Scalable Production of Chiral 3,3-Difluoro-4-Aminopyrrolidine Derivatives via Tartaric Acid Resolution
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed fluorinated heterocycles at the forefront of medicinal chemistry innovation. Specifically, the chiral 3,3-difluoro-4-aminopyrrolidine scaffold has emerged as a critical structural motif in the development of next-generation therapeutics, including LRRK2 inhibitors for Parkinson's disease and PI3K-delta inhibitors for immune disorders. However, the commercial viability of these drug candidates often hinges on the ability to produce their chiral intermediates efficiently and economically. Patent CN113429328A addresses this critical bottleneck by disclosing a robust preparation method for chiral 3,3-difluoro-4-aminopyrrolidine derivatives. This technology represents a significant paradigm shift from traditional, cost-prohibitive separation techniques to a scalable diastereomeric salt resolution process. By leveraging the stereochemical discrimination capabilities of inexpensive chiral tartaric acid, this method offers a viable pathway for the reliable pharmaceutical intermediate supplier to deliver high-purity materials essential for clinical and commercial drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of single enantiomers for complex fluorinated pyrrolidines has relied heavily on chiral preparative chromatography. While this technique boasts high separation efficiency and excellent selectivity in a laboratory setting, it presents formidable challenges when transitioning to industrial scales. The primary drawback lies in the exorbitant cost associated with chiral stationary phases and the specialized equipment required to operate them effectively. Furthermore, chromatographic processes are inherently batch-limited and struggle to achieve the continuous throughput necessary for multi-kilogram or ton-scale production. The operational complexity, combined with the significant solvent consumption and waste generation, renders chiral chromatography economically unfeasible for the cost reduction in API manufacturing that modern pharmaceutical supply chains demand. Consequently, many promising drug candidates containing this scaffold face delayed development timelines due to the lack of a scalable synthetic route.
The Novel Approach
In stark contrast to chromatographic limitations, the methodology described in CN113429328A employs a classical yet highly optimized diastereomeric salt resolution strategy. This approach capitalizes on the distinct physical properties, specifically solubility and crystallinity, of diastereomeric salts formed between the racemic amine and a chiral acid. By selecting chiral tartaric acid—either the D- or L-isomer—as the resolving agent, the process converts the enantiomeric mixture into separable diastereomeric salts. The elegance of this method lies in its simplicity: it utilizes standard crystallization unit operations that are ubiquitous in fine chemical plants, thereby eliminating the need for exotic equipment. This transition from chromatography to crystallization not only drastically simplifies the operational workflow but also aligns perfectly with the requirements for commercial scale-up of complex pharmaceutical intermediates, ensuring a stable and continuous supply of chiral building blocks.
Mechanistic Insights into Diastereomeric Salt Resolution
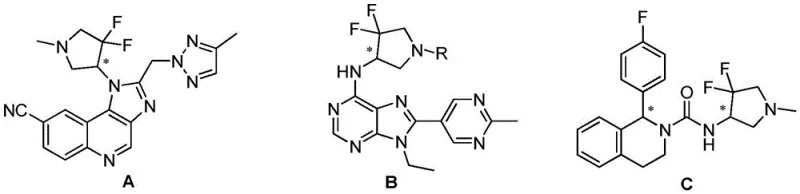
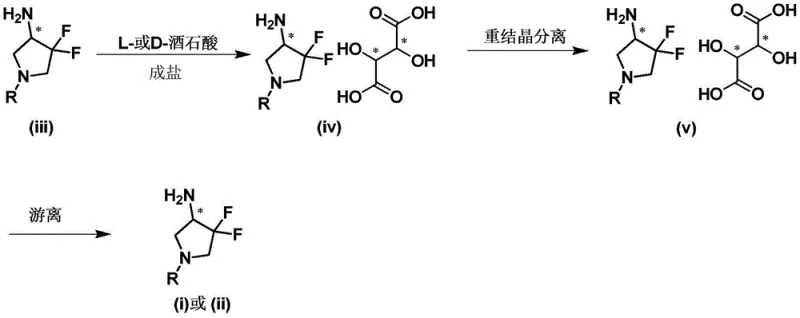
The core chemical principle driving this synthesis is the formation of diastereomeric ammonium tartrates through an acid-base reaction between the basic amino group of the pyrrolidine derivative and the carboxylic acid groups of tartaric acid. When a racemic mixture of 3,3-difluoro-4-aminopyrrolidine reacts with a single enantiomer of tartaric acid (e.g., D-tartaric acid), two distinct diastereomeric salts are generated: one comprising the (S)-amine and D-acid, and the other comprising the (R)-amine and D-acid. Although these salts share similar chemical connectivity, their three-dimensional spatial arrangements differ significantly, leading to divergent lattice energies and solvation characteristics. In a carefully selected solvent system, such as ethanol or ethyl acetate, one diastereomer exhibits markedly lower solubility than the other. This differential solubility is the thermodynamic driver that allows for the selective precipitation of the desired salt upon cooling, effectively enriching the solid phase with one enantiomer while leaving the other in the mother liquor.
