity, and stringent regulatory hurdles regarding residual metal limits in final drug products. The technology described in CN108912161B directly addresses these pain points by introducing a non-toxic, highly active alternative that aligns perfectly with the principles of green chemistry, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the silylcyanation of aldehydes to form cyanohydrin silyl ethers has relied heavily on transition metal catalysis. These conventional methods, while chemically viable, suffer from inherent structural and economic deficiencies that hinder large-scale industrial adoption. The primary drawback is the reliance on precious or toxic metals like Ru, Pt, and Ti, which are subject to extreme price volatility and supply chain insecurity. Furthermore, the toxicity of these metals necessitates rigorous and expensive purification steps to ensure that residual metal levels in the final API meet strict regulatory guidelines (often in the ppm or ppb range). This downstream processing adds significant time and cost to the manufacturing timeline. Additionally, many traditional protocols require harsh reaction conditions, including elevated temperatures or the use of hazardous solvents, which increases energy consumption and operational risk, making them less attractive for modern, sustainability-focused manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach utilizing the NCN tridentate pincer ligand stabilized magnesium-lithium bimetallic compound offers a streamlined and economically superior alternative. This method leverages the synergistic effects of dual metal centers to activate both the nucleophile (trimethylsilyl cyanide) and the electrophile (aldehyde) simultaneously. As illustrated in the general reaction scheme below, the process facilitates the efficient conversion of diverse aldehyde substrates into their corresponding silyl ethers with remarkable speed and selectivity.
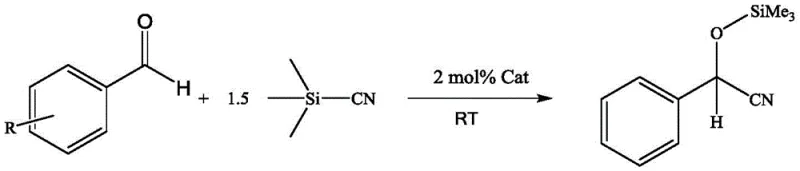
The reaction proceeds smoothly at room temperature in chloroform, eliminating the need for energy-intensive heating or cooling cycles. The patent data indicates that this system is universally applicable to both aromatic and aliphatic aldehydes, achieving conversion rates approaching 100%. By replacing expensive transition metals with abundant magnesium and lithium, the process drastically reduces the raw material cost basis. Moreover, the absence of toxic heavy metals simplifies the workup procedure, potentially allowing for direct crystallization or simple extraction, thereby enhancing the overall throughput and reducing the environmental footprint of the synthesis.
Mechanistic Insights into NCN Tridentate Pincer Mg-Li Catalysis
For the technical audience, understanding the mechanistic underpinnings of this catalyst is crucial for appreciating its superior performance. The core of this innovation lies in the unique structure of the catalyst, depicted below, where a central magnesium atom is coordinated by an NCN tridentate pincer ligand and associated with a lithium cation.
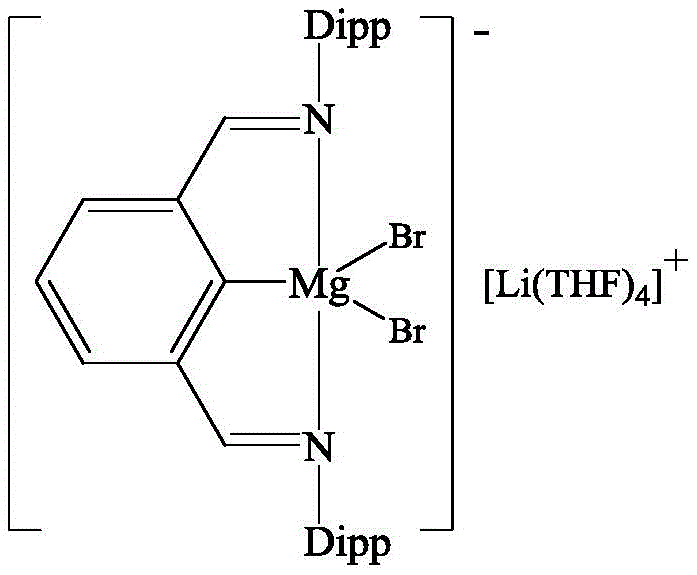
The tridentate nature of the ligand provides exceptional thermal and chemical stability to the magnesium center, preventing catalyst decomposition during the reaction cycle. Mechanistically, the electron-withdrawing character of the three donor atoms (Carbon and two Nitrogens) bonded to the magnesium reduces the electron density on the metal center. This renders the magnesium more electrophilic, enhancing its ability to coordinate with and activate the lone pair electrons on the nitrile group of the trimethylsilyl cyanide. Simultaneously, the lithium ion plays a critical cooperative role by coordinating with the carbonyl oxygen of the aldehyde substrate. Due to its strong polarization capability, the lithium ion significantly increases the positive charge character of the carbonyl carbon, making it much more susceptible to nucleophilic attack. This dual-activation mechanism—where Mg activates the cyanide source and Li activates the aldehyde—lowers the activation energy of the rate-determining step, resulting in the observed high reaction rates and yields even at ambient temperatures.
This synergistic bimetallic effect also contributes to the high selectivity observed in the patent examples. Unlike monometallic Lewis acids that might promote side reactions such as aldol condensations or polymerization of the aldehyde, the precise geometric arrangement enforced by the pincer ligand ensures that the reaction proceeds exclusively through the desired silylcyanation pathway. This specificity is vital for maintaining high purity profiles in pharmaceutical intermediates, as it minimizes the formation of difficult-to-remove impurities. The robustness of this catalytic cycle allows for consistent performance across a wide variety of electronic environments on the aldehyde ring, from electron-rich methoxy groups to electron-deficient nitro and halogen substituents, demonstrating the versatility required for complex API synthesis.
