Scalable Nickel-Catalyzed Synthesis of Fluorinated Alcohols for Advanced Pharmaceutical Intermediates
Scalable Nickel-Catalyzed Synthesis of Fluorinated Alcohols for Advanced Pharmaceutical Intermediates
The strategic incorporation of fluorine atoms into organic molecules has become a cornerstone of modern medicinal chemistry, profoundly enhancing the metabolic stability, lipophilicity, and bioavailability of active pharmaceutical ingredients (APIs). Patent CN112047809A discloses a groundbreaking preparation method for alcohol compounds containing fluorine atoms, addressing a critical gap in synthetic methodology where direct preparation routes were previously unreported or inefficient. This technology leverages a nickel-catalyzed reductive coupling between halogenated alkanes (specifically trifluoroolefins) and epoxides in the presence of zinc powder, offering a robust pathway to valuable fluorinated building blocks. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in accessing complex fluorinated scaffolds with high efficiency and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fluorine-containing alcohol compounds has been fraught with significant chemical and operational challenges that hinder efficient commercial scale-up of complex intermediates. Conventional routes often rely on harsh reagents such as organolithiums or Grignard reagents which are extremely sensitive to moisture and air, necessitating rigorous anhydrous conditions and cryogenic temperatures that drastically increase energy consumption and safety risks. Furthermore, existing methods frequently suffer from poor functional group tolerance, requiring extensive protecting group manipulations that lengthen the synthetic sequence and reduce overall atom economy. The lack of direct methods to couple fluorinated olefins with epoxides has forced chemists to utilize multi-step sequences that generate substantial waste and drive up the cost of goods, creating bottlenecks in the supply chain for high-purity intermediates.
The Novel Approach
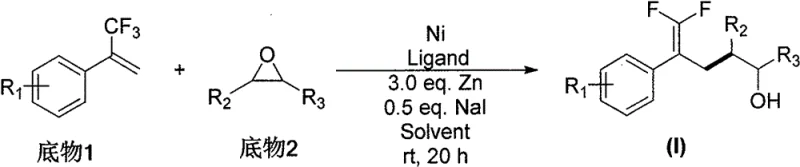
The novel approach detailed in CN112047809A revolutionizes this landscape by introducing a nickel-catalyzed system that operates under remarkably mild conditions, typically at 30°C, eliminating the need for energy-intensive cooling or heating protocols. This method utilizes a catalytic system comprising NiBr2·diglyme and a bipyridine ligand, activated by zinc powder, to facilitate the reductive coupling of trifluoroolefins with epoxides directly. As illustrated in the general reaction scheme below, this transformation proceeds with exceptional efficiency, achieving isolated yields as high as 93% in optimized examples while maintaining 100% product purity after standard purification. The simplicity of the feeding mode and the tolerance to ambient conditions significantly lower the barrier for entry, making this a superior choice for cost reduction in API manufacturing compared to legacy technologies.
Mechanistic Insights into Nickel-Catalyzed Reductive Coupling
The core of this technological breakthrough lies in the intricate catalytic cycle mediated by the nickel species, which enables the activation of the carbon-fluorine bond adjacent to the olefinic system without defluorination. The mechanism likely initiates with the reduction of the Ni(II) precatalyst to a low-valent Ni(0) or Ni(I) species by the zinc powder, which then undergoes oxidative addition or radical coordination with the trifluoroolefin substrate. This generates a fluorinated alkyl-nickel intermediate that is sufficiently nucleophilic to attack the electrophilic epoxide ring, triggering a ring-opening event that installs the hydroxyl functionality with precise regiocontrol. The presence of additives such as sodium iodide and triethylamine hydrochloride plays a crucial role in modulating the reactivity of the nickel center and stabilizing the transition states, ensuring that the reaction proceeds smoothly to afford the desired fluorinated alcohol product with high fidelity.
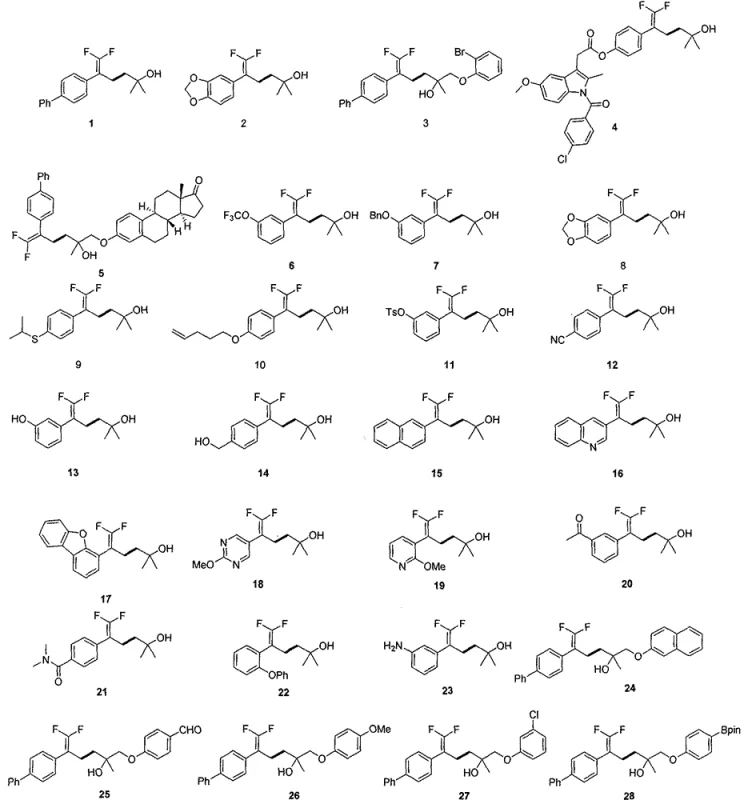
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions such as homocoupling of the olefin or polymerization of the epoxide, which are common pitfalls in radical-mediated processes. The specific ligand environment provided by 4,4'-di-tert-butyl-2,2'-bipyridine creates a steric and electronic pocket that favors the cross-coupling pathway over competing decomposition channels. This high level of chemoselectivity is critical for pharmaceutical applications, as it reduces the burden on downstream purification processes and ensures that the final material meets stringent purity specifications required for clinical candidates. The ability to tolerate diverse functional groups on both the olefin and epoxide partners further underscores the robustness of this catalytic manifold, allowing for the late-stage functionalization of complex molecular architectures.
