Advanced Synthesis of Naphthoxazole Phosphine Oxide Ligands for Catalytic Applications
The landscape of organometallic catalysis is continually evolving, driven by the demand for more efficient and sustainable ligand systems. A significant breakthrough in this domain is detailed in patent CN111825716B, which discloses a novel class of naphthoxazole phosphine oxide compounds and their streamlined synthesis. These compounds serve as precursors to advanced P,N-bidentate ligands, often referred to as PHOX ligands, which are critical for asymmetric catalysis in the pharmaceutical and fine chemical sectors. The patent outlines a robust, one-pot methodology that utilizes 1,4-naphthoquinone, diphenyl(dialkyl)phosphine oxide, and imines as key building blocks. By employing copper acetate as a dual-function catalyst and oxidant, this process achieves high yields under relatively mild conditions compared to historical precedents. For R&D directors and procurement specialists seeking reliable naphthoxazole phosphine oxide supplier partnerships, this technology represents a pivotal shift towards cost-effective and scalable ligand manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxazole ring, a fundamental skeleton in many bioactive molecules and ligands, has been fraught with synthetic challenges. Traditional multi-step synthesis methods often necessitate the use of highly reactive and hazardous reagents such as n-butyllithium, zinc chloride, or concentrated hydrochloric acid. These conditions not only pose significant safety risks in a manufacturing environment but also result in poor atom economy and lower overall yields due to the accumulation of losses across multiple isolation steps. Furthermore, the requirement for stringent anhydrous conditions and cryogenic temperatures for organolithium reagents drastically increases energy consumption and operational complexity. For a supply chain head, these factors translate into higher production costs, longer lead times, and increased difficulty in maintaining consistent quality control during the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
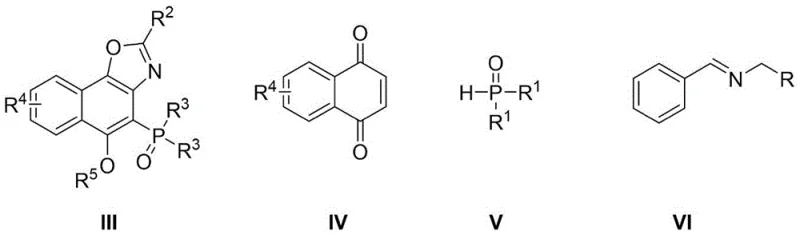
In stark contrast, the methodology described in CN111825716B offers a transformative one-pot strategy that simplifies the synthetic pathway considerably. The process initiates with the reaction of 1,4-naphthoquinone and diphenyl(dialkyl)phosphine oxide in toluene under a nitrogen atmosphere at 120°C. Subsequently, the imine and copper acetate are introduced directly into the same vessel, allowing the oxidative cyclization to proceed efficiently without intermediate workups. This telescoped approach not only reduces solvent usage and waste generation but also significantly shortens the total processing time. The ability to utilize commercially available starting materials like 1,4-naphthoquinone and various substituted imines enhances the versatility of the route, allowing for the rapid generation of diverse ligand libraries. This innovation directly addresses the need for cost reduction in electronic chemical manufacturing and pharmaceutical intermediate production by streamlining the workflow.
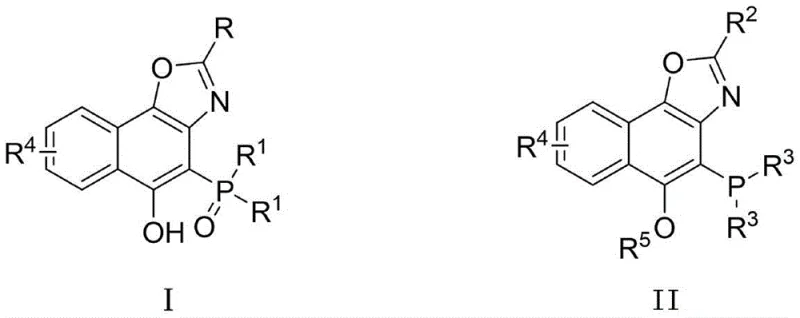
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this synthetic advancement lies in the elegant utilization of copper acetate, which functions simultaneously as a Lewis acid catalyst and an oxidant. The mechanism likely involves the initial coordination of the copper species to the phosphine oxide and the quinone system, facilitating a nucleophilic attack or radical coupling process that constructs the C-P and C-N bonds requisite for the naphthoxazole framework. The oxidative nature of the copper salt promotes the aromatization of the intermediate dihydro-species, driving the equilibrium towards the stable oxazole product. This dual role eliminates the need for external stoichiometric oxidants, which are often expensive or generate toxic byproducts. From a purity perspective, the mild reaction temperature of 120°C in toluene helps minimize thermal degradation and side reactions that are common in harsher acidic or basic environments. Consequently, the resulting crude product exhibits a cleaner impurity profile, reducing the burden on downstream purification processes such as column chromatography or recrystallization.
