Advanced Rare Earth Catalysis for High-Purity Quinazoline-2-thione Intermediates
Advanced Rare Earth Catalysis for High-Purity Quinazoline-2-thione Intermediates
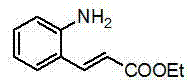
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing heterocyclic scaffolds, particularly those with significant biological activity. A pivotal advancement in this domain is detailed in Chinese Patent CN103232400A, which introduces a novel method for preparing quinazoline-2-thione derivatives. This technology leverages a sophisticated silicon-amino rare earth compound, specifically [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3, to catalyze the addition reaction between ethyl o-aminocinnamate and various isothiocyanates. Unlike conventional synthetic routes that rely on harsh acidic conditions or complex multi-step sequences, this innovation offers a streamlined, solvent-free approach that operates under mild thermal conditions. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, providing a robust platform for generating diverse libraries of bioactive quinazoline cores.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazoline-2-thiones has been fraught with significant technical and safety challenges that hinder large-scale adoption. Traditional methodologies often depend on the condensation of ortho-amino aromatic ketones or alcohols with isothiocyanates in the presence of strong protonic acids, such as hydrochloric acid, and reducing agents. For instance, prior art describes reactions requiring temperatures around 65°C for extended periods, yet yielding mediocre results, often hovering around 31%. Furthermore, the necessity for strong acids introduces severe safety hazards, including corrosion risks and the generation of hazardous waste streams that complicate environmental compliance. Alternative routes involving the cyclization of thiourea intermediates derived from acrylic acid derivatives often necessitate complicated starting materials and reflux conditions in methanol, which increases both the raw material costs and the complexity of the technological process. These legacy methods struggle with poor chemoselectivity, leading to difficult purification profiles and inconsistent batch-to-batch quality.
The Novel Approach
In stark contrast, the methodology disclosed in CN103232400A revolutionizes the synthesis landscape by employing a highly active silicon-amino rare earth catalyst system. This novel approach eliminates the need for volatile organic solvents, conducting the reaction under neat (solvent-free) conditions, which drastically reduces the environmental footprint and simplifies the reactor setup. The reaction proceeds efficiently at moderate temperatures ranging from room temperature to 80°C, depending on the specific isothiocyanate substrate, with reaction times typically between 3 to 48 hours. By utilizing lanthanum, samarium, neodymium, or ytterbium-based catalysts, the process achieves exceptional chemoselectivity, ensuring that the addition reaction occurs precisely at the desired sites without side reactions. This results in substantially higher yields, with some embodiments reporting conversion rates as high as 98%, thereby maximizing raw material utilization and minimizing waste generation compared to the inefficient acid-catalyzed predecessors.
Mechanistic Insights into Silicon-Amino Rare Earth Catalyzed Cyclization
The core of this technological breakthrough lies in the unique electronic and steric properties of the silicon-amino rare earth complex, [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3. In this catalytic cycle, the rare earth metal center (Ln), particularly when Ln is Lanthanum (La), acts as a potent Lewis acid. It coordinates with the nitrogen or sulfur atoms of the isothiocyanate and potentially the carbonyl oxygen of the ester group in ethyl o-aminocinnamate, thereby activating the electrophilic centers for nucleophilic attack. The bulky trimethylsilylamino ligands provide a protective steric environment that prevents catalyst deactivation while allowing substrate access. The lithium chloride bridge further stabilizes the complex structure, facilitating a cooperative catalytic effect that lowers the activation energy barrier for the cyclization step. This precise activation mechanism allows the reaction to proceed smoothly without the need for external proton sources or harsh thermal activation, preserving sensitive functional groups on the aromatic rings.
![Structure of the silicon-amino rare earth catalyst [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3](/insights/img/quinazoline-2-thione-rare-earth-catalysis-pharma-supplier-20260308044733-02.png)
From an impurity control perspective, this catalytic system offers distinct advantages due to its high specificity. Traditional acid-catalyzed methods often promote polymerization of the acrylic double bond or hydrolysis of the ester moiety, leading to complex impurity profiles that are difficult to separate. However, the mild nature of the rare earth catalyst minimizes these side reactions. The reaction pathway is directed specifically towards the formation of the quinazoline ring through a concerted addition-cyclization mechanism. Furthermore, the catalyst loading is optimized to be between 1% and 10% molar equivalent, with 2.5% being preferred, which strikes a balance between reaction rate and ease of removal. Post-reaction workup involves simple aqueous quenching and extraction, avoiding the need for complex metal scavenging resins often required with transition metal catalysts, thus ensuring the final API intermediate meets stringent purity specifications with minimal heavy metal residue.