How to Synthesize Fluorinated Alcohols Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the reagents and the order of addition to maximize yield and reproducibility. The patent outlines a standardized protocol where the nickel catalyst, ligand, zinc powder, and additives are pre-mixed in a polar aprotic solvent such as N,N-dimethylacetamide (DMAc) before the introduction of the substrates. This pre-activation step ensures that the catalytic species is fully generated and ready to engage with the reactants immediately upon mixing. While the specific molar ratios and reaction times can be adjusted based on the specific substrates used, the general procedure provides a reliable framework for accessing a wide array of fluorinated alcohol derivatives. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided in the section below.
- Prepare the catalytic system by mixing NiBr2·diglyme (10 mol%), ligand (12 mol%), Zinc powder (3.0 eq), NaI (0.5 eq), and Et3N·HCl (1.2 eq) in DMAc solvent under inert atmosphere.
- Add the trifluoroolefin substrate and epoxide substrate sequentially to the reaction mixture and stir at 30°C for approximately 10 hours.
- Quench the reaction with ethyl acetate, filter off solid residues through a short silica plug, and purify the crude product via column chromatography to obtain high-purity fluorinated alcohols.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed technology translates into tangible strategic benefits that extend far beyond simple yield improvements. The shift from precious metal catalysts to earth-abundant nickel significantly reduces the raw material costs associated with catalysis, while the mild reaction conditions lower the utility costs related to heating and cooling infrastructure. Moreover, the operational simplicity of the process, which does not require glovebox techniques or strictly anhydrous solvents, reduces the training burden on operators and minimizes the risk of batch failures due to environmental exposure. These factors collectively contribute to a more resilient and cost-effective supply chain for fluorinated intermediates, ensuring consistent availability for downstream drug manufacturing processes.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts in favor of nickel represents a direct and substantial cost saving in the bill of materials for every kilogram produced. Additionally, the high atom economy of the coupling reaction minimizes waste generation, reducing the costs associated with waste disposal and solvent recovery. The ability to achieve high yields (up to 93%) with minimal byproduct formation means that less starting material is required to produce the same amount of API, further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate ambient air and moisture to a significant degree, mitigates the risk of supply disruptions caused by sensitive reagent degradation or logistical delays in obtaining ultra-dry solvents. The use of commercially available and stable starting materials, such as trifluoroolefins and simple epoxides, ensures that the supply chain is not dependent on exotic or hard-to-source precursors. This stability allows for better inventory planning and reduces the lead time for high-purity intermediates, enabling faster response to market demands.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic profile being manageable at 30°C, avoiding the thermal runaway risks associated with highly exothermic organometallic reactions. The use of zinc powder as a reductant generates benign zinc salts as byproducts, which are easier to handle and dispose of compared to the heavy metal waste streams generated by other catalytic systems. This alignment with green chemistry principles facilitates regulatory approval and supports corporate sustainability goals, making it an attractive option for environmentally conscious manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorinated alcohol synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows and for assessing the quality profile of the resulting intermediates.
Q: What are the key advantages of this nickel-catalyzed method over traditional fluorination techniques?
A: This method utilizes earth-abundant nickel instead of expensive precious metals, operates at mild temperatures (30°C), and tolerates air and moisture better than many organolithium or Grignard-based fluorination routes, significantly simplifying operational complexity.
Q: What is the functional group compatibility of this synthesis route?
A: The reaction demonstrates excellent compatibility with various functional groups including esters, ethers, halides, and heterocycles, allowing for the direct synthesis of complex drug intermediates without extensive protecting group strategies.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of simple feeding modes, non-sensitive reagents, and mild reaction conditions (30°C) makes this process highly scalable and safe for industrial manufacturing, reducing the need for specialized cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Alcohols Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the nickel-catalyzed synthesis described in CN112047809A for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fluorinated alcohol meets the exacting standards required for global drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your path to market with our expertise in fluorine chemistry and process development.