Furthermore, the structural diversity achievable through this method is substantial, as evidenced by the wide range of substituents tolerated on the imine and phosphine oxide components. The patent details successful syntheses with R groups ranging from electron-rich species like 4-methoxyphenyl to electron-deficient moieties such as 4-trifluoromethylphenyl and halogens like bromine and iodine. This tolerance is crucial for fine-tuning the steric and electronic properties of the final ligand to optimize its performance in specific catalytic transformations. The subsequent conversion of the phosphine oxide (Compound I) to the phosphine ligand (Compound II) via alkylation and reduction further demonstrates the robustness of the scaffold. The reduction step using phenylsilane and acetic acid is particularly noteworthy for its efficiency in converting the P=O bond to P-H or P-C bonds without affecting the sensitive oxazole ring, ensuring high-purity OLED material or catalyst precursor standards are met.
How to Synthesize Naphthoxazole Phosphine Oxide Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling, making it highly attractive for process chemistry teams. The protocol begins by charging a reaction vessel with 1,4-naphthoquinone and the appropriate diphenyl(dialkyl)phosphine oxide in toluene, followed by heating under nitrogen. After an initial incubation period, the imine and copper catalyst are added, and the mixture is maintained at elevated temperature to drive the cyclization to completion. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and timing required to achieve the reported high yields. This level of procedural clarity ensures that the transition from laboratory discovery to pilot plant production can be managed with minimal technical risk, supporting the commercial scale-up of complex pharmaceutical intermediates.
- React 1,4-naphthoquinone and diphenyl(dialkyl)phosphine oxide in toluene at 120°C under nitrogen for 12 hours.
- Add imine and copper acetate to the mixture and continue stirring at 120°C for another 12 hours to obtain Compound I.
- Perform alkylation with methyl/ethyl iodide followed by reduction with phenylsilane to convert Compound I into the novel ligand Compound II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers compelling economic and logistical benefits. The shift from multi-step, hazardous chemistries to a streamlined one-pot process fundamentally alters the cost structure of ligand production. By eliminating the need for specialized equipment required for cryogenic reactions or handling pyrophoric reagents, capital expenditure and operational overheads are significantly reduced. Moreover, the use of toluene as a solvent, a common and recoverable industrial solvent, simplifies waste management and solvent recycling protocols. These factors collectively contribute to substantial cost savings in the manufacturing of high-value catalytic ligands, allowing end-users to secure a more competitive pricing structure for their final API or specialty chemical products.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as n-butyllithium and strong mineral acids removes a major cost driver from the bill of materials. Additionally, the one-pot nature of the reaction reduces the number of unit operations, thereby lowering labor costs and energy consumption associated with heating, cooling, and intermediate isolations. The high atom economy of the oxidative cyclization ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal fees. This logical deduction of cost benefits makes the process highly attractive for large-scale production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals like 1,4-naphthoquinone and simple imines mitigates the risk of raw material shortages that often plague supply chains dependent on exotic or custom-synthesized precursors. The robustness of the reaction conditions, which tolerate a wide range of functional groups, ensures consistent batch-to-batch quality even with slight variations in feedstock quality. This reliability is essential for maintaining continuous production schedules in the fast-paced pharmaceutical and agrochemical industries, where delays can have cascading effects on downstream drug development timelines.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, which simplifies the engineering requirements for scaling up from kilogram to tonne quantities. The absence of heavy metal contaminants beyond the catalytic copper, which can be removed via standard workup procedures, aligns well with increasingly stringent environmental regulations regarding heavy metal residues in pharmaceutical products. The simplified waste stream, primarily consisting of organic solvents and copper salts, is easier to treat and dispose of compared to the complex mixtures generated by traditional multi-step syntheses, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this ligand system into their existing catalytic processes.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: The novel one-pot method eliminates the need for harsh reagents like n-butyllithium and strong acids, significantly improving operational safety and atom economy while maintaining high yields.
Q: What is the typical yield range for the naphthoxazole phosphine oxide compounds?
A: According to the patent data, the synthesis of Compound I achieves yields ranging from moderate to excellent, with specific examples demonstrating yields up to 86% depending on the substituents.
Q: Can this ligand be used for large-scale industrial amination reactions?
A: Yes, the ligand derived from these compounds has been proven effective for the efficient amination of amines or nitrobenzenes with alcohols, showing high potential for industrial scalability due to the robust synthesis route.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthoxazole Phosphine Oxide Supplier
The technological potential of the naphthoxazole phosphine oxide scaffold is immense, offering a versatile platform for developing next-generation catalysts. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to bulk manufacturing is seamless. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that employ state-of-the-art analytical techniques to verify the identity and purity of every batch. We understand that in the high-stakes world of catalytic ligand supply, consistency is paramount, and our infrastructure is designed to deliver exactly that.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer picture of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than assumptions. Let us collaborate to bring this efficient and sustainable chemistry to your production line.