Following the isolation of the enriched salt, the final step involves a straightforward alkaline dissociation to liberate the free chiral amine. By treating the purified tartrate salt with a base such as sodium hydroxide or potassium carbonate in an aqueous or biphasic system, the ammonium salt is neutralized. This shifts the equilibrium towards the free base form, which can then be extracted into an organic solvent. This dissociation step is crucial as it regenerates the valuable chiral amine in its neutral form, ready for downstream coupling reactions in drug synthesis. The entire sequence—from salt formation to crystallization and final liberation—is designed to maximize optical purity while minimizing material loss. The patent highlights that controlling parameters such as temperature gradients during cooling and the stoichiometric ratio of acid to amine is vital for achieving high enantiomeric excess (ee), ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Chiral 3,3-Difluoro-4-Aminopyrrolidine Efficiently
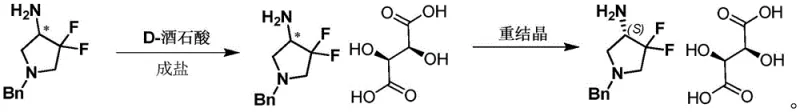
The practical implementation of this resolution technology involves a streamlined three-stage protocol that balances yield and optical purity. The process begins with the dissolution of the racemic starting material in a suitable alcohol solvent, followed by the addition of the chiral resolving agent under heated conditions to ensure complete salt formation. Subsequent controlled cooling induces the crystallization of the target diastereomer, which is then isolated via filtration. Finally, the chiral integrity is unlocked through basic hydrolysis. This standardized approach allows manufacturers to reproducibly access both (S) and (R) enantiomers simply by switching the chirality of the tartaric acid used. For a detailed visualization of a specific embodiment involving N-benzyl-3,3-difluoro-4-aminopyrrolidine, refer to the reaction scheme below which illustrates the high-yield formation of the D-tartrate salt.
- Prepare a salt mixture by reacting racemic 3,3-difluoro-4-aminopyrrolidine derivative with chiral tartaric acid (D- or L-isomer) in a solvent like ethanol.
- Separate the desired diastereomeric salt from the mixture via recrystallization, utilizing solubility differences in solvents such as ethyl acetate or alcohols.
- Dissociate the purified chiral salt in an alkaline solution (e.g., sodium hydroxide) to release the free chiral 3,3-difluoro-4-aminopyrrolidine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this tartaric acid resolution method translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift away from chromatographic purification fundamentally alters the cost structure of producing these high-value intermediates. By replacing expensive chiral columns and specialized instrumentation with commodity chemicals like tartaric acid and standard crystallizers, the manufacturing process becomes significantly more cost-effective. This reduction in capital expenditure and operating costs directly contributes to cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing models in long-term supply agreements. Furthermore, the reliance on widely available reagents mitigates the risk of supply chain disruptions often associated with specialized chromatographic media.
- Cost Reduction in Manufacturing: The elimination of chiral chromatography removes a major cost center from the production budget. Tartaric acid is a bulk commodity chemical with a stable global supply, contrasting sharply with the high cost and limited lifespan of chiral HPLC columns. Additionally, the solvent systems employed (ethanol, ethyl acetate, water) are inexpensive and easily recyclable, further driving down the variable costs per kilogram. This economic efficiency enables the production of high-purity chiral amines at a fraction of the cost of traditional methods, making early-stage drug projects more financially viable.
- Enhanced Supply Chain Reliability: Scalability is the cornerstone of supply chain security. Crystallization processes are inherently easier to scale from gram to ton quantities compared to chromatographic separations, which often face non-linear scaling challenges. This method ensures that as a drug candidate progresses from clinical trials to commercial launch, the supply of the key intermediate can be ramped up seamlessly without requiring entirely new processing technologies. The robustness of the recrystallization step also means that the process is less sensitive to minor fluctuations in raw material quality, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method offers a greener alternative to chromatography. The reduction in solvent volume and the avoidance of silica-based waste streams simplify waste treatment protocols. The use of aqueous workups and common organic solvents facilitates easier solvent recovery and recycling, aligning with modern green chemistry principles. This environmental compatibility not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations, safeguarding the manufacturing license to operate and enhancing the sustainability profile of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of chiral fluorinated pyrrolidines. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the operational nuances of this resolution technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their synthetic routes.
Q: Why is tartaric acid resolution preferred over chiral chromatography for this intermediate?
A: Chiral chromatography is expensive and difficult to scale for industrial production due to equipment limitations and high operational costs. Tartaric acid resolution utilizes inexpensive, readily available resolving agents and standard crystallization equipment, making it far more suitable for large-scale manufacturing.
Q: What specific enantiomers can be obtained using this method?
A: The method allows for the selective production of either the (S)- or (R)-enantiomer. Using D-tartaric acid yields the (S)-3,3-difluoro-4-aminopyrrolidine derivative salt, while L-tartaric acid yields the (R)-isomer salt, providing flexibility for different synthetic pathways.
Q: What are the typical reaction conditions for the resolution step?
A: The resolution typically involves dissolving the racemate and tartaric acid in a solvent like ethanol at elevated temperatures (70-90°C), followed by controlled cooling (e.g., to 0°C or -10°C) to induce crystallization of the less soluble diastereomeric salt.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3,3-Difluoro-4-Aminopyrrolidine Derivative Supplier
The technological advancements detailed in CN113429328A underscore the immense potential of chiral fluorinated pyrrolidines in modern drug discovery, yet realizing this potential requires a manufacturing partner with deep process expertise. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise temperature control and crystallization dynamics required for this resolution process, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to quality assurance, we guarantee the delivery of chiral intermediates that consistently exceed the demanding standards of the global pharmaceutical industry.
We invite R&D leaders and procurement specialists to collaborate with us to optimize their supply chains for these critical building blocks. By leveraging our technical capabilities, you can secure a stable source of high-quality materials while achieving significant economic efficiencies. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next breakthrough in medicinal chemistry.