How to Synthesize Cyanohydrin Silyl Ethers Efficiently
Implementing this technology in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The protocol outlined in the patent emphasizes the importance of maintaining anhydrous and anaerobic conditions to preserve the activity of the sensitive organometallic catalyst. The reaction is typically conducted in a glovebox or under a rigorous nitrogen atmosphere using dry chloroform as the solvent. The molar ratio of catalyst to substrate is optimized at 2 mol%, which strikes a balance between catalytic efficiency and cost-effectiveness. The reaction time is notably short, ranging from 15 minutes to 6 hours depending on the steric and electronic nature of the aldehyde substrate, allowing for rapid turnaround times in process development.
- Prepare the reaction system under anhydrous and oxygen-free conditions, typically within a nitrogen-protected glovebox, ensuring all glassware and solvents like chloroform are strictly dry.
- Introduce the NCN tridentate pincer ligand stabilized magnesium-lithium bimetallic catalyst into the reaction vessel containing the solvent, followed by the addition of trimethylsilyl cyanide (TMSCN).
- Add the aldehyde substrate (aromatic or aliphatic) to the mixture and stir at room temperature for 15 minutes to 6 hours until conversion is complete, monitored via NMR spectroscopy.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Mg-Li catalytic technology offers profound strategic advantages for procurement managers and supply chain heads. The most immediate benefit is the drastic reduction in raw material costs associated with catalyst procurement. By eliminating the dependence on precious metals like Platinum and Ruthenium, which are subject to geopolitical supply risks and fluctuating market prices, manufacturers can stabilize their cost of goods sold (COGS). Furthermore, the non-toxic nature of magnesium and lithium removes the regulatory burden associated with heavy metal residues. This translates to simplified quality control workflows and reduced analytical testing costs, as there is no need for sophisticated ICP-MS monitoring for trace toxic metals in the final product. The elimination of expensive metal scavenging resins or complex purification steps further drives down the operational expenditure.
Supply chain reliability is significantly enhanced by the use of earth-abundant elements. Magnesium and lithium are widely available globally, ensuring a secure and continuous supply of catalyst precursors compared to the constrained supply chains of rare transition metals. This security of supply is critical for long-term API production contracts where continuity is paramount. Additionally, the mild reaction conditions (room temperature) and the use of common solvents like chloroform reduce the energy load on the manufacturing facility. Lower energy consumption not only reduces utility costs but also aligns with corporate sustainability goals and carbon reduction targets. The simplicity of the reaction setup also implies easier scale-up; processes that run efficiently at room temperature with low catalyst loading are generally more robust when transitioning from kilogram to multi-ton scales, reducing the risk of batch failures and ensuring consistent delivery timelines to customers.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with an earth-abundant magnesium-lithium system fundamentally alters the cost structure of cyanohydrin production. The removal of precious metals eliminates a major variable cost component, while the high catalytic activity (requiring only 2 mol% loading) ensures that material usage is minimized. Furthermore, the absence of toxic metals negates the need for costly downstream purification steps such as metal scavenging or extensive chromatography, leading to substantial savings in both materials and labor. The mild reaction conditions also result in lower energy consumption, contributing to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: Relying on magnesium and lithium mitigates the supply chain risks associated with critical raw materials. Unlike platinum group metals which are often sourced from geopolitically unstable regions, Mg and Li have diverse and robust global supply chains. This diversity ensures that production schedules are not disrupted by raw material shortages. The stability of the catalyst and the simplicity of the reaction conditions also contribute to higher batch success rates, ensuring that delivery commitments to downstream pharmaceutical clients are met consistently. This reliability is a key differentiator in the competitive landscape of contract development and manufacturing organizations (CDMOs).
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at ambient temperature and pressure, removing the engineering complexities associated with high-pressure or high-temperature reactors. From an environmental standpoint, the non-toxic nature of the catalyst aligns with increasingly stringent global environmental regulations regarding heavy metal discharge and waste management. The "green" profile of this chemistry facilitates easier permitting for new manufacturing lines and reduces the liability associated with hazardous waste disposal. This compliance advantage future-proofs the manufacturing asset against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented catalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the operational realities and advantages of the Mg-Li bimetallic system.
Q: What are the primary advantages of the Mg-Li bimetallic catalyst over traditional transition metal catalysts?
A: Unlike traditional catalysts based on Ruthenium, Manganese, or Platinum which are toxic and expensive, the Mg-Li bimetallic system utilizes earth-abundant, non-toxic metals. It operates under mild room temperature conditions with high selectivity and eliminates the need for costly heavy metal removal processes in downstream purification.
Q: What is the substrate scope for this catalytic silylcyanation reaction?
A: The patented method demonstrates exceptional versatility, effectively catalyzing both aromatic aldehydes (including those with electron-donating or withdrawing groups like halogens, nitro, and methoxy) and aliphatic aldehydes. The reaction achieves nearly 100% conversion across this broad range of substrates.
Q: How does the catalyst loading compare to conventional methods?
A: The process requires a catalyst loading of only 2 mol% relative to the substrate. This low loading, combined with the high turnover efficiency and the ability to run reactions at room temperature, significantly reduces the overall material cost and energy consumption compared to high-temperature or high-loading transition metal protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanohydrin Silyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Mg-Li bimetallic catalytic technology described in CN108912161B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly integrate such innovative green chemistry solutions into our manufacturing portfolio. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the specific analytical requirements of complex intermediates, guaranteeing that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite procurement leaders and R&D directors to explore how this advanced catalytic method can optimize your supply chain and reduce manufacturing costs. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring safer, more sustainable, and cost-effective chemical solutions to your pipeline, securing your supply chain with the reliability and quality that NINGBO INNO PHARMCHEM is known for.