How to Synthesize Quinazoline-2-thione Efficiently
To implement this advanced synthesis route effectively, strict adherence to anhydrous and oxygen-free conditions is paramount to maintain the integrity of the rare earth catalyst. The process begins with the preparation of the catalyst, followed by the direct mixing of substrates in a controlled inert atmosphere. The reaction parameters, such as temperature and time, must be tuned based on the electronic nature of the isothiocyanate substituent; for example, aryl isothiocyanates typically react faster at 50°C, while alkyl variants may require slightly higher temperatures or longer durations. The following guide outlines the standardized operational procedure derived from the patent embodiments to ensure reproducible high-yield outcomes suitable for pilot and commercial scale operations.
- Prepare the reaction vessel under anhydrous and oxygen-free conditions, preferably under an argon atmosphere, to ensure catalyst stability.
- Mix ethyl o-aminocinnamate and the selected isothiocyanate (1.0 to 1.2 equivalents) with the silicon-amino rare earth catalyst (1% to 10% molar loading).
- Stir the mixture at temperatures between 50°C and 80°C for 3 to 48 hours, then quench with aqueous acetic acid and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rare earth catalyzed process translates into tangible strategic benefits regarding cost structure and operational reliability. The elimination of reaction solvents is a primary driver for cost reduction, as it removes the expenses associated with purchasing, storing, recovering, and disposing of large volumes of organic solvents. Additionally, the high atom economy and superior yields mean that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS). The simplicity of the workup procedure, which avoids complex distillation or specialized filtration steps, further reduces utility consumption and labor hours, making the overall manufacturing process significantly more lean and efficient compared to legacy technologies.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the drastic simplification of the process workflow. By operating under solvent-free conditions, the facility saves substantially on solvent procurement and waste treatment costs, which are often hidden but significant burdens in fine chemical manufacturing. The catalyst itself, while sophisticated, is used in very low loadings (as low as 1-2.5%), and its high turnover frequency ensures that the cost per mole of product remains competitive. Moreover, the avoidance of corrosive acids like hydrochloric acid reduces equipment maintenance costs and extends the lifespan of reactor vessels, contributing to long-term capital expenditure savings.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the starting materials—ethyl o-aminocinnamate and various isothiocyanates—are commercially available and widely produced commodity chemicals, ensuring a stable and resilient supply chain. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or mixing efficiency, leading to consistent batch quality and reliable delivery schedules. This reliability is crucial for downstream pharmaceutical customers who require uninterrupted supply of high-quality intermediates to maintain their own production timelines and regulatory filings.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is inherently safer and more straightforward due to the absence of flammable solvents and hazardous acidic reagents. The solvent-free nature significantly reduces the risk of thermal runaway and fire hazards, simplifying safety assessments and regulatory approvals for plant expansion. Furthermore, the reduced generation of chemical waste aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations, positioning manufacturers as responsible partners in the global supply chain and mitigating the risk of future compliance-related shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rare earth catalyzed synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and product quality attributes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using rare earth catalysts over traditional acid catalysis for quinazoline synthesis?
A: Traditional methods often require harsh protonic acids like hydrochloric acid and high temperatures, leading to safety hazards and lower yields (around 31%). The rare earth catalyzed method operates under mild, solvent-free conditions with significantly higher chemoselectivity and yields up to 98%.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process is highly scalable because it eliminates the need for reaction solvents, which simplifies downstream processing and waste treatment. The catalyst loading is low (1-10%), and the workup involves standard extraction and chromatography techniques familiar to process chemists.
Q: What is the substrate scope for the isothiocyanate component?
A: The catalyst system demonstrates excellent universality, accommodating a wide range of aryl isothiocyanates (including electron-withdrawing and electron-donating groups like nitro, chloro, and methoxy) as well as alkyl isothiocyanates, maintaining high efficiency across diverse substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline-2-thione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the rare earth catalyzed synthesis described in CN103232400A for producing high-value quinazoline-2-thione intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this laboratory-scale innovation are fully realized in an industrial setting. Our state-of-the-art facilities are equipped to handle air-sensitive catalytic reactions under strict inert atmospheres, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required by global pharmaceutical clients.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this solvent-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our quinazoline derivatives and to discuss detailed route feasibility assessments for your target molecules.
